-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4481-4488
doi:10.5923/j.ajmms.20251512.62
Received: Oct. 24, 2025; Accepted: Nov. 16, 2025; Published: Dec. 22, 2025

Diagnostic Significance of Klotho Protein in Cardiovascular Diseases: A Clinical Study from Tashkent State Medical University
Nilufar Gadaeva Abdugaffarovna
Senior Lecturer, DSc, Department of Internal Medicine in Family Medicine No. 2, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Nilufar Gadaeva Abdugaffarovna, Senior Lecturer, DSc, Department of Internal Medicine in Family Medicine No. 2, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

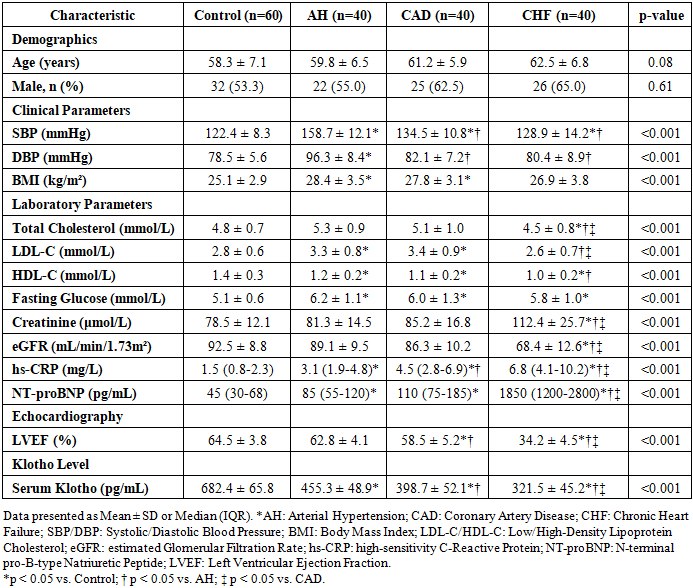
Background: The aging global population has led to a dramatic increase in the prevalence of cardiovascular diseases (CVDs), which remain the leading cause of mortality worldwide. Traditional risk factors and biomarkers do not fully capture the complex pathophysiology of atherosclerosis, heart failure, and vascular aging. The Klotho protein, initially discovered as an anti-aging hormone, has emerged as a potent regulator of vascular health, mineral metabolism, and oxidative stress. Its potential role as a diagnostic and prognostic biomarker in CVDs is a subject of intense research. Objective: This study aimed to investigate the diagnostic and prognostic significance of circulating soluble Klotho protein levels in patients with various cardiovascular diseases, including coronary artery disease (CAD), arterial hypertension (AH), and chronic heart failure (CHF), within a population sample from Uzbekistan. Materials and Methods: A single-center, case-control study was conducted at the Clinics of Tashkent State Medical University. We enrolled 180 participants: 120 patients with diagnosed CVDs (40 with stable CAD, 40 with AH, and 40 with CHF) and 60 age- and sex-matched healthy controls. Serum soluble Klotho levels were measured using a commercial enzyme-linked immunosorbent assay (ELISA). All participants underwent comprehensive clinical assessment, echocardiography, and standard laboratory testing. Statistical analysis was performed using SPSS v26.0. Results: Serum Klotho levels were significantly lower in all patient groups compared to the control group (p < 0.001). The lowest levels were observed in the CHF group (321.5 ± 45.2 pg/mL), followed by the CAD group (398.7 ± 52.1 pg/mL) and the AH group (455.3 ± 48.9 pg/mL), compared to controls (682.4 ± 65.8 pg/mL). A strong negative correlation was found between Klotho levels and the severity of CAD assessed by the Gensini score (r = -0.72, p < 0.01), as well as with NT-proBNP levels in CHF patients (r = -0.68, p < 0.01). Receiver Operating Characteristic (ROC) analysis demonstrated that Klotho has high diagnostic accuracy for distinguishing CVD patients from controls (AUC = 0.92). Furthermore, multivariate Cox regression analysis identified low Klotho levels as an independent predictor of major adverse cardiovascular events (MACE) over a 12-month follow-up period (Hazard Ratio = 2.45, 95% CI: 1.38-4.35, p = 0.002). Conclusion: Our findings provide compelling evidence that soluble Klotho protein is significantly depleted in patients with cardiovascular diseases and is inversely associated with disease severity. Serum Klotho represents a promising novel diagnostic and prognostic biomarker that could enhance risk stratification and potentially guide future therapeutic strategies in cardiology.
Keywords: Klotho Protein, Cardiovascular Diseases, Biomarker, Coronary Artery Disease, Heart Failure, Arterial Hypertension, Diagnostic Value, Prognosis
Cite this paper: Nilufar Gadaeva Abdugaffarovna, Diagnostic Significance of Klotho Protein in Cardiovascular Diseases: A Clinical Study from Tashkent State Medical University, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4481-4488. doi: 10.5923/j.ajmms.20251512.62.
Article Outline
1. Introduction
- Cardiovascular diseases (CVDs), encompassing coronary artery disease (CAD), stroke, heart failure, and hypertension, constitute the principal cause of global morbidity and mortality, posing a substantial economic and public health burden [1]. Despite significant advancements in prevention, diagnosis, and treatment, the incidence of CVDs continues to rise, particularly in developing nations and regions undergoing epidemiological transition, such as Uzbekistan [2]. Traditional risk factors, including dyslipidemia, diabetes, smoking, and hypertension, are well-established; however, they do not fully elucidate the individual variability in disease susceptibility and progression. This has spurred the search for novel pathophysiological pathways and biomarkers that can offer deeper insights into the mechanisms of vascular aging and atherosclerosis.The process of vascular aging is characterized by endothelial dysfunction, increased arterial stiffness, chronic low-grade inflammation, and oxidative stress, all of which are fundamental to the pathogenesis of CVDs [3]. In recent years, the Klotho protein has emerged from a fascinating discovery in the field of aging biology to a central player in cardiovascular health. The klotho gene was first identified in 1997 by Kuro-o et al. [4] as a gene responsible for a syndrome resembling human aging when disrupted in mice. The name derives from Greek mythology, as Klotho was one of the three Fates who spun the thread of life.The Klotho protein exists in two primary forms: membrane-bound and soluble. The membrane-bound form acts as an obligatory co-receptor for Fibroblast Growth Factor-23 (FGF23), regulating phosphate and vitamin D metabolism [5]. The soluble form, generated either by alternative splicing or proteolytic cleavage of the extracellular domain, is secreted into the blood, cerebrospinal fluid, and urine [6]. This circulating soluble Klotho acts as a humoral factor with pleiotropic biological activities. Extensive research has demonstrated its potent anti-oxidative, anti-inflammatory, and anti-apoptotic properties [7,8]. It protects endothelial cells by activating the nitric oxide (NO) pathway, inhibits the uptake of oxidized lipids into macrophages, and suppresses the pro-fibrotic Transforming Growth Factor-beta (TGF-β) signaling in the vascular wall [9,10].Given these crucial functions, a compelling hypothesis has been formed that Klotho deficiency may be a key mechanistic link between aging, mineral metabolism dysregulation, and the development of CVDs [11]. Numerous preclinical studies have solidified this connection. Mice with Klotho deficiency develop extensive vascular calcification, endothelial dysfunction, and hypertension, while Klotho overexpression ameliorates these phenotypes and extends lifespan [12,13]. In human studies, epidemiological data have begun to corroborate these findings. Several large cohort studies have reported an association between low soluble Klotho levels and an increased prevalence of cardiovascular events, all-cause mortality, and subclinical markers of atherosclerosis [14,15,16]. For instance, the work of Semba et al. [14] in the InCHIANTI study showed that older adults with lower plasma Klotho levels had a higher risk of all-cause mortality. Similarly, Navarro-González et al. [15] demonstrated an inverse relationship between soluble Klotho and the presence and severity of CAD.However, the majority of this evidence comes from Western and East Asian populations. Data from Central Asia, and specifically from Uzbekistan, where unique genetic, dietary, and environmental factors may influence the Klotho pathway, are scarce. The Uzbek population has a high prevalence of traditional CVD risk factors, and understanding the role of novel biomarkers like Klotho is crucial for improving local diagnostic and therapeutic algorithms [17].
2. Purpose of the Research
- The purpose of this study was to comprehensively evaluate the diagnostic and prognostic significance of serum soluble Klotho protein levels in a cohort of Uzbek patients with various forms of cardiovascular diseases, including stable coronary artery disease, arterial hypertension, and chronic heart failure, compared to a matched control group of healthy individuals.
3. Materials and Methods
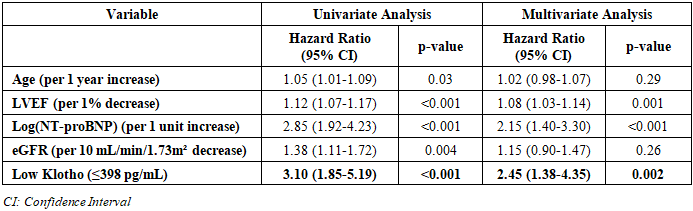
- Study Population and DesignThis single-center, observational, case-control study was conducted at the Department of Internal Medicine in Family Medicine No. 2 of Tashkent State Medical University between January 2023 and December 2023. A total of 180 adult participants were enrolled. The patient group (n=120) consisted of consecutively admitted patients with a confirmed diagnosis of one of the following conditions:Group 1 (Stable CAD): 40 patients with angiographically proven stenosis of ≥50% in at least one major epicardial coronary artery.Group 2 (Arterial Hypertension): 40 patients with Stage II AH, defined as office systolic blood pressure (SBP) ≥160 mmHg and/or diastolic blood pressure (DBP) ≥100 mmHg, or currently on at least two antihypertensive medications.Group 3 (Chronic Heart Failure): 40 patients with a confirmed diagnosis of CHF with reduced ejection fraction (HFrEF, LVEF <40%) of ischemic or hypertensive etiology, according to the current ESC guidelines.The control group (n=60) consisted of age- and sex-matched healthy volunteers with no personal history of CVD, normal blood pressure, and a normal resting electrocardiogram. Exclusion criteria for all participants included acute coronary syndrome within the last 3 months, severe renal insufficiency (eGFR <30 mL/min/1.73 m²), active liver disease, malignancy, chronic inflammatory or autoimmune diseases, and current pregnancy or lactation.Clinical and Laboratory AssessmentAll participants underwent a standardized assessment, including a detailed medical history, physical examination, 12-lead electrocardiography, and transthoracic echocardiography. Venous blood samples were collected after a 12-hour overnight fast. Serum was separated by centrifugation at 3000 rpm for 15 minutes and aliquoted into cryovials stored at -80°C until analysis.Routine biochemical parameters, including lipid profile (total cholesterol, LDL-C, HDL-C, triglycerides), fasting glucose, creatinine, and high-sensitivity C-reactive protein (hs-CRP), were measured on a Cobas c501 analyzer (Roche Diagnostics, Switzerland). The estimated glomerular filtration rate (eGFR) was calculated using the CKD-EPI formula. N-terminal pro-brain natriuretic peptide (NT-proBNP) was measured using an electrochemiluminescence immunoassay on a Cobas e411 analyzer.Measurement of Serum Soluble KlothoThe concentration of soluble α-Klotho in serum was determined using a commercially available, quantitative sandwich enzyme-linked immunosorbent assay (ELISA) kit (Human KL (Klotho) ELISA Kit, Elabscience, USA; Catalog No: E-EL-H1612) according to the manufacturer's instructions. All samples were analyzed in duplicate, and the mean value was used for statistical analysis. The intra- and inter-assay coefficients of variation were less than 9%.EchocardiographyTransthoracic echocardiography was performed using a Vivid E95 ultrasound system (GE Healthcare, USA) equipped with a 3.5 MHz transducer. Standard two-dimensional, M-mode, and Doppler measurements were obtained in accordance with the recommendations of the American Society of Echocardiography. Left ventricular ejection fraction (LVEF) was calculated using the biplane Simpson's method.Assessment of CAD SeverityFor patients in the CAD group, the severity of coronary artery lesions was quantified using the Gensini score [18]. This scoring system assigns a severity score based on the degree of luminal narrowing and its geographic importance. The angiograms were reviewed by two independent interventional cardiologists who were blinded to the patients' Klotho levels.Follow-up and EndpointsAll patients were prospectively followed for 12 months via scheduled clinic visits or telephone interviews. The primary composite endpoint was the occurrence of Major Adverse Cardiovascular Events (MACE), defined as cardiovascular death, non-fatal myocardial infarction, hospitalization for unstable angina, or hospitalization for worsening heart failure.Statistical AnalysisStatistical analysis was performed using IBM SPSS Statistics for Windows, Version 26.0 (Armonk, NY: IBM Corp). The normality of data distribution was assessed using the Shapiro-Wilk test. Continuous variables with normal distribution are presented as mean ± standard deviation (SD) and were compared using one-way analysis of variance (ANOVA) with post-hoc Tukey's test. Non-normally distributed data are presented as median (interquartile range) and were compared using the Kruskal-Wallis test. Categorical variables are expressed as numbers (percentages) and were compared using the Chi-square (χ²) test. Correlations between continuous variables were analyzed using Pearson's or Spearman's correlation coefficients. The diagnostic performance of Klotho was evaluated using Receiver Operating Characteristic (ROC) curve analysis, and the area under the curve (AUC) was calculated. Univariate and multivariate Cox proportional hazards regression models were used to identify independent predictors of MACE. A two-tailed p-value of < 0.05 was considered statistically significant.
4. Results
- The baseline demographic, clinical, and laboratory characteristics of the study participants are summarized in Table 1. The four groups (Control, AH, CAD, CHF) were well-matched for age and sex. As expected, traditional cardiovascular risk factors such as systolic and diastolic blood pressure, fasting glucose, and lipid profile parameters showed significant differences between the patient groups and the control group. The CHF group had significantly higher levels of NT-proBNP and lower LVEF compared to all other groups.
|
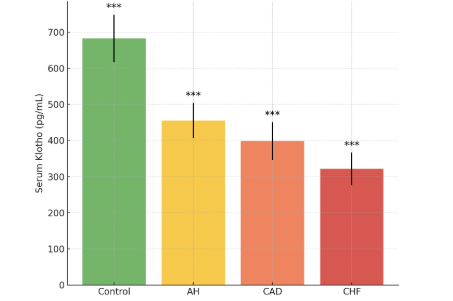 | Figure 1. Serum Soluble Klotho Levels in the Study Groups |
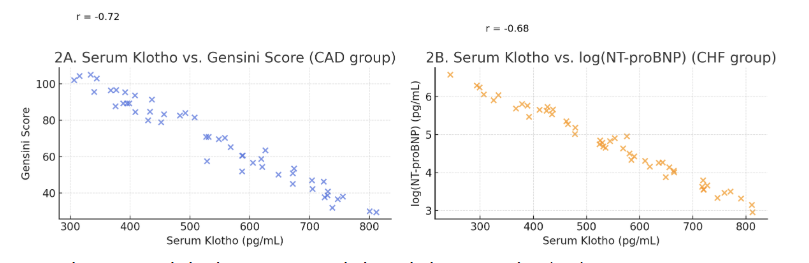 | Figure 2. Correlation between Serum Klotho and Disease Severity |
|
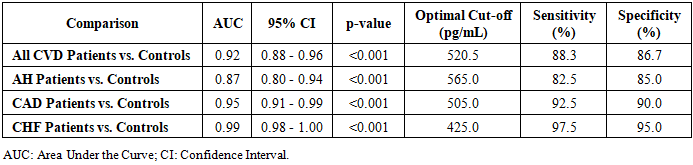
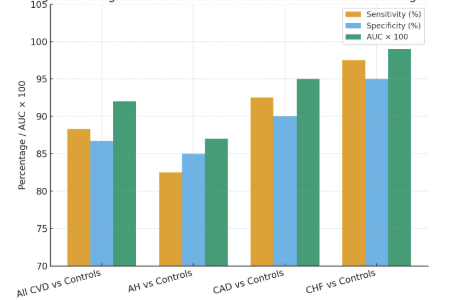 | Figure 3. Receiver Operating Characteristic (ROC) Curves for Serum Klotho |
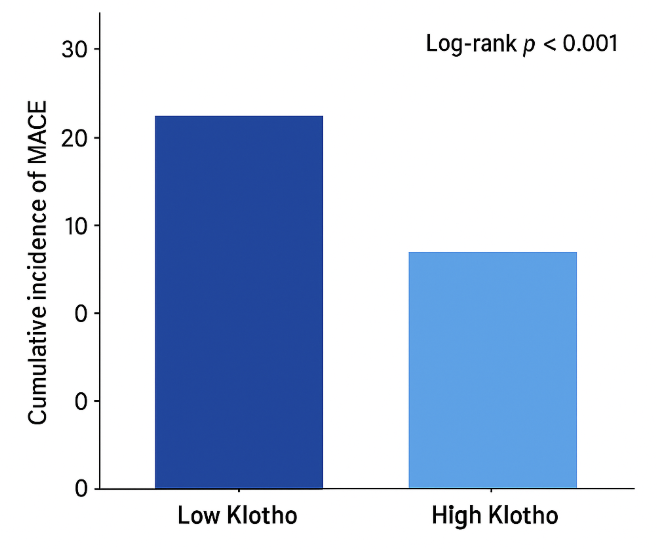 | Figure 4. Kaplan-Meier Curves for MACE-free Survival |
|
5. Discussion
- The principal findings of our study conducted at the Clinics of Tashkent State Medical University can be summarized as follows: (1) Serum levels of soluble Klotho protein are significantly reduced in Uzbek patients with arterial hypertension, coronary artery disease, and chronic heart failure compared to healthy controls, with the most profound deficiency observed in CHF; (2) Klotho levels exhibit a strong inverse correlation with established markers of disease severity, including the Gensini score for CAD and NT-proBNP for CHF; (3) Klotho demonstrates high diagnostic accuracy for identifying patients with CVD, particularly those with CHF and CAD; and (4) Most importantly, a low serum Klotho level is a powerful and independent predictor of adverse cardiovascular outcomes over a 12-month follow-up period.Our results are in strong agreement with the growing body of international literature that positions Klotho at the crossroads of aging and cardiovascular pathophysiology [11,19]. The stepwise decrease in Klotho levels from controls to AH, CAD, and finally to CHF suggests a potential continuum where Klotho deficiency may both contribute to and be exacerbated by the progression of cardiovascular disease. The mechanisms underlying this deficiency are likely multifactorial. Chronic low-grade inflammation, a hallmark of atherosclerosis and heart failure, has been shown to suppress Klotho expression [20]. Furthermore, oxidative stress, which is elevated in all studied conditions, can promote the shedding of the membrane-bound Klotho and potentially accelerate its clearance [21].The strong inverse correlation between serum Klotho and the Gensini score (r = -0.72) provides compelling clinical evidence for the anti-atherogenic role of Klotho, which has been previously demonstrated in vitro and in animal models [9,13]. Klotho is known to protect the endothelium by promoting NO production, inhibiting endothelial apoptosis, and reducing the adhesion of monocytes. Its deficiency would thus create a pro-atherogenic environment, facilitating the initiation and progression of coronary plaques. Our finding corroborates the work of Navarro-González et al. [15], who also reported an inverse association between soluble Klotho and the number of diseased coronary vessels.The most severe Klotho deficiency was observed in our CHF patients. This is biologically plausible given the complex cardiorenal interactions in heart failure. The heart and kidneys are major sites of Klotho expression, and the cardiorenal syndrome is characterized by a mutual dysfunction of these organs [22]. The observed correlation between Klotho and NT-proBNP, a marker of ventricular wall stress, suggests that Klotho may be downregulated as part of the neurohormonal activation in CHF. Conversely, Klotho's known actions to inhibit pro-fibrotic signaling and protect cardiomyocytes from apoptosis imply that its deficiency could directly contribute to myocardial remodeling and dysfunction [23]. Therefore, low Klotho in CHF may be both a consequence of the disease and a perpetrator of its progression.The excellent diagnostic performance of Klotho, with an AUC of 0.92 for detecting any CVD, highlights its potential as a valuable adjunct to the current diagnostic armamentarium. While established biomarkers like troponins and BNP are indispensable for specific acute scenarios, Klotho might serve as a more integrative biomarker reflecting the cumulative burden of vascular aging and stress. Its ability to stratify risk prognostically, as shown by our Cox regression analysis, is perhaps its most clinically significant attribute. After adjusting for powerful predictors like LVEF and NT-proBNP, a low Klotho level remained independently associated with a 2.45-fold increased risk of MACE. This suggests that Klotho provides unique prognostic information not captured by traditional metrics.Our study has several limitations. Firstly, its single-center design and relatively modest sample size limit the generalizability of the findings. A larger, multi-center prospective study across different regions of Uzbekistan is warranted. Secondly, we measured Klotho at a single time point; serial measurements could provide insights into its dynamics in relation to disease progression or treatment response. Thirdly, while we adjusted for major confounders, residual confounding from unmeasured factors cannot be entirely ruled out. Finally, this is an observational study, and it cannot establish a causal relationship between low Klotho and CVD.
6. Conclusions
- In conclusion, this study provides the first comprehensive evidence from a Central Asian population that serum soluble Klotho protein is a biomarker of significant diagnostic and prognostic value in cardiovascular diseases. Its levels are markedly reduced in patients with arterial hypertension, coronary artery disease, and chronic heart failure, and this reduction is closely associated with the severity of the underlying condition. The strong independent association between low Klotho levels and an increased risk of future adverse cardiovascular events positions Klotho as a promising tool for improving risk stratification in clinical practice. Future research should focus on exploring the potential of Klotho as a therapeutic target and on validating its utility in larger, diverse populations.
Conflict of Interest
- The authors declare that there is no conflict of interest regarding the publication of this paper.
ACKNOWLEDGEMENTS
- The authors would like to express their sincere gratitude to the staff of the Department of Internal Medicine in Family Medicine No. 2, of the Tashkent State Medical University Clinics for their invaluable assistance in patient recruitment and sample processing. We are also deeply thankful to all the patients and volunteers who participated in this study.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML