-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4452-4455
doi:10.5923/j.ajmms.20251512.55
Received: Nov. 20, 2025; Accepted: Dec. 10, 2025; Published: Dec. 12, 2025

Description of the Results of a Comparative Study of Immunoglobulin Content in the Serum of Women with Pre-Cervical Tumor
Nazarov B. B., Karimova N. N.
Bukhara State Medical Institute named after Abu Ali ibn Sino, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The study examined changes in serum immunoglobulins in women with precancerous cervical lesions. Cervical cancer in women is characterized by a severe course, poor treatment efficacy, and often fatal outcome. Precancerous cervical diseases have been proven to be among the main risk factors for this medical and socio-economic problem [1]. Therefore, it is advisable to have biological markers and clinical laboratory criteria that clearly identify the disease. Taking into account the above, this study aimed to conduct a comparative investigation of the serum concentrations of humoral immune factors in women with precancerous cervical conditions.
Keywords: Immunoglobulin, Humoral immunity, Biological marker, Humoral factor, Plasma cells
Cite this paper: Nazarov B. B., Karimova N. N., Description of the Results of a Comparative Study of Immunoglobulin Content in the Serum of Women with Pre-Cervical Tumor, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4452-4455. doi: 10.5923/j.ajmms.20251512.55.
1. Introduction
- Cervical cancer in women is distinguished by a severe course, low treatment success, and frequent fatal outcomes. It has been shown that precancerous conditions of the cervix are among the key risk factors for this medical and socio-economic issue. Thus, early detection and uncomplicated treatment of precancerous cervical conditions are of great importance. A variety of diagnostic methods are recommended and must be under continuous monitoring. However, one of the biggest challenges is initial detection, because women who consider themselves healthy and have no pain or discomfort symptoms are unlikely to seek medical help. This situation is also influenced by cultural and social factors; the problem of timely medical consultation remains a leading issue [2].In many cases, precancerous cervical lesions are detected incidentally during a primary medical visit for another issue. Therefore, it is reasonable to develop and utilize biological markers and clinical laboratory criteria that would definitively diagnose this condition [3].The immune system is one of the most sensitive systems in the body with respect to early pathological changes. Its immunocompetent cells and humoral immune factors respond to ongoing asymptomatic, subclinical conditions with quantitative and qualitative changes. It has been shown that shifts in their quantities and balance can indicate the presence of pre-disease states in the organism [4].Taking the above into account, the objective of this research was set to comparatively study the serum concentrations of humoral immune factors in women with precancerous cervical lesions.
2. Materials and Methods
- A total of 252 women of various ages were enrolled in the clinical study. Immunological investigations were performed on 76 of these patients; 15 healthy women with no detected cervical pre-cancerous or cancerous conditions were included as the control group.All participants were divided into four representative study groups:- Group 1 (n=23): Women diagnosed with precancerous cervical lesions (dysplastic process) who had not received any treatment.- Group 2 (n=26): Women with precancerous cervical lesions (dysplastic process) who received conservative treatment.- Group 3 (n=26): Women with early-stage cervical cancer (stage 0–I) who underwent radiation and chemotherapy.- Group 4 (Control, n=15): Healthy women in whom no precancerous or cancerous cervical lesions were observed during the study period.The serum concentration of IgA was determined using ELISA test kits from Vector Best (Novosibirsk, Russia). IgM, IgG, and IgE were measured using ELISA test kits from Xema (Moscow, Russia). Enzyme-linked immunosorbent assays were carried out on a MR-96A analyzer (Mindray Co. Ltd, China) manufactured in 2022.The obtained data were statistically processed using standard methods of variance statistics with the Excel software. The arithmetic mean (M) and standard error of mean (m) were calculated. Differences were evaluated for statistical significance using the Fisher–Student criterion (P).
3. Results
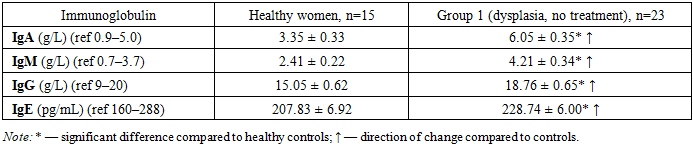
- Serum levels of all measured immunoglobulins in the patient groups were significantly higher than in healthy women (P<0.05 to P<0.001), although all values remained within the upper reference ranges (P>0.05). This suggests that, while these pathologies induce significant changes in immunoglobulin concentrations, the overall immune response activation remains relatively low. Nevertheless, the findings indicate that these immune proteins can respond to even minimal antigenic stimulation.
|
|
|
4. Discussion
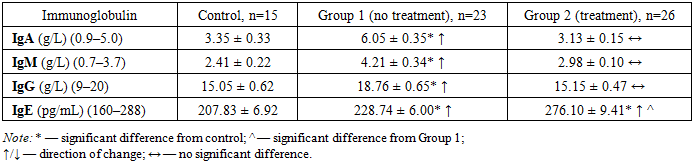
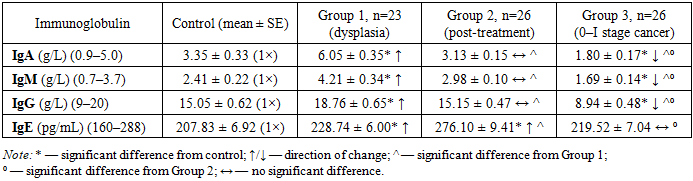
- In women with precancerous cervical conditions (dysplasia), serum immunoglobulin levels were significantly elevated compared to healthy controls (P<0.05–P<0.001), with the same direction of increase across all classes. However, the intensity of change differed: for example, the increase in IgG was less pronounced than that of IgA and IgM. Specifically, IgA increased 1.81-fold, IgM 1.75-fold, and IgG 1.25-fold in patients relative to controls (all P<0.05–P<0.001).In untreated patients with dysplasia (Group 1), IgA, IgM, IgG, and IgE all rose significantly (same direction, varying degrees). In treated patients (Group 2), these markers significantly decreased (P<0.05) to reach levels comparable to healthy women, with no significant differences remaining (except IgE). IgE was the only exception, continuing to rise even after therapy, remaining significantly higher than in both healthy and untreated women.These quantitative immunoglobulin changes and imbalances are likely linked to inflammation associated with the dysplastic process and immune system activity. After standard treatment, the marked decrease in immunoglobulin levels correlates with therapeutic success; the persistent elevation of IgE may be explained by lack of antihistamine therapy and the increasing allergic component of the inflammatory process.Thus, significant increases in IgA, IgM, IgG, and IgE (1.10–1.81-fold) in dysplasia can serve as diagnostic and treatment-response indicators, whereas their decline following radiation and chemotherapy (up to 1.43–1.86-fold reductions relative to controls) can serve as prognostic markers of treatment efficacy. We propose using these changes together with clinical symptoms and pre-pathological diagnostics as combined diagnostic and prognostic biomarkers, which is a novel recommendation from our study.
5. Conclusions
- 1. In women with precancerous cervical lesions, serum immunoglobulin levels showed a uniform trend of increase, although the degree of change varied. Specifically, IgA was 1.81 times higher than normal, IgM 1.75 times, and IgG 1.25 times in patients (P<0.05–P<0.001).2. In untreated women (dysplasia), IgA, IgM, IgG, and IgE increased significantly, whereas in treated women these indicators significantly decreased to reach control levels, with no significant differences from controls. The exception was IgE, which continued to rise after treatment (remaining significantly higher than in healthy and untreated women).3. It was shown that the significant increases of IgA, IgM, IgG, and IgE (1.10–1.81-fold) in precancerous cervical conditions can be used as diagnostic and treatment-effect markers, while their decreases during radiation and chemotherapy (up to 1.43–1.86-fold lower than normal) serve as prognostic markers. Using these together with clinical symptoms and pre-cancer diagnostics as combined diagnostic and prognostic biomarkers is recommended for the first time.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML