-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4422-4430
doi:10.5923/j.ajmms.20251512.50
Received: Nov. 12, 2025; Accepted: Dec. 2, 2025; Published: Dec. 12, 2025

Clinical and Genetic Parallels of Uterine Fibroids
Dinara Irnazarova1, Dilchekhra Yuldasheva1, Akmal Irnazarov2
1Department of Obstetrics and Gynecology, Tashkent State Medical University, Tashkent, Uzbekistan
2Department of Faculty and Hospital Surgery №1, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Dinara Irnazarova, Department of Obstetrics and Gynecology, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Uterine fibroids are the most common neoplasm in women in gynecological practice, causing significant morbidity and impacting negatively on a woman's quality of life. Despite its widespread prevalence, uterine fibroids remain a relatively understudied disease. At the Multidisciplinary Clinic of the Tashkent Medical Academy, we studied 200 women of reproductive and premenopausal age in 2021-2023. All women underwent a general clinical and molecular genetic examination. The polymorphic marker of the estrogen and progesterone gene in the women studied is associated with the development of uterine fibroids.
Keywords: Uterine fibroids (UF), Genetic polymorphism, Estrogen receptor genes (ER), Progesterone receptor genes (PR)
Cite this paper: Dinara Irnazarova, Dilchekhra Yuldasheva, Akmal Irnazarov, Clinical and Genetic Parallels of Uterine Fibroids, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4422-4430. doi: 10.5923/j.ajmms.20251512.50.
Article Outline
1. Introduction
- Uterine fibroids (UF), also called uterine leiomyomas, are benign smooth muscle tumors of the uterus affecting reproductive and perimenopausal women. [1]. UFs are the most common solid and symptomatic tumors in women and are the main indication for hysterectomy, the definitive and effective radical surgical treatment for leiomyomas. However, many women have future reproductive goals and wish to preserve the uterus [2]. The prevalence of UF ranges from 12-25 to 70-80% of all gynecological diseases, reaching a maximum in late reproductive and premenopausal age [3].Despite the numerous studies conducted in this field, a consensus among scientists regarding the pathogenesis of UF remains elusive. The etiology of UF remains a subject of considerable debate, with a multitude of factors contributing to its development. These factors are believed to interact in a complex manner, influenced by both genetic and epigenetic elements. Despite their benign nature, uterine leiomyomas are symptomatic and cause various symptoms such as abdominal pain, heavy menstrual bleeding, pelvic pain and infertility [4,5]. The growth of these tumors is positively regulated by the hormones estrogen and progesterone, so the tumors usually decrease in size after the onset of menopause [5]. Myomas are comprised of disordered bundles of smooth muscle cells, accompanied by an abundance of various ECM proteins, including collagens, fibronectins and laminins [6]. The etiology of leiomyomas is considered to be monoclonal tumors resulting from the transformation of a solitary myometrial progenitor cell, influenced by various genetic and epigenetic alterations such as sex steroid hormones, cytokines, and growth factors [4,7]. Despite the advancement of research in this field, the existing knowledge concerning the pathophysiology of leiomyoma remains limited and our understanding of the underlying causes of the disease remains deficient. A recent meta-analysis of epidemiological and sequencing data has indicated that mutations in mediator complex subunit 12 (MED12) may be more prevalent in leiomyomas affecting black women. A more profound comprehension of the protein dysregulation associated with these leiomyoma subtypes may facilitate the identification of non-surgical targeted therapeutic strategies in future studies.The function of progesterone signaling in the development and growth of UFs is a pivotal area of investigation. The role of progesterone in promoting UF cell proliferation is well-documented, with the activation of several signaling pathways at both the genetic and epigenetic level having been demonstrated, including the Akt/MEK/ERK pathway. A further observation of particular relevance in the present study is that there is significantly higher progesterone receptor expression in UF tissue samples than in normal myometrium tissue samples [9]. The underlying causes of fibroids are not yet fully established, however, a mounting body of evidence from epidemiological, clinical, and experimental sources support a pivotal role for ovarian steroid hormones in the growth and pathogenesis of UF [10].It has been determined that certain factors have the capacity to influence the molecular and genetic processes of proliferation, apoptosis, hypertrophy and hyperplasia of UF. Among these factors, the most significant are estrogens, progesterone, and their respective receptors [11,12]. The molecular mechanism through which estrogen stimulates myoma growth involves the estrogen receptor genes (ER), which is induced by estrogen through the expression of progesterone receptor genes (PR), thereby allowing leiomyomas to respond to progesterone [12]. Conversely, progesterone has been observed to induce leiomyomas cell growth by means of the expression of a specific set of genes that regulate the processes of apoptosis and proliferation [13]. The role that estrogens play is multifaceted, and involves progesterone, growth factors, genetic and epigenetic factors [14]. The combined action of these two sex hormones is a prerequisite for UF growth, i.e. the stimulatory effects of estrogen and progesterone are complementary. The action of estrogen is known to form the conditions for progesterone-mediated growth in target tissues [15,16]. Upon internalization, the ER, facilitating its entry into the nucleus and subsequent gene expression, activates the hormone estrogen (estradiol) within the cytoplasm of the cell. In the nucleus, the receptor complex then stimulates the expression of estrogen-dependent genes. Increased expression of ERα and ERβ is realized through overproduction of estrogens and prostaglandins, which entails disruption of apoptosis mechanisms leading to accumulation of DNA errors. It is vital to note that cells evade apoptosis, and estrogen-dependent proliferation of transformed cells results in tumor formation [17]. Aberrations in various chromosomes have been observed in the ER and signaling pathways, implicating them in the pathobiology of fibroid [14,18].The presence of estrogen, in conjunction with ERα, has been demonstrated to render UF susceptible to the effects of progesterone through the induction of PR expression (Picture I). As illustrated in Picture I, estrogen, in conjunction with ERα, induces PR expression, thereby rendering UF susceptible to the effects of progesterone. PR subsequently binds to numerous DNA sites within leiomyoma smooth muscle cells, regulating a multitude of genes and promoting proliferation, survival, and the aberrant production of the extracellular matrix [19].
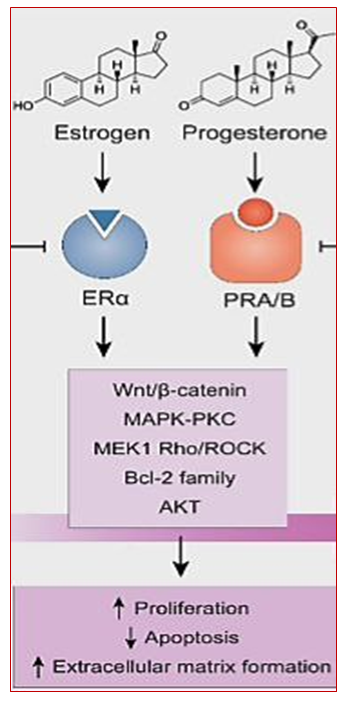 | Figure 1. Нormonal action on myometrial tissue as a cause of UF [19] |
2. Aim of the Work
- The purpose of the research was to study the clinical course of uterine fibroids and the association of genetic polymorphism of ESR1 (rs2228480/594) and PGR (rs1042838) receptor genes with this pathology.
3. Materials and Methods
- In order to accomplish this objective, an open prospective cohort study was conducted from 2021 to 2023, which was based on clinical and laboratory examinations of 200 women of reproductive and premenopausal age who were admitted to the Women's Health Centre and Gynecology Department of the Multidisciplinary Clinic of the Tashkent Medical Academy. The participants were stratified into three distinct groups: the first cohort consisted of patients diagnosed with myomas (n=102), which were further categorized as symptomatic (n=53) or asymptomatic (n=49). The second cohort comprised healthy subjects (n=98) who served as the control group. All the women who were the subject of this study underwent a comprehensive examination including clinical and anamnestic, laboratory, instrumental (ultrasound duplex scanning), histological, molecular genetic (PCR) methods of research. Following database formation, a statistical method of data processing was employed. In the course of the study, the ethical principles stipulated by the World Medical Association Declaration of Helsinki "Ethical Principles for Scientific and Medical Research Involving Human Subjects" (revised in 2024) [23] and diagnostic measures were observed in accordance with of the National Clinic Protocol for Diagnostic Investigations of the Ministry of Health of the Republic of Uzbekistan [24].
4. Results
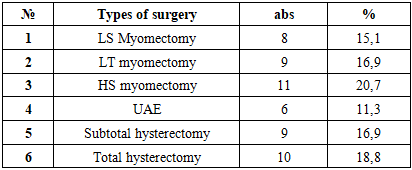
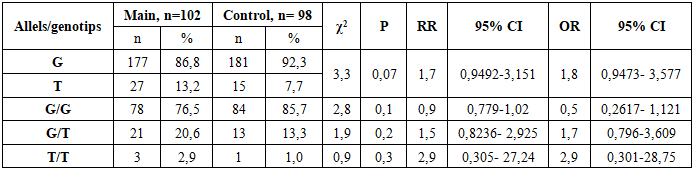
- The investigation of the age demographics of the subjects revealed that the mean age was between 18 and 54 years. Specifically, the mean age was determined to be 42.6±1 years (n=49) in women within the primary cohort who were characterized as having asymptomatic fibroids, and 43.5±0.2 years (n=53) in those who were symptomatic. Notably, the mean age in the control group (n=98) was found to be 38.7±0.9 years (p<0.001). Furthermore, a risk analysis of symptomatic fibroids development was performed in the study population. The analysis revealed that 1.9% of women (n=53) exhibited high risk (31 points), 84.9% exhibited medium risk, and 11.3% exhibited low risk (p<0.001). In contrast, the analysis of asymptomatic fibroids development revealed that the proportion of women with high risk (p<0.001) was non-zero; more than half of the women (52.2%, n=49) exhibited medium risk, and 47.8% exhibited low risk. Risk factors predisposing to the development of the disease are hereditary predisposition, aggravated gynecological history, obesity and lifestyle of the examined persons [25].The present study set out to analyses the outcomes of surgical intervention for women with symptomatic UF (n=53). The selection of surgical methodology for the treatment of symptomatic fibroids was determined by various factors, including but not limited to: age, the presence of reproductive goals, and the severity of clinical symptoms exhibited by the study participants. The presence of anemia in woman with abnormal uterine bleeding (AUB), symptoms of rapid growth and pelvic pain were also considered (Table 1).
|
|
|
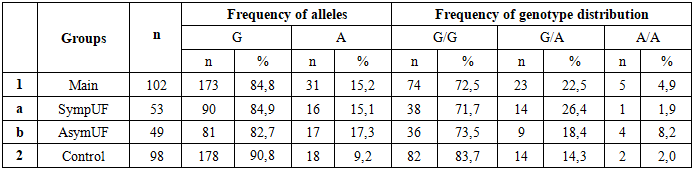
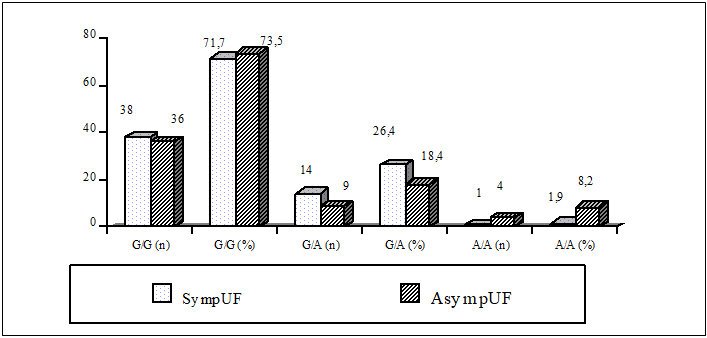 | Figure 2. The Differences in the Frequency of Alleles and Genotypes of the rs2228480/594 Polymorphism of the ESR1 Gene in Symptomatic and Asymptomatic UF Groups |
|
|
|
5. Discussion
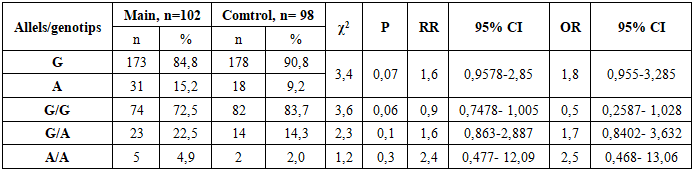
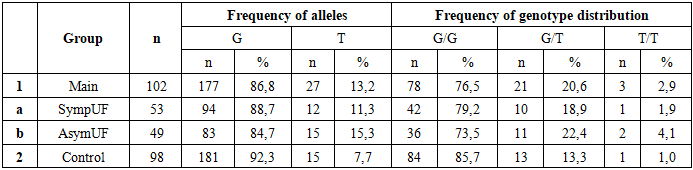
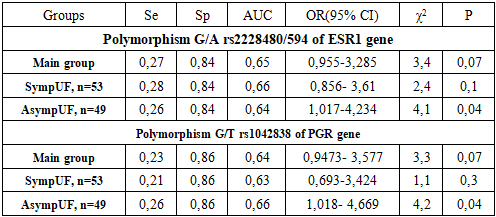
- Uterine fibroid is more frequent in late reproductive and perimenopausal age, which is consistent with the literature. Women with symptomatic fibroids and a high index of risk factors for the disease have a significantly impaired quality of life. As a result, these women develop clinical symptoms such as abnormal uterine bleeding leading to severe anemia, rapid growth of the myoma, pelvic pain and infertility. On ultrasound in women with a mixed form of myomatous nodes, the median uterine volume is greater than 2 times that of asymptomatic UF. This, in turn, leads to the use of radical, organ-removing surgical methods of treatment of this pathology.The current understanding of the role of genetic abnormalities in UF development is insufficient. The extent to which chromosomal abnormalities in tumor cells contribute to the manifestation of clinical symptoms as a result of molecular genetic processes at the level of genes and intragenic interactions remains to be fully elucidated. In order to ascertain the role of polymorphic variants of predictor genes in the development of fibroids, molecular-genetic studies of ESR1 and PGR receptor genes were carried out, and their gene-gene interaction in the development and clinical course of UF was studied.The investigation of allelic variants and genotype frequency of ESR1 receptor gene polymorphism (rs2228480/594) in women diagnosed with UF revealed that individuals carrying a heterogeneous unfavorable G/A genotype exhibited a 2.1-fold higher risk of developing myoma when compared to the control group (OR=2.1; 95% CI 0.936-4.95), whereas individuals possessing the G/G genotype appeared to demonstrate a protective effect against fibroids development (OR=0.5). The involvement of polymorphic loci of ESR1 genes in the pathogenesis of myoma has been demonstrated in a number of studies, which corroborates the findings of the present study. The authors' data further demonstrates that the presence of mutant genotype in women diagnosed with UF can be explained by the influence of ESR1 Polymorphism on myoma development, a process that occurs indirectly.In the study of allele and genotype frequency distribution of the PGR receptor gene polymorphism (rs1042838) in UF, it was established that the homozygous G/G genotype exhibited a protective effect concerning myoma (OR=0.5; 95% CI 0.261-1.121), while the G/T (OR=1.7; 95% CI 0.796-3.609) and T/T (OR=2.9; 95% CI 0.371-28.75) PGR genotypes demonstrated a promoter effect in relation to UF. The probability of identifying this genotype was found to be statistically significant within the patient population of the primary cohort, resulting in an elevated risk of developing the disease from an initial value of 1.7 to a value of 2.9. This finding suggests the potential inefficacy of gestagen-based preparations in women with symptomatic UF, highlighting the necessity of further investigation. Progesterone, through PR-A and PR-B receptors, plays a pivotal role in instigating the sequence of biological disorders, and, along with estradiol, functions as a controller of this process. It has been demonstrated that progesterone exerts its inhibitory effect on ER expression and functions directly through PR. The active involvement of progesterone in the peripheral growth of myomas is well-documented, with estrogens being instrumental in augmenting the expression of the PR in both the myometrium and myoma [15,22]. Consequently, population analyses suggest that polymorphisms in ESR1 and PGR genes are associated with susceptibility to the disease, as evidenced by genetic polymorphisms. In light of the multifaceted etiopathogenesis of UF, a gene-gene interaction analysis was conducted with the aim of predicting the genetic risk of UF development in women. In this study, the interaction between the favorable and unfavorable genotypes of the ESR1 rs2228480/594 and PGR rs1042838 genes was analyzed in patients with UF and in control groups, to identify the haplotype combinations. This approach enabled us to identify unfavorable genotypes of polymorphism polymorphisms of diverse genes implicated in various aspects of this pathological process, in patients exhibiting both symptomatic and asymptomatic symptoms. It was observed that the presence of certain genotypes, including homozygous genes, did not reach a level of statistical significance. An analysis of the combination of unfavorable genotypes of the studied genes among patients with UF and a control sample demonstrated that the risk for myoma development in women with symptomatic (OR=4.7) and asymptomatic fibroids (OR=4.2) was significantly associated with having two or more unfavorable gene combinations, fourfold higher than in the control group. The analysis also revealed a high prognostic significance of unfavorable haplotypes in the development of UF, with an area under the curve (AUC) value of 0.7. Conducted molecular genetic studies have confirmed that evaluating polymorphic loci in isolation is insufficient for predicting the risk of UF development. The gene-gene interaction of ESR1 and PGR has been demonstrated to have a pronounced independent effect on the development of symptomatic myoma, in addition to contributing to the phenotypic development of the disease complication. The observed stability in the connection between polymorphisms in receptor genes in symptomatic and asymptomatic subjects indicates the value of genetic testing, particularly in instances of a family history of UF, in predicting the likelihood of UF development and progression among women in the reproductive and perimenopausal ages. Prediction of UF patients will allow for the anticipation of the manifestation of disease symptoms and its complication, the implementation of timely conservative or organ-preserving surgical procedures, and enhancement of the quality of life for women afflicted by UF. The investigation of epigenetic and genetic factors associated with the development of myomas, which can lead to a heightened risk of disease, constitutes a crucial step in enhancing our comprehension of cellular transformation processes and the identification of diagnostic biomarkers, facilitating early diagnosis of UF. This, in turn, represents a significant advancement in the realm of personalized predictive medicine.
6. Conclusions
- The risk of developing uterine fibroids depends on the presence of cumulative risk factors, which are more pronounced in women with symptomatic fibroids. The associative role of genetic polymorphism of genes: A/A ESR1 (rs2228480/594) (OR=2.1) and G/T and T/T PGR (rs1042838) (OR=1.9) with uterine fibroid development was demonstrated. Two intragenic combinations of unfavorable genotypes of G/A ESR1 rs2228480/594 + G/T PGR rs1042838 predictor genes in patients increased the risk of myoma development from 3.6 to 4.4 times (OR=3.6-4.4; 95% CI 1.829-10.81). At this stage of the work, it would be interesting to further investigate the association of epigenetic risk factors with genetic determinants of fibroids.
ACKNOWLEDGEMENTS
- We would like to express our deep gratitude to all the patients who gave informed consent and participated in this study, and to the staff of the Department of Obstetrics and Gynecology of Family Medicine, Tashkent Medical Academy, and the Department of Molecular Genetics, Institute of Hematology, Uzbekistan.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML