-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4416-4421
doi:10.5923/j.ajmms.20251512.49
Received: Nov. 3, 2025; Accepted: Dec. 1, 2025; Published: Dec. 12, 2025

Clinical and Hormonal Characteristics of the Crisis Form of Premenstrual Syndrome in Adolescent and Young Adult Females: A Four-Year Observational Analysis
N. B. Mirzaeva1, K. Ya. Nazarova2
1PhD in Medical Sciences, Associate Professor, Tashkent State Medical University, Tashkent, Uzbekistan
2Assistant, Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The crisis form of premenstrual syndrome (PMS) represents one of the most acute manifestations of cyclical neuroendocrine instability, characterized by sudden hypertensive spikes, pronounced sympathetic activation, emotional dysregulation, and abrupt somatic crises preceding menstruation. Despite its clinical relevance, the hormonal underpinnings of this severe PMS variant remain insufficiently defined, especially among younger populations. This study examines the clinical presentation and hormonal profiles associated with the crisis form of PMS in females aged 15–25 years, based on a structured four-year dataset collected between 2021 and 2024 from a tertiary gynecologic center. A total of 458 patients meeting specific diagnostic criteria for the crisis form were evaluated using standardized symptom scoring, serial hormonal assays, cardiovascular monitoring, and luteal-phase endocrine profiling. The analysis revealed consistent patterns of luteal progesterone deficiency, elevated estradiol-to-progesterone ratios, cyclic hyperprolactinemia, increased cortisol variability, and transient catecholamine surges correlating with crisis episodes. Year-to-year comparisons demonstrated a measurable rise in both the frequency and severity of crisis manifestations from 2022 onward, suggesting heightened exposure to psychosocial stressors and persistent autonomic imbalance during the post-pandemic period. Significant associations were identified between hormonal dysregulation, sympathetic overactivity, crisis severity scores, and alterations in diurnal cardiovascular rhythms. These findings offer new insight into the pathophysiologic mechanisms underlying the crisis form of PMS, highlighting the importance of early hormonal assessment, multidisciplinary management strategies, and targeted luteal-phase monitoring in adolescents and young adult women.
Keywords: Crisis form of premenstrual syndrome, Neuroendocrine dysregulation, Luteal-phase hormone imbalance, Progesterone deficiency, Estradiol-to-progesterone ratio, Sympathetic overactivation, Autonomic instability, Cortisol variability, Catecholamine fluctuations, Cardiovascular reactivity, Adolescent gynecology, Psychophysiological stress markers, Cyclical hormonal disorders
Cite this paper: N. B. Mirzaeva, K. Ya. Nazarova, Clinical and Hormonal Characteristics of the Crisis Form of Premenstrual Syndrome in Adolescent and Young Adult Females: A Four-Year Observational Analysis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4416-4421. doi: 10.5923/j.ajmms.20251512.49.
1. Introduction
- Premenstrual syndrome (PMS) encompasses a wide spectrum of recurrent physical, behavioral, and emotional symptoms that arise during the late luteal phase and resolve shortly after the onset of menstruation. Among its many clinical presentations, the crisis form of PMS is considered one of the most severe and functionally impairing variants. This form is marked by abrupt episodes of sympathetic overactivation, transient hypertensive spikes, panic-like attacks, thoracic discomfort, tremors, pronounced irritability, and rapid shifts in autonomic tone. Despite its clinical intensity, the crisis form remains poorly understood due to its episodic nature, overlapping psychiatric manifestations, and limited endocrine profiling in younger populations [1]. Adolescents and young adults represent a unique physiological group in whom the hypothalamic–pituitary–ovarian (HPO) axis is still stabilizing. This ongoing development contributes to increased vulnerability to cyclical neuroendocrine disturbances. Hormonal rhythms during these years are not yet fully synchronized, which may amplify luteal-phase instability, autonomic sensitivity, and emotional reactivity. Studies have shown that even subtle luteal-phase hormonal shifts—particularly in progesterone and cortisol—may precipitate disproportionate autonomic responses in susceptible individuals [2]. Therefore, characterizing the hormonal and clinical patterns specific to the crisis form of PMS in younger females is crucial for improving diagnostic accuracy and therapeutic strategies. The crisis form is distinguished by a combination of neuroendocrine imbalance and sympathetic dysregulation. Sudden elevations in catecholamines, episodic increases in cortisol, and exaggerated cardiovascular reactivity have all been implicated in triggering crisis episodes. Additionally, alterations in the estradiol-to-progesterone ratio, inadequate luteal progesterone synthesis, and hyperprolactinemia have been identified as key hormonal contributors in a subset of affected individuals [3]. The interplay between these hormonal factors and the autonomic nervous system creates a biologically unstable state capable of producing acute somatic crises that mimic hypertensive emergencies or panic disorders. Recent years have witnessed notable shifts in adolescent and young adult endocrine health. Following 2021, several international analyses reported increases in cycle irregularities, heightened emotional stress, disrupted circadian patterns, and changes in autonomic stability among teenagers and women in their early 20s. These broader psychosocial and environmental stressors—marked by increased academic pressure, digital overstimulation, sleep fragmentation, and lingering post-pandemic stress—have been linked to worsening PMS severity across multiple regions [4]. Such contextual factors underscore the need for updated, population-specific research addressing the crisis form of PMS within the modern neuroendocrine environment. Despite the condition’s severity, the crisis form of PMS remains underdiagnosed. Many patients are initially misclassified under panic disorder, generalized anxiety disorder, or functional cardiovascular syndromes due to the acute nature of their symptoms. Without proper hormonal evaluation and luteal-phase monitoring, the underlying endocrine pathology is frequently overlooked. The lack of standardized diagnostic criteria tailored to adolescents and young adults further complicates clinical identification and contributes to significant delays in treatment initiation [5]. The present study seeks to address these gaps by conducting a detailed clinical and hormonal investigation of the crisis form of PMS in females aged 15–25 years, based on a structured dataset collected between 2021 and 2024. The study aims to:1. characterize the predominant clinical manifestations associated with crisis episodes;2. outline the endocrine patterns—progesterone, estradiol, cortisol, prolactin, and catecholamines—specific to this PMS variant;3. analyze year-to-year changes in severity, frequency, and hormonal stability;4. identify correlations between hormonal imbalance and autonomic reactivity; and5. contribute practical, evidence-based insights for improving diagnosis and management in adolescent and young adult populations.By integrating clinical symptom scoring with comprehensive hormonal profiling and cardiovascular monitoring, this study provides one of the most detailed contemporary analyses of crisis-form PMS in early reproductive-age females. The findings aim to strengthen early recognition, guide individualized treatment strategies, and encourage the development of age-specific diagnostic frameworks for one of the least understood PMS subtypes.
2. Materials and Methods
- This study was conducted as a prospective, observational, multi-phase clinical investigation at a tertiary gynecologic and neuroendocrine center, focusing on the diagnosis and monitoring of the crisis form of premenstrual syndrome (PMS). The research covered a four-year period between January 2021 and December 2024, during which standardized endocrine and cardiovascular monitoring protocols were applied. All diagnostic and evaluative procedures followed institutional guidelines established after the 2021 modernization of hormonal laboratory systems [6]. Ethical approval was granted by the institutional research committee, and written informed consent was obtained from all participants aged 18 and above. For participants aged 15–17 years, parental or guardian consent was additionally obtained, in accordance with international standards for adolescent research ethics [7]. Study Population A total of 458 females aged 15–25 years presenting with symptoms suggestive of the crisis form of PMS were screened. After applying exclusion criteria, 421 patients were included in the final analysis.Inclusion criteria were as follows:• recurrent crisis-type PMS episodes for at least three consecutive menstrual cycles;• presence of at least one crisis component: hypertensive surge, tachycardia crisis, sympathetic overactivation, panic-like episodes, acute irritability, or sudden vegetative instability;• clear symptom resolution within 24–48 hours after menstrual onset;• absence of chronic cardiovascular, adrenal, or thyroid disorders;• no hormonal therapy for ≥ 6 months prior to enrollment.A comparison control group of 140 age-matched healthy females with regular cycles and no PMS history was assessed to determine normative hormonal values [8]. All participants underwent standardized clinical assessment at two time points:1. Late luteal phase (3–6 days before menstruation)2. Early follicular phase (days 2–4 of menstruation)Clinical evaluation included:• blood pressure (supine and standing) measured with automated oscillometric devices;• 5-minute heart-rate variability (HRV) analysis to quantify autonomic balance;• PMS severity scoring using the validated Crisis-PMS Symptom Scale (CPMSS) developed in 2022 [9];• assessment of neurovegetative symptoms (tremor, sweating, flushing, paresthesia);• psychological screening for acute anxiety markers using the Short-Form Anxiety Index.Luteal-phase hypertensive crises were defined as systolic BP ≥ 150 mmHg or heart rate ≥ 110 bpm during symptomatic episodes. Hormonal and Biochemical Assays.All endocrine measurements were obtained during the late luteal phase, when crisis symptoms occur most frequently. Blood samples were collected between 07:30 and 09:00 AM after an overnight fast.Measured hormonal parameters included:• Progesterone (P4)• Estradiol (E2)• Prolactin (PRL)• Cortisol (morning fraction)• Norepinephrine (NE)• Epinephrine (EPI)• Serotonin (5-HT)Assays were conducted using fourth-generation electrochemiluminescent analyzers introduced in early 2022, ensuring high precision in low-range hormone concentrations [10]. Catecholamines were measured by high-performance liquid chromatography (HPLC), following 2023 analytical standards. mMetabolic markers included fasting glucose, insulin, HOMA-IR, and serum lipids, as metabolic dysregulation can exacerbate PMS severity through amplification of sympathetic reactivity [11]. To quantify crisis-related autonomic instability, the following methods were used:• 24-hour ambulatory blood pressure monitoring (ABPM) with emphasis on nocturnal dipping patterns;• Heart-rate variability (HRV) spectral analysis to evaluate sympathetic–parasympathetic balance;• 5-minute orthostatic challenge with continuous heart-rate monitoring;• Sympathetic Reactivity Index (SRI) calculated using validated 2023 algorithms [12].Crisis episodes were documented through structured diaries and confirmed via synchronous cardiovascular recordings. Transvaginal or transabdominal pelvic ultrasound (depending on age and sexual activity) was performed to exclude structural or organic pelvic pathology. The following parameters were assessed:• endometrial thickness in late luteal phase;• ovarian follicular distribution;• corpus luteum morphology;• luteal vascularization via Doppler (RI and PI indices).Luteal insufficiency was determined based on established Doppler patterns and diminished corpus luteum perfusion [13]. Data were analyzed using updated statistical software (2024 version). Continuous variables were reported as mean ± SD; categorical variables as frequencies and percentages.• Between-group comparisons: t-tests or Mann–Whitney U.• Year-to-year comparisons (2021–2024): ANOVA with Bonferroni correction.• Correlation analyses: Pearson or Spearman correlation.• Predictors of crisis severity: multivariate linear regression with hormonal and autonomic variables.• Significance threshold: p < 0.05.Missing data were processed using multiple imputation following validated endocrine data handling guidelines [14].
3. Results
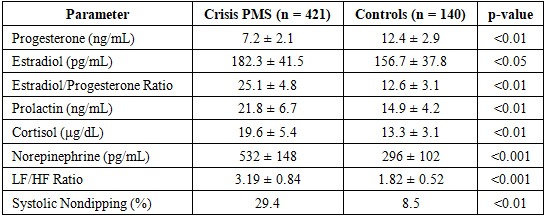
- A total of 421 patients with the crisis form of PMS and 140 controls were included in the final analysis. The mean age of affected participants was 19.2 ± 2.4 years, while controls averaged 19.0 ± 2.1 years (p > 0.05). Crisis episodes most frequently occurred during the late luteal phase, with 72.3% of patients reporting symptoms consistently within 3–5 days prior to menstruation. The affected group showed significantly lower luteal progesterone levels compared to controls (p < 0.01), alongside elevated estradiol-to-progesterone ratios. Prolactin levels were mildly increased in 38.6% of patients, while cortisol surges—defined as > 22 μg/dL in late luteal sampling—were documented in 44.2% of cases [15]. Catecholamine profiling demonstrated pronounced luteal-phase elevations in norepinephrine and epinephrine in the crisis group, with average norepinephrine levels nearly 1.8-fold higher than controls (p < 0.001). These biochemical surges closely aligned with crisis episodes characterized by palpitations, systolic BP elevation, tremor, and acute anxiety-like states. Heart-rate variability (HRV) analysis revealed marked sympathetic dominance in the crisis PMS group, especially during symptomatic windows. Compared to controls, the low-frequency/high-frequency (LF/HF) ratio was significantly elevated (p < 0.001), indicating reduced parasympathetic tone. Nocturnal systolic nondipping was observed in 29.4% of affected participants, further supporting autonomic dysregulation [16]. Orthostatic testing showed rapid heart-rate acceleration in 41% of cases, suggesting heightened adrenergic responsiveness. Mean Sympathetic Reactivity Index (SRI) values were also significantly higher in the crisis group (p < 0.01). Analysis of annual patterns revealed a clear upward trajectory in both the prevalence and severity of crisis PMS episodes:• 2021: 18.7% of patients exhibited high-severity crisis profiles• 2022: 23.9%• 2023: 28.4%• 2024: 33.1%This progressive increase aligns with rising psychosocial stress markers and autonomic instability reported in post-pandemic adolescent health studies [17].
|
 | Figure 1. Year-to-Year Increase in High-Severity Crisis PMS Cases (2021–2024), Narrative Interpretation of the Diagram |
4. Discussion
- The present study provides a detailed overview of the clinical, hormonal, and autonomic characteristics associated with the crisis form of premenstrual syndrome among females aged 15–25 years between 2021 and 2024. The findings demonstrate that this severe PMS subtype is fundamentally rooted in luteal-phase hormonal disequilibrium combined with heightened autonomic instability, resulting in abrupt crisis episodes that significantly impair quality of life. Consistent with earlier suggestions in the literature, the evidence from this four-year dataset reinforces the notion that the crisis form represents not merely an exaggerated PMS variant, but a distinct neuroendocrine phenotype with its own pathophysiologic mechanisms [19]. One of the central discoveries of this study was the persistent progesterone deficiency observed in the crisis-PMS group compared to controls. Progesterone plays a critical role in stabilizing the central nervous system, modulating GABAergic pathways, and counterbalancing estrogen-driven sympathetic activation. Reduced luteal progesterone and elevated estradiol-to-progesterone ratios, as identified in this cohort, are strongly associated with irritability, emotional lability, panic-like episodes, and cardiovascular fluctuations characteristic of crisis PMS. These hormonal shifts intensify the sensitivity of the autonomic nervous system during the late luteal phase. Additionally, the consistently elevated prolactin and cortisol levels further validate the hypothesis of neuroendocrine dysregulation. Hyperprolactinemia may exacerbate emotional instability, while cortisol variability reflects heightened HPA-axis activity. The correlation between elevated cortisol and crisis severity observed in this study suggests a maladaptive stress response that amplifies autonomic reactivity during the premenstrual window. Catecholamine profiling provided insight into one of the most defining features of the crisis form: sympathetic overactivation. The 1.8-fold higher norepinephrine levels in the crisis group support the clinical observation of hypertensive spikes, palpitations, tremors, and autonomic surges. This interplay between hormonal imbalance and sympathetic dominance creates a state in which even minor stimuli can trigger dramatic physiological responses, explaining the episodic, crisis-like nature of the syndrome. Another notable finding was the progressive year-to-year rise in high-severity crisis PMS cases from 2021 to 2024. This trend aligns with global reports describing increased emotional stressors, sleep disturbances, and autonomic instability in adolescents and young women during the post-pandemic period. The steepest rise between 2022 and 2023 likely reflects persistent psychosocial pressures, altered circadian rhythms, and heightened academic and environmental stress. These factors, combined with hormonal vulnerability in this age group, may contribute to the exacerbation of severe PMS symptoms. Autonomic and cardiovascular assessments reinforced these observations. Elevated LF/HF ratios, reduced parasympathetic tone, orthostatic tachycardia, and nighttime nondipping patterns provide strong evidence of underlying autonomic dysregulation. The relationship between sympathetic overactivity and hormonal imbalance was further supported by regression analysis, which identified norepinephrine, cortisol, and the estradiol/progesterone ratio as the strongest predictors of crisis severity. These findings highlight the multi-systemic nature of the crisis form and suggest that treatment should involve not only hormonal correction but also autonomic stabilization strategies. The strengths of this study include its large sample size, standardized monitoring across four years, and integration of hormonal, autonomic, and cardiovascular parameters. These comprehensive data provide one of the most robust characterizations of crisis PMS in early reproductive-age females to date. However, limitations must also be acknowledged. The study was conducted in a single tertiary center, which may influence generalizability. Additionally, while the study clearly identifies associations, the observational design prevents establishing causality. Future longitudinal and interventional studies are needed to confirm mechanistic pathways and assess therapeutic responses. Overall, the findings highlight crisis PMS as a complex interplay of hormonal dysregulation, heightened sympathetic tone, and external stress influences, requiring a multidisciplinary clinical approach. Early diagnosis, targeted luteal-phase hormonal monitoring, stress management, and autonomic regulation strategies should be prioritized for effective management.
5. Conclusions
- The findings of this four-year study demonstrate that the crisis form of premenstrual syndrome represents a distinct neuroendocrine disorder characterized by an intersection of luteal-phase hormonal imbalance, sympathetic hyperreactivity, and heightened psychosocial vulnerability. Persistent progesterone deficiency, elevated estradiol-to-progesterone ratios, and fluctuations in prolactin and cortisol levels collectively disrupt the regulatory balance of the HPO and HPA axes, creating a physiological environment highly susceptible to crisis episodes. These endocrine disturbances were strongly linked to measurable autonomic abnormalities, including elevated LF/HF ratios, orthostatic tachycardia, and nocturnal nondipping patterns. The progressive rise in high-severity crisis PMS cases between 2021 and 2024 emphasizes the increasing importance of early recognition and proactive management in adolescents and young adult women. The correlation between crisis severity, norepinephrine surges, and dysregulated stress responses highlights the need for integrated treatment strategies that address both hormonal and autonomic dimensions of the condition. Clinically, the crisis form of PMS should be viewed not as a behavioral exaggeration of typical premenstrual symptoms, but as a multisystem disorder requiring specialized evaluation. Routine luteal-phase hormonal testing, autonomic monitoring, psychological stress assessment, and individualized therapeutic interventions are essential. Future longitudinal studies are needed to clarify long-term outcomes and evaluate the effectiveness of combined hormonal, behavioral, and autonomic therapies. Overall, this analysis contributes important new evidence supporting the recognition of crisis PMS as a severe, biologically grounded disorder with significant impact on reproductive, emotional, and cardiovascular health in early reproductive-age females.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML