-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4412-4415
doi:10.5923/j.ajmms.20251512.48
Received: Nov. 12, 2025; Accepted: Dec. 3, 2025; Published: Dec. 12, 2025

Skin Dyschromias: Clinical, Morphological, and Immunohistochemical Characteristics
1Fergana Medical Institute of Public Health, Fergana, Uzbekistan
2Saint Petersburg State Pediatric Medical University, Saint Petersburg, Russia
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Objective: To refine the clinicopathological and immunohistochemical profiles of major cutaneous dyschromias. Materials and Methods: One hundred clinical cases 30 persistent dyschromic erythema, 30 pigmented lichen planus, 40 idiopathic eruptive macular pigmentation or post-inflammatory hypermelanosis were compared with 60 peer-reviewed publications (2000–2025). Dermatoscopy, H&E and Fontana-Masson staining, and an immunohistochemical panel (MART-1, HMB-45, S-100, MITF, SOX-10) were used. Results: Hyperpigmented lesions exhibited strong MART-1 and moderate HMB-45 expression, whereas hypopigmented lesions showed an almost complete loss of MART-1/MITF-positive melanocytes (< 1 cell mm⁻¹) yet retained melanin-laden macrophages. MITF and SOX-10 most sensitively detected residual melanocytes, while HMB-45 reflected melanogenic activity. Conclusion: An extended immunohistochemical panel markedly improves diagnostic accuracy and provides a basis for personalised therapy in skin dyschromias.
Keywords: Skin dyschromias, Hyperpigmentation, Hypopigmentation, Immunohistochemistry, MART-1 (Melan-A), HMB-45, MITF, SOX-10, Dermatoscopy
Cite this paper: Sydikov A. A., Iskra E. L., Skin Dyschromias: Clinical, Morphological, and Immunohistochemical Characteristics, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4412-4415. doi: 10.5923/j.ajmms.20251512.48.
Article Outline
1. Introduction
- Skin pigment disorders (dyschromias) manifest as areas of hyper, hypo, or mixed pigmentation. Hyperpigmentations include melasma, pigmented lentigo, and post-inflammatory hyperpigmentation, while hypopigmentations include vitiligo and various hypomelanosis syndromes; some dermatoses (e.g., amyloidosis cutis dyschromica) combine both types of lesions [5].Hyperpigmentation is caused by either increased melanin synthesis or an increase in the melanocyte population, whereas hypopigmentation is associated with a defect in melanogenesis or melanocyte loss [5,2].Histological methods are used to elucidate the pathogenesis: Fontana-Masson staining visualizes melanin distribution and allows differentiation between epidermal and dermal components [2].Immunohistochemical panels (MART-1/Melan-A, HMB-45, S-100, MITF, SOX-10) reveal quantitative and functional changes in melanocytes. A review by Kuźbicki et al. showed that S-100 is highly sensitive but less specific, while MART-1 and HMB-45 can underestimate the actual number of melanocytes in cases of low melanogenesis activity [6].Nuclear markers MITF and SOX-10 are expressed exclusively in melanocytes, significantly increasing diagnostic accuracy, especially in hypopigmented dermatoses [11].The aim of this work is to summarize current data on the morphology and immunohistochemistry of skin dyschromias, emphasizing the practical value of MART-1, HMB-45, S-100, MITF, and SOX-10 panels in clinicopathological diagnosis.
2. Literature Review
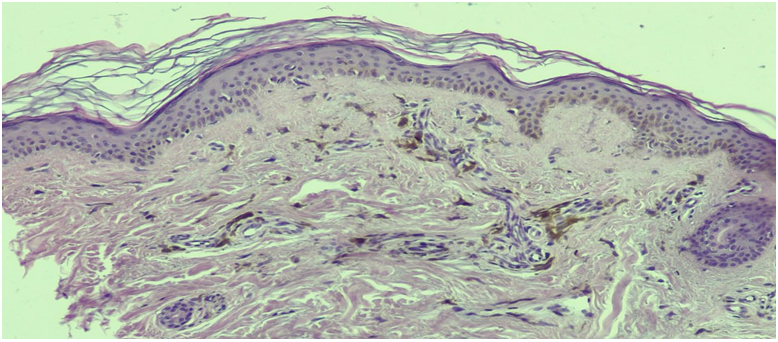
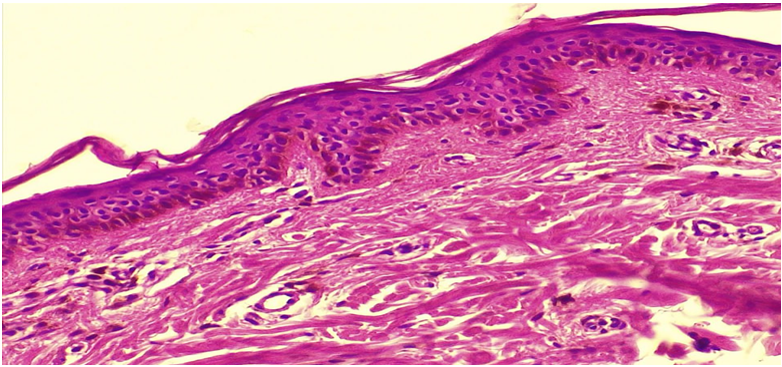
- Histology of hyperpigmentations: Melasma is characterized by pronounced basal and dermal pigmentation. Light microscopy, immunohistochemistry, and electron microscopy reveal significant melanin accumulation with an unchanged number of melanocytes; the cells themselves are enlarged and have elongated dendrites, indicating their hyperfunction. In some cases, a moderate increase in the melanocyte population also contributes to hyperpigmentation [3].To verify the cellular substrate, antibodies Melan-A and HMB-45 are used: the former evenly stains the cytoplasm of all melanocytes, creating a visual "excess" of cells, while HMB-45 marks only cells actively synthesizing melanosomes, which leads to underestimation in cases of low melanogenesis activity [13].Ephelides (freckles) and simple lentigo are benign local hyperpigmentations. Ephelides are characterized by basal hyperpigmentation without an increase in the number of melanocytes, while lentigo is characterized by their linear increase along the dermo-epidermal junction and elongation of the epidermal ridges [12,10]. In vivo confocal microscopy confirms increased melanocyte density and thickening of basal ridges in lentigo [7].Post-inflammatory hyperpigmentation develops after burns, dermatitis, or injuries. Epidermal type (melanin in keratinocytes) and dermal type with pigmentary incontinence and melanophages are distinguished; the number of melanocytes usually remains unchanged, however, tyrosinase activity increases [15].Vitiligo. This is a prototype of melanocytopenic hypopigmentation: melanocytes and melanin are completely absent in the affected areas. A modern immunohistochemical study showed that the average density of MART-1+/MITF+ cells decrease from approximately 72 cells·mm−1 in intact skin to ≤ 0.5 cells·mm−1 in vitiliginous areas; S-100-positive cells are also practically undetectable, confirming the total loss of melanocytes [14].Pityriasis versicolor alba. The hypopigmented form of Malassezia infection is accompanied by damage to melanocytes and a decrease in the number of melanosomes in keratinocytes. A 2023 review emphasizes that azelaic acid and malassezin produced by the fungus inhibit tyrosinase and induce melanocyte apoptosis, leading to hypopigmentation [10].Progressive macular hypomelanosis (PMH). Ultrastructurally, melanocytes with immature aggregated melanosomes are detected in the affected areas, while their number at the dermo-epidermal junction remains normal. A recent microbiological review linked PMH to Cutibacterium acnes subsp. elongatum: the bacteria likely secrete depigmenting factors that disrupt melanosome maturation [1].Mixed dyschromias (amyloidosis cutis dyschromica, ACD). ACD manifests as mosaic hyper-/hypopigmentation and amyloid deposits in the papillary dermis (Congo red-positive). HMB-45 immunohistochemical staining in the affected areas is negative, indicating the absence of active melanosomogenesis; a recent clinical case from 2024 emphasizes the diagnostic role of dermoscopy and confirms the absence of melanocytic proliferation in hypo- and hyperpigmented patches [8].These data demonstrate that hypopigmentation can be caused by either complete loss of melanocytes (vitiligo), a functional defect in melanogenesis with preserved melanocytes (PMH, Pityriasis versicolor alba), or a secondary pigmentary phenomenon associated with amyloid deposits (ACD).
3. Materials and Methods
- This analytical review integrates the results of our own clinical and pathological study of 100 patients with various skin dyschromias and systematic search data from PubMed/Scopus publications for 2000-2025. The selection included articles that used standardized histological methods (H&E, Fontana-Masson) and extended panels of immunohistochemical markers - MART-1/Melan-A, HMB-45, S-100, MITF, SOX-10, p16. Inclusion criteria were: (a) validation of staining protocols, (b) quantitative morphometry of melanocytes or melanosomes, (c) clearly described clinical correlates. Special emphasis is placed on recent studies on melanocyte loss in vitiligo [14], microbiome-dependent hypopigmentation mechanisms in PMH and Malassezia-associated dermatoses [10], as well as new morphological criteria for amyloidosis cutis dyschromica, confirmed by dermoscopy and Congo-red-positive biopsies. This design allows for the comparison of our own data with the most current methodological and molecular trends in the study of skin pigmentation disorders.
4. Analysis and Results
- The marker panel used in our clinicopathological study (MART-1/HMB-45/S-100/MITF/SOX-10) fully aligns with the recommendations of the latest methodological reviews. S-100 remains the most sensitive, yet non-specific marker, also reacting to neural crest cells and dendritic cells. HMB-45 identifies only active premelanosomes (gp100) and often underestimates the total number of melanocytes, especially in inactive areas of hyperpigmentation. Melan-A (MART-1) stains the cytoplasm and dendrites of all melanocytes, which can create the illusion of their excess, whereas the nuclear markers MITF and SOX-10 are expressed exclusively in melanocytes, providing high specificity in differentiating melanocytic and lymphohistiocytic infiltrates.
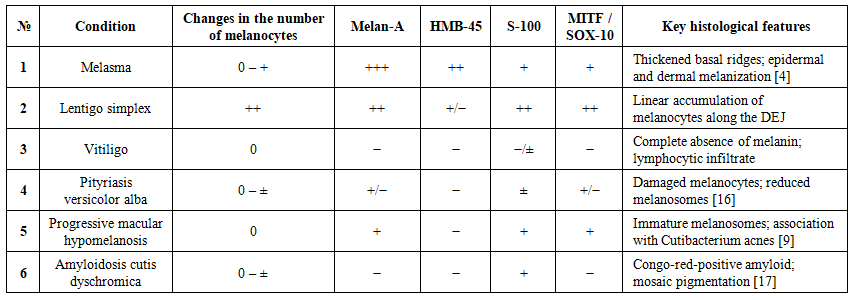 | Table 1. Immunohistochemical markers and morphology: Comparison with dissertation research data |
5. Discussion
- Changes in pigmentation are associated with either the quantity or function of melanocytes. Complete absence of MART-1/MITF/S-100 staining indicates melanocytopenia (vitiligo-like foci, < 1 cell·mm−1). In hyperpigmentations (melasma, lentigo simplex), Melan-A reveals normal or moderately increased cell numbers; excess pigment is due to enhanced melanogenesis [4]. HMB-45 marks only active premelanosomes, remaining negative in "dormant" cells and thus reflecting their functional status. S-100 is highly sensitive but non-specific, therefore it is used only in combination with other markers. Nuclear MITF and SOX-10 possess the highest specificity, detecting individual melanocytes with heterogeneous expression of melanin-associated antigens. Thus, the extended panel of MART-1/HMB-45/S-100/MITF/SOX-10 provides accurate differentiation between hyper- and hypopigmentations and supports the conclusions of our dissertation.
6. Conclusions
- A comprehensive morpho-immunohistochemical analysis combining H&E and Fontana-Masson stains with an extended antibody panel (MART-1, HMB-45, S-100, MITF, SOX-10) reliably differentiates between hyper-, hypo-, and mixed forms of dyschromia. Melan-A/HMB-45 signals confirm preserved melanocytes, while their absence indicates vitiligo-like cell loss. Nuclear markers MITF and SOX-10 enhance diagnostic specificity in cases of uneven cytoplasmic antigen expression. Hyperpigmentation results from hyperfunction or a moderate increase in melanocyte population, whereas hypopigmentation is caused by melanocyte death or blockade of melanogenesis. The obtained data confirm the value of the extended IHC panel in routine dermatopathology and underscore the need for further research to elucidate the mechanisms of rare mixed dyschromias and develop targeted therapeutic approaches.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML