-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4321-4324
doi:10.5923/j.ajmms.20251512.25
Received: Nov. 8, 2025; Accepted: Nov. 25, 2025; Published: Dec. 8, 2025

Morphological and Morphometric Characteristics of the Thyroid Gland in White Rats Under Selenium Deficiency
O. G. Aslanov1, R. R. Baymuradov2
1Independent Researcher, Department of Anatomy, Clinical Anatomy (OSTA), Bukhara State Medical Institute named after Abu Ali ibn Sino, Uzbekistan
2PhD., Associate Professor, Department of Anatomy, Clinical Anatomy (OSTA), Bukhara State Medical Institute named after Abu Ali ibn Sino, Uzbekistan
Correspondence to: O. G. Aslanov, Independent Researcher, Department of Anatomy, Clinical Anatomy (OSTA), Bukhara State Medical Institute named after Abu Ali ibn Sino, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study examines the morphological and morphometric characteristics of the thyroid gland in white rats under selenium deficiency. Key histological alterations, variations in follicular dimensions, colloid condition, and vascular responses were analyzed. The findings demonstrate that selenium deficiency results in significant structural and functional disturbances of the thyroid gland, indicating the high sensitivity of endocrine tissues to microelement imbalance.
Keywords: Selenium, Microelement deficiency, Thyroid gland, Morphology, Morphometry, Experimental study, White rats
Cite this paper: O. G. Aslanov, R. R. Baymuradov, Morphological and Morphometric Characteristics of the Thyroid Gland in White Rats Under Selenium Deficiency, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4321-4324. doi: 10.5923/j.ajmms.20251512.25.
1. Introduction
- The functional activity of the thyroid gland is essential for maintaining hormonal balance, metabolic processes, and tissue growth in the organism. Trace elements, particularly selenium (Se), serve as indispensable cofactors for the synthesis of thyroid hormones—thyroxine (T4) and triiodothyronine (T3)—and for the activity of deiodinase enzymes that activate these hormones. Recent scientific studies have demonstrated that selenium deficiency affects not only thyroid hormone metabolism, but also leads to pronounced alterations in the morphological structure of the gland.Vasiliu et al. (2020) reported degenerative changes in follicular structure, reduced colloid density, and morphologic remodeling of the epithelial layer in conditions of selenium deficiency [4]. Hofstee (2020) showed that maternal selenium deficiency in experimental animals caused disturbances in thyroid activity and hormone balance in offspring, with a pronounced sex-specific pattern [1]. Köhrle (2023) highlighted the interrelated mechanisms of selenium, iodine, and iron in thyroid hormone synthesis and demonstrated that deficiencies of these microelements destabilize morphological integrity [2].Furthermore, Wang (2023) emphasized the protective role of selenium in the pathogenesis of thyroid disorders, noting that Se deficiency intensifies dystrophic processes in follicular epithelium [5]. According to Milanović et al. (2024), the combination of selenium deficiency and hypothyroidism drastically reduces the activity of selenoproteins such as glutathione peroxidase, thereby increasing the susceptibility of thyroid tissue to oxidative stress [3].Overall, these scientific data indicate that selenium is essential for maintaining the morphological stability of the thyroid gland, and that its deficiency can significantly alter follicular architecture, colloid quality, and endocrine function. Therefore, investigating the influence of selenium deficiency on morphological and morphometric parameters of the thyroid gland in an experimental white rat model remains a relevant scientific task.The aim of this study was to determine the histomorphometric characteristics of the thyroid gland in white outbred rats under conditions of selenium (Se) deficiency, including alterations in follicular structure, colloid density, epithelial layer morphology, and elements of the microcirculatory system. The study also sought to evaluate the impact of selenium deficiency on the functional activity and morphometric parameters of the thyroid gland and to provide a scientific basis for the main pathogenetic mechanisms underlying endocrine imbalance associated with Se deficiency.
2. Materials and Methods
- Experimental studies were conducted on male white outbred rats weighing 150–170 g, maintained under standard vivarium conditions. The animals were divided into two main groups:Group I – Control: rats maintained on a standard diet.Group II – Selenium-deficient (Se–): rats fed a specialized diet with selenium content below physiological levels.The experimental model used diets produced by ALTROMIN Spezialfutter GmbH & Co. KG (Germany): C 1000, C 1035, C 1038, C 1040, C 1045, and a modified C 1035 diet adapted for selenium deficiency. The duration of the experiment was set at 10 weeks. All procedures were performed according to ethical certificate No. 36/2024.Histological preparations of the thyroid gland were obtained. Standard hematoxylin–eosin (H&E) staining was performed, and PAS reaction along with additional staining methods were used to evaluate colloid homogeneity. Morphometric measurements were conducted using a calibrated micrometric scale, including parameters such as follicle diameter, epithelial height, colloid area, arteriolar diameter, and capillary density.Both parametric and nonparametric statistical methods were applied. Data collection, cleaning, and systematization were performed using Microsoft Excel 2016, while statistical analysis was conducted in IBM SPSS Statistics v.26 (IBM Corp., USA). The distribution of quantitative variables was assessed using the Shapiro–Wilk test (n < 50) or the Kolmogorov–Smirnov test (n > 50). The Mann–Whitney U-test was applied for non-normally distributed data, whereas the Student’s t-test was used for normally distributed variables. Graphical visualization was performed with STATGRAPHICS 5.1. The mean (M), standard error (m), variance, and coefficient of variation were calculated, and percentage differences relative to the control group were evaluated. A significance level of p < 0.05 was accepted.
3. Results
- Selenium (Se) is a crucial element for thyroid physiology. Approximately 14 mg of selenium is present in the human body, incorporated as selenocysteine in glutathione peroxidase (GPx) and iodothyronine deiodinase enzymes. GPx, a Se-dependent enzyme, catalyzes the breakdown of hydrogen peroxide (H₂O₂), thereby protecting tissues from oxidative stress and cellular damage. In the thyroid gland, H₂O₂ is produced in response to TSH stimulation and is required by thyroid peroxidase (TPO) for the iodination of thyroglobulin, which drives T4 synthesis. Excessive H₂O₂ has been shown to inactivate TPO in vitro, suggesting that its accumulation can impair thyroid hormone production.In regions where selenium and iodine deficiency coexist, reduced Se intake may decrease maternal T4 metabolism, potentially protecting the developing fetal brain from hypothyroidism. However, after birth, the hyperplastic and hyperstimulated iodine-deficient thyroid produces excessive reactive oxygen species, which—combined with decreased GPx activity—may contribute to thyroid tissue damage.
|
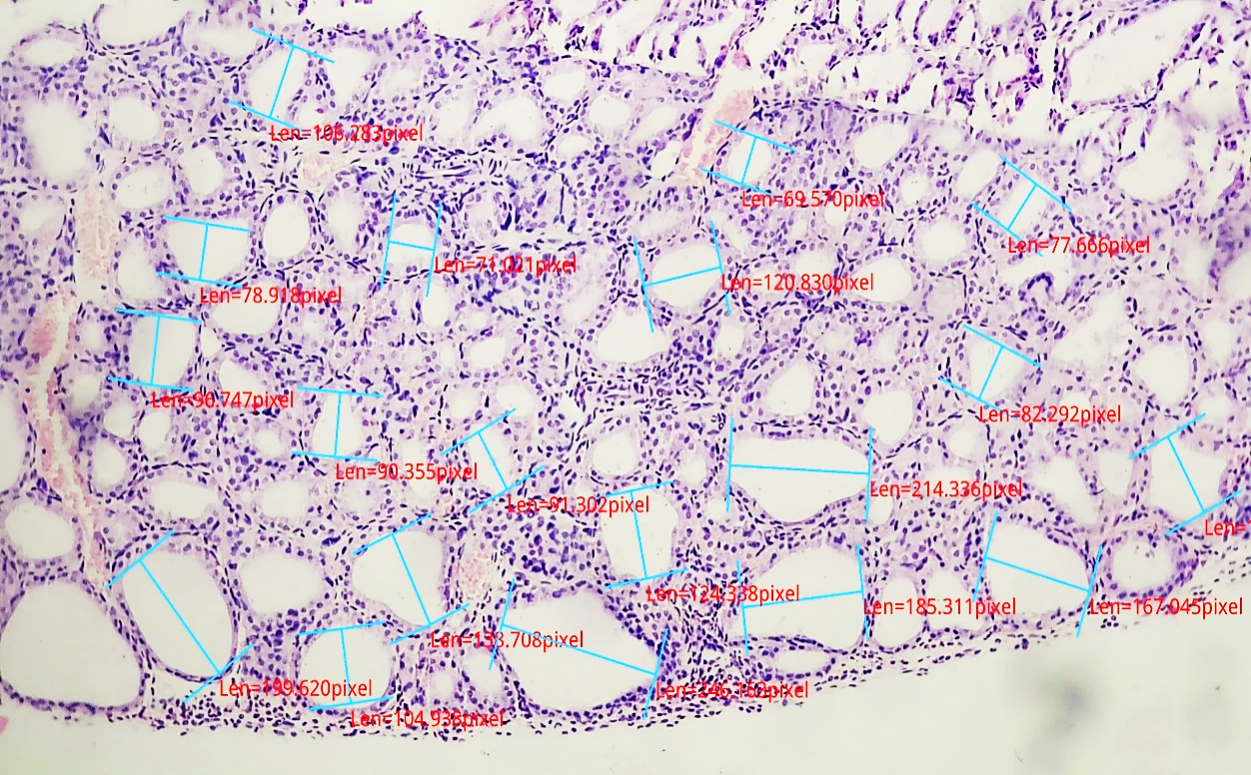 | Figure 3. Morphometric parameters of the thyroid gland in 6-month-old selenium-deficient white rats. Hematoxylin–eosin staining, ×200 magnification |
4. Conclusions
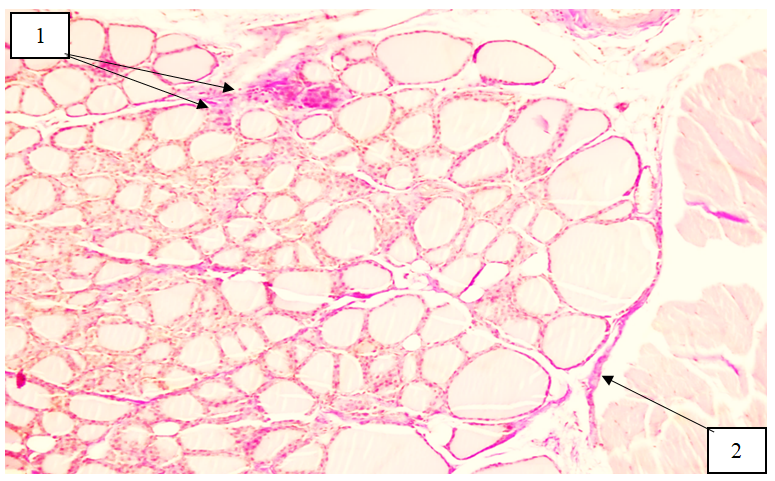
- In selenium-deficient white laboratory rats, notable morphological and morphometric alterations of the thyroid gland were observed. Follicles in the peripheral regions of the gland were relatively larger compared with those in the central zones. A decrease in the height of the epithelial cells was detected, and thyrocytes predominantly exhibited cuboidal to flattened morphology. Selenium deficiency also led to a reduction in follicular diameter, accompanied by a decreased amount of colloid within the follicular lumen and an increase in the density of the colloid material. In hematoxylin–eosin–stained sections, the colloid demonstrated intense eosinophilic staining. Selenium deficiency resulted in impaired degradation of hydrogen peroxide, which led to its excessive accumulation. Consequently, the harmful effects of elevated H₂O₂ were evident throughout the organism, including in the thyroid gland, contributing to structural alterations and functional instability of the tissue.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML