-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4313-4315
doi:10.5923/j.ajmms.20251512.23
Received: Nov. 7, 2025; Accepted: Nov. 23, 2025; Published: Dec. 8, 2025

Optimization of Diagnosis and Therapy of Nephrotoxicity in Patients with Metastatic Breast Cancer Considering Prognostic Factors
Sokhiba S. Mirakhmedova1, Nodir M. Rakhimov2
1Assistant of the Oncology Department at Bukhara State Medical Institute, Bukhara, Uzbekistan
2DSc, Professor, Director of the Samarkand Interregional Hospice, Uzbekistan
Correspondence to: Sokhiba S. Mirakhmedova, Assistant of the Oncology Department at Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Nephrotoxicity remains one of the most common and clinically significant complications of systemic therapy in patients with metastatic breast cancer. The diversity of treatment regimens, including cytotoxic agents, targeted therapies, hormonal drugs, and bisphosphonates, increases the risk of renal damage and often necessitates dose modifications or discontinuation of effective anticancer treatment. The aim of this study is to optimize the diagnosis and management of nephrotoxicity by incorporating prognostic factors that influence its incidence and severity. This article analyzes the key mechanisms of nephrotoxicity, identifies important clinical and laboratory predictors, and reviews current approaches to early detection of renal injury, including the use of novel biomarkers. Special attention is given to personalized treatment strategies based on risk stratification and evidence-based nephroprotective measures. Improving diagnostic methods and therapeutic tactics may reduce the severity of renal complications, enhance the effectiveness of systemic therapy, and improve the quality of life in patients with metastatic breast cancer.
Keywords: Metastatic breast cancer, Nephrotoxicity, Prognostic factors, Renal injury biomarkers, Renal dysfunction diagnostics, Systemic therapy, Chemotherapy, Targeted therapy, Bisphosphonates, Nephrotoxicity prevention, Pharmacological nephroprotection, Personalized treatment, Risk stratification
Cite this paper: Sokhiba S. Mirakhmedova, Nodir M. Rakhimov, Optimization of Diagnosis and Therapy of Nephrotoxicity in Patients with Metastatic Breast Cancer Considering Prognostic Factors, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4313-4315. doi: 10.5923/j.ajmms.20251512.23.
Article Outline
1. Introduction
- Metastatic breast cancer (MBC) remains a major contributor to global cancer-related morbidity and mortality despite significant advances in systemic therapy. Modern treatment approaches - including anthracycline- and taxane-based chemotherapy, platinum derivatives, targeted agents such as CDK4/6 inhibitors, HER2-directed therapies, and bone-modifying agents—have improved survival outcomes but increased the risk of treatment-related toxicities, particularly nephrotoxicity [2,1].Renal complications in patients receiving systemic therapy arise from multifactorial mechanisms, including direct tubular toxicity, glomerular injury, renal microangiopathy, or hemodynamic alterations induced by anti-VEGF agents [3]. Platinum-based drugs such as cisplatin remain among the most nephrotoxic agents used in breast cancer management, frequently causing electrolyte imbalance, acute tubular injury, and reduced glomerular filtration rate [4,7]. Bisphosphonates and denosumab, widely used to manage bone metastases, also carry risks of renal impairment, especially in patients with pre-existing chronic kidney disease [5].Early recognition of nephrotoxicity is critical, yet traditional markers such as serum creatinine often fail to detect early renal injury. Novel biomarkers—including neutrophil gelatinase–associated lipocalin (NGAL), kidney injury molecule-1 (KIM-1), and cystatin C—have demonstrated superior sensitivity for early detection and prognostication of renal dysfunction [6]. Incorporating these biomarkers into clinical practice may enable earlier therapeutic adjustments and reduce irreversible kidney damage.Considering the heterogeneity of patients with metastatic breast cancer, identifying prognostic factors that predispose to nephrotoxicity—such as age, comorbidities, cumulative toxicity from multiple lines of therapy, and baseline renal function—is essential for optimizing treatment strategies. Risk-adapted modifications, including dose adjustments, alternative drug selections, and nephroprotective interventions, play a pivotal role in preventing treatment interruption and protecting long-term outcomes [8].This article examines the mechanisms, diagnostic approaches, prognostic factors, and therapeutic strategies related to nephrotoxicity in metastatic breast cancer. Special attention is given to the integration of prognostic markers and personalized medicine approaches aimed at improving renal outcomes while maintaining effective oncological treatment.
2. Materials and Methods of Research
- A retrospective case–control study was conducted to identify prognostic markers influencing therapeutic outcomes in patients with metastatic breast cancer. The study included a detailed analysis of clinical observations and medical records of women diagnosed with breast carcinoma who met the predefined inclusion criteria. The investigation covered the period from 2022 to 2024 and was performed at the Tashkent City Branch of the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR). The research material consisted of medical data from 105 women with histologically verified breast cancer, stage pT3–4N0M1, who underwent palliative treatment during the study period. The choice of this particular disease stage was purposeful: it allowed examination of a cohort with uniform staging yet significant heterogeneity in clinical presentation and progression patterns. The presence of metastases in the lymphatic system and distant organs enabled a reliable assessment of the biological behavior of the tumor and its prognostic impact on treatment outcomes.Patients ranged in age from 26 to 78 years, with an even age distribution across age groups. The mean age was 56 ± 11.3 years.The cohort was divided into two groups following the case–control design:• Main group (cases): 32 patients who demonstrated documented disease progression (local recurrence or distant metastasis) within 6–24 months after treatment.• Control group: 63 patients who showed no signs of disease progression during the same follow-up period.This stratification enabled comparative evaluation of prognostic factors associated with progression versus stable disease.Upon admission, all patients underwent a comprehensive clinical and diagnostic workup according to the standard protocol for the initial evaluation of breast cancer patients in specialized oncology facilities. This included:• physical examination and medical history• laboratory investigations (complete blood count, biochemical profile, renal function tests)• imaging studies (mammography, breast and abdominal ultrasound, CT or MRI as indicated)• assessment of molecular tumor subtype (ER, PR, HER2 status)• evaluation of metastatic spread (bone scan, PET-CT where available)Given that nephrotoxicity was a major endpoint of the broader research, renal function was evaluated using: serum creatinine and blood urea nitrogen, estimated glomerular filtration rate (eGFR, CKD-EPI formula), urinalysis with proteinuria assessment, electrolyte panel (Na⁺, K⁺, Ca²⁺, Mg²⁺). Advanced biomarkers (NGAL, KIM-1, cystatin C) were assessed in a subset of patients to detect early renal injury.
3. Results and Discussion
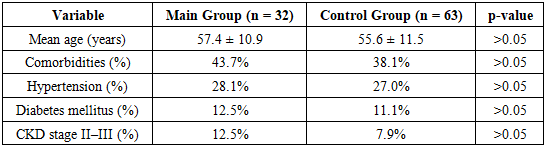
- A total of 105 women with breast cancer stage pT3–4N0M1 were included in the analysis. Of these, 32 patients (30.5%) formed the main group with documented disease progression within 6–24 months, and 63 patients (60.0%) comprised the control group with no evidence of progression during the same observation period.The mean age of the total cohort was 56 ± 11.3 years (range: 26–78 years). There were no statistically significant age differences between the study groups (p > 0.05). Comorbid conditions were recorded in both groups, including hypertension (27.6%), type 2 diabetes mellitus (11.4%), and chronic kidney disease stage II–III (9.5%). The distribution of comorbidities between the groups did not differ significantly.
|
|
4. Conclusions
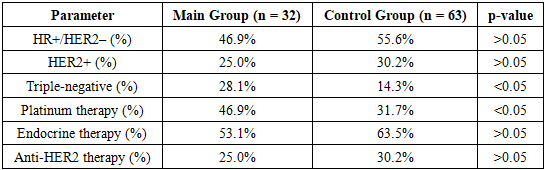
- This study provides important insights into the prognostic significance, clinical patterns, and diagnostic challenges of nephrotoxicity in patients with breast cancer stage pT3–4N0M1 undergoing palliative treatment. By incorporating a case–control design and analyzing 105 clinical cases, the research demonstrated that renal complications are not only frequent but also strongly associated with disease progression, treatment intensity, and baseline patient characteristics. The findings highlight that triple-negative breast cancer, baseline renal dysfunction, platinum-based chemotherapy, and multiple comorbidities are significant predictors of both nephrotoxicity and unfavorable oncologic outcomes. These prognostic factors underscore the crucial need for individualized treatment strategies, particularly for patients with biologically aggressive tumors or pre-existing renal vulnerability.Nephrotoxicity emerged as a major determinant of treatment continuity, contributing to dose reductions, interruptions, and discontinuations—factors known to compromise progression-free survival. This reinforces the importance of early detection and proactive nephroprotection to preserve both renal function and therapeutic efficacy. The study also demonstrated the clinical utility of novel renal biomarkers—especially NGAL and KIM-1—whose superior sensitivity and earlier response compared with conventional markers position them as valuable tools for detecting subclinical kidney injury. Integrating these biomarkers into routine monitoring protocols may significantly enhance timely intervention and prevent irreversible renal damage.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML