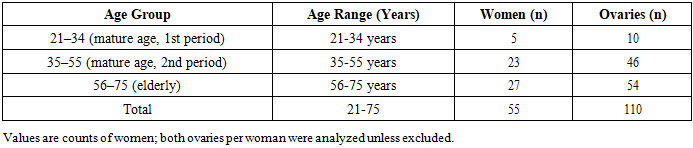-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4307-4312
doi:10.5923/j.ajmms.20251512.22
Received: Nov. 2, 2025; Accepted: Nov. 21, 2025; Published: Dec. 8, 2025

Morphological and Morphometric Features of the Ovaries of Women of Different Age Groups
Firuza Saifulloevna Nabieva
Assistant, Department of Oncology, Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan
Correspondence to: Firuza Saifulloevna Nabieva, Assistant, Department of Oncology, Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: The female ovary undergoes coordinated structural remodeling across the lifespan. High-quality, calibrated morphometry from histologically normal ovaries is limited, especially regarding cortical/medullary thickness, the tunica albuginea, primordial follicle density, and right–left asymmetry. Objective: To quantify age-related changes in ovarian macro- and micro-morphology and to evaluate right–left asymmetry in a cohort of women undergoing hysterectomy with bilateral salpingo-oophorectomy for benign uterine disease. Methods: We performed a retrospective cross-sectional study of 55 women (110 ovaries) aged 21–75 years, treated between January 2024 and January 2025 at Carmen PLUS and the Bukhara branch of the RSSaPMCOaR. Ovaries were macroscopically normal at surgery and histologically free of pathology. Macroscopic measures (length, width, thickness, weight, volume) and histomorphometry on calibrated H&E sections (cortex and medulla thickness, tunica albuginea thickness, and primordial follicle density per mm²) were obtained for each Right and Left ovary. Age was analyzed in strata (21–34, 35–55, 56–75 years) and as a continuous variable. Pre-specified analyses included age-group comparisons, paired right–left testing, and regression models adjusted for BMI, with multiplicity control. Results: Across age, ovaries showed a consistent pattern of cortical and medullary thinning, progressive thickening of the tunica albuginea, and declining primordial follicle density. Patient-level means differed across age strata, and regression analyses supported independent associations of age with these morphometric traits. Right–left comparisons demonstrated asymmetry in selected parameters at the cohort level. Conclusions: Ovarian morphology exhibits reproducible age-related remodeling characterized by cortical/medullary thinning, tunica albuginea thickening, and depletion of primordial follicles, with evidence of lateral asymmetry. These quantitative data provide a structural context for clinical assessment of ovarian reserve and imaging findings.
Keywords: Ovary, Histomorphometry, Age, Cortex, Medulla, Tunica albuginea, Primordial follicles, Right–left asymmetry
Cite this paper: Firuza Saifulloevna Nabieva, Morphological and Morphometric Features of the Ovaries of Women of Different Age Groups, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4307-4312. doi: 10.5923/j.ajmms.20251512.22.
Article Outline
1. Introduction
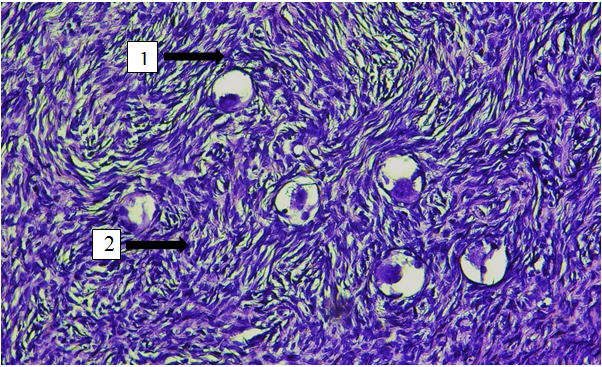
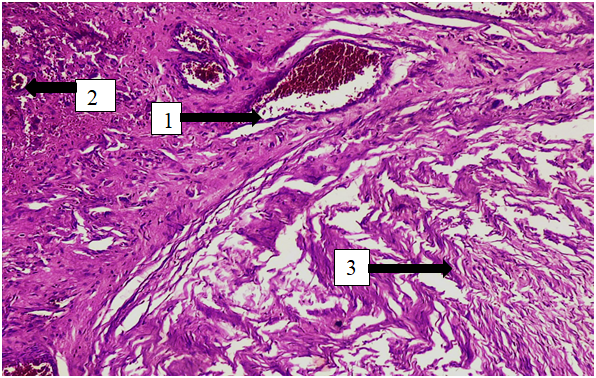
- The development of the female reproductive system is a multistage biological process. During early embryogenesis (2–7 weeks), morphogenesis of both the female and male reproductive systems proceeds in parallel, closely linked to the formation of the excretory system. At this “indifferent stage,” bipotential gonads arise alongside paired Wolffian and Müllerian ducts and the urogenital sinus, while primordial germ cells proliferate, migrate, and colonize the gonadal ridges [1].The ovarian rudiment begins to form by the fifth week of embryonic development. Its origins include:Coelomic epithelium → precursor of follicular cells and some luteal cells.Mesenchyme → gives rise to stromal connective tissue, theca cells, and steroidogenic elements.Gonocytes → precursors of oogonia, which develop into oocytes of different orders [2,10].Morphological remodeling continues throughout life. After the age of 20, ovarian dimensions fluctuate subtly, typically 4–4.5 × 2–2.5 cm with a diameter of 1–2 cm and mass of 6–7.5 g. With advancing age, involuted ovaries shrink to ~2 × 1 × 0.5 cm and 1–2 g [3,8]. A consistent asymmetry is noted, with the right ovary often ~70% larger than the left, a feature of potential physiological and clinical relevance.The ovarian surface also reflects reproductive activity. In infants and prepubertal girls, the capsule appears smooth and pink. At puberty, cyclic ovulation leads to formation of corpora lutea and corpora albicantia, producing surface irregularity. With aging, the capsule becomes coarsely nodular, showing fibrotic depressions [4,9].Histologically, the ovary is organized into cortex and medulla. The cortex comprises spindle-shaped stromal cells resembling fibroblasts within a collagen framework, housing follicles at different developmental stages (primordial, primary, secondary, tertiary/graafian) [5,6]. In reproductive-age women, primordial follicles are located peripherally, while growing follicles occupy deeper cortical zones. With age, the cortex thickens until mid-reproductive life, then thins progressively with menopause. The medulla shows the opposite trend, with greater stromal fibrosis and vascular sclerosis in later life [3,8].Age-dependent changes also affect extracellular matrix and vasculature. By the third decade, cortical stroma increasingly contains collagen bundles and vascular proliferation. After age 50, the capsule thickens, stromal sclerosis intensifies, the tunica albuginea becomes fibrotic, and follicle diversity diminishes. Hyalinosis, vascular sclerosis, and arterial wall thickening appear, ultimately reducing cortical blood supply. In advanced senescence, ovaries may become flattened fibrous plates [2,7].ObjectiveTo systematically quantify age-related morphometric changes in the human ovary, including external dimensions, weight, cortical and medullary thickness, tunica albuginea thickness, and primordial follicle density, and to assess right–left asymmetry across age groups.
2. Materials and Methods
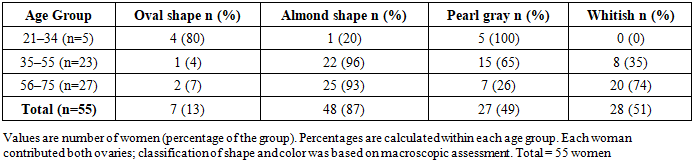
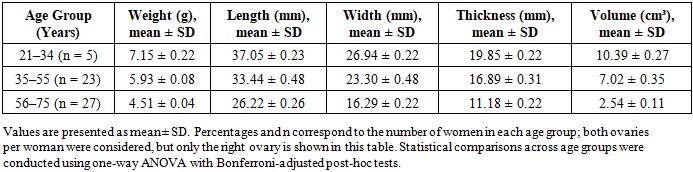
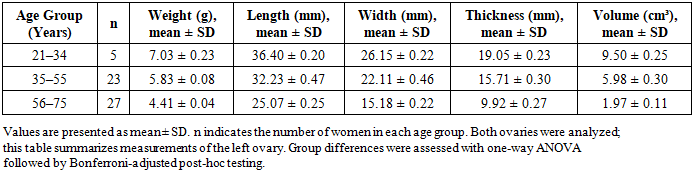
- We retrospectively analyzed data from 55 women treated at Carmen PLUS and the Bukhara branch of the RSSAPMCOaR between January 2024 and January 2025. Patient information (age, obstetric/gynecologic history) and histology/morphometry of excised ovaries were reviewed from institutional archives. Only macroscopically and histologically normal ovaries were included; those with cysts, tumors, endometriosis, or inflammatory changes were excluded.The study cohort was stratified into three age groups:21–34 years (n = 5)35–55 years (n = 23)56–75 years (n = 27)In total, 110 ovaries were analyzed. For each ovary, shape, coloration, weight, and external dimensions (length, width, thickness, calculated volume) were recorded. Histological assessment included measurement of cortex and medulla thickness, tunica albuginea thickness, and primordial follicle counts per mm² on calibrated sections. Representative baseline characteristics and ovarian morphology are summarized in Table 1 (age distribution) and Table 2 (macroscopic appearance).
|
|
|
|
|
|
3. Results
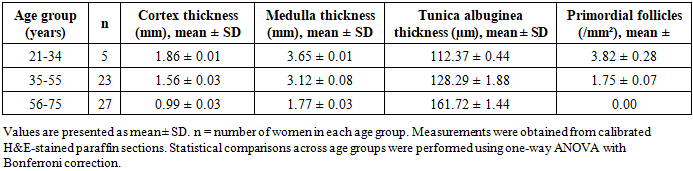
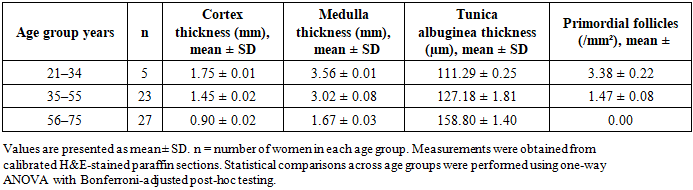
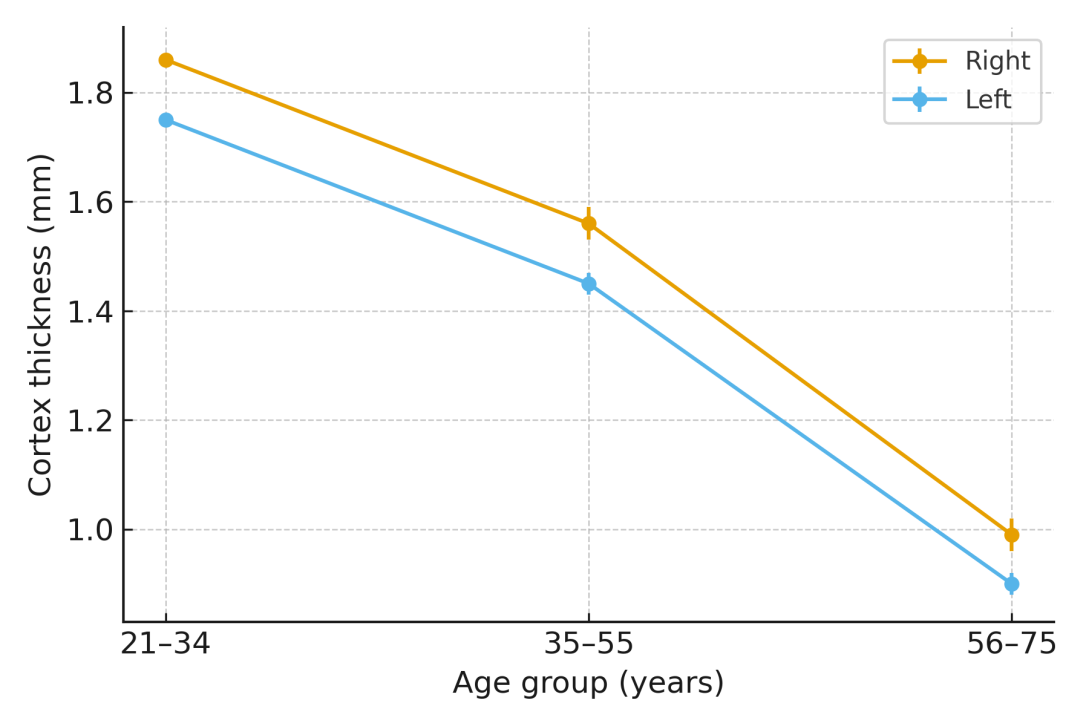
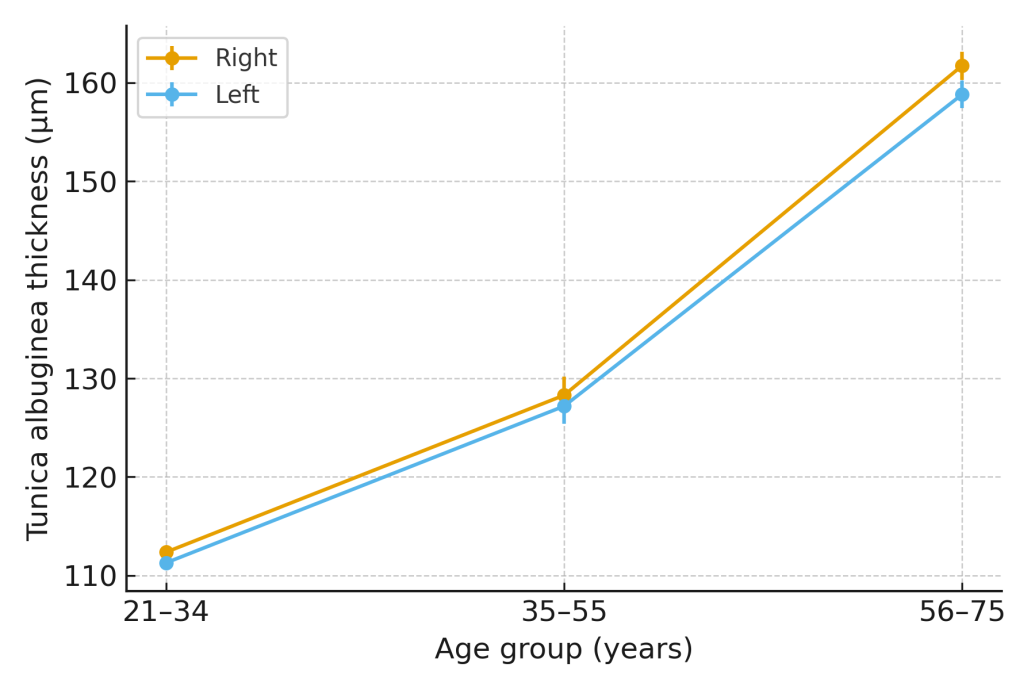
- Quantitative analysis demonstrated a progressive thickening of the tunica albuginea with advancing age (Tables 5 and 6; Figure 3). In women aged 21–34 years, mean tunica thickness was 112.37 ± 0.44 µm in the right ovary and 111.29 ± 0.25 µm in the left. This increased significantly in the 35–55 year group (128.29 ± 1.88 µm and 127.18 ± 1.81 µm, respectively) and further in women aged 56–75 years (161.72 ± 1.44 µm and 158.80 ± 1.40 µm; p < 0.001, ANOVA).
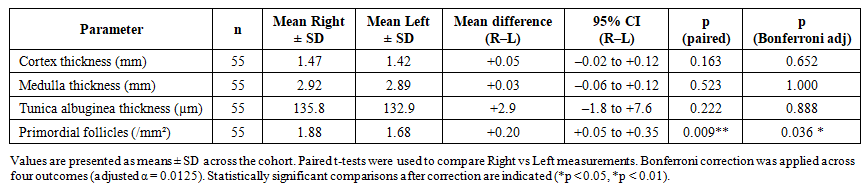 | Table 7. Paired comparison of right vs left ovarian morphometric parameters |
4. Discussion
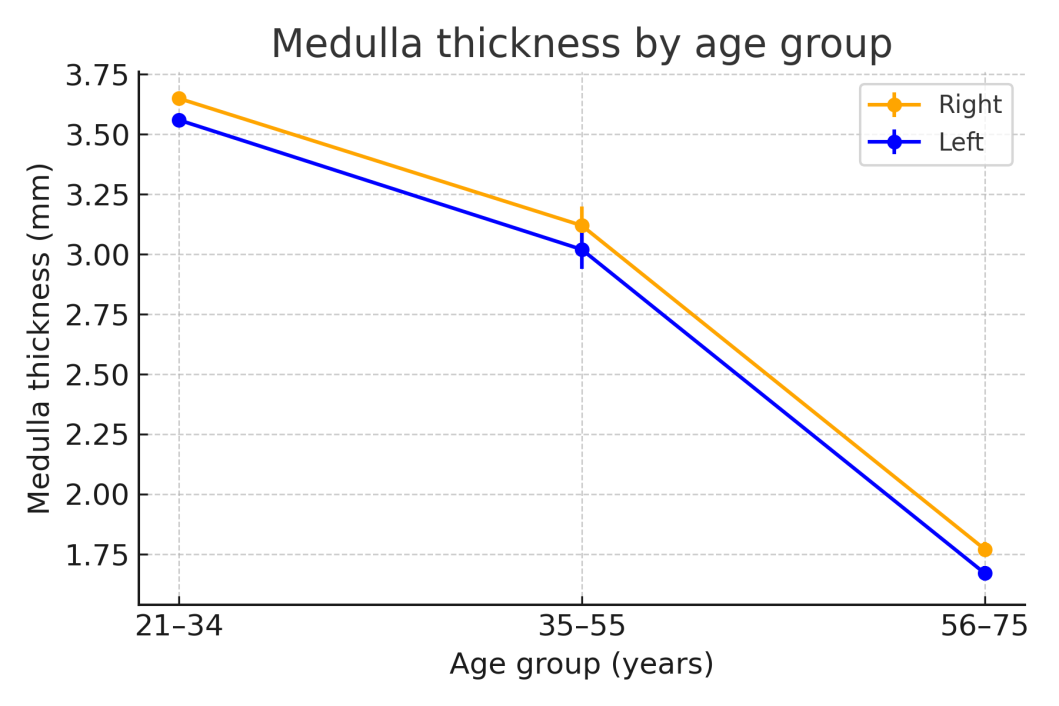
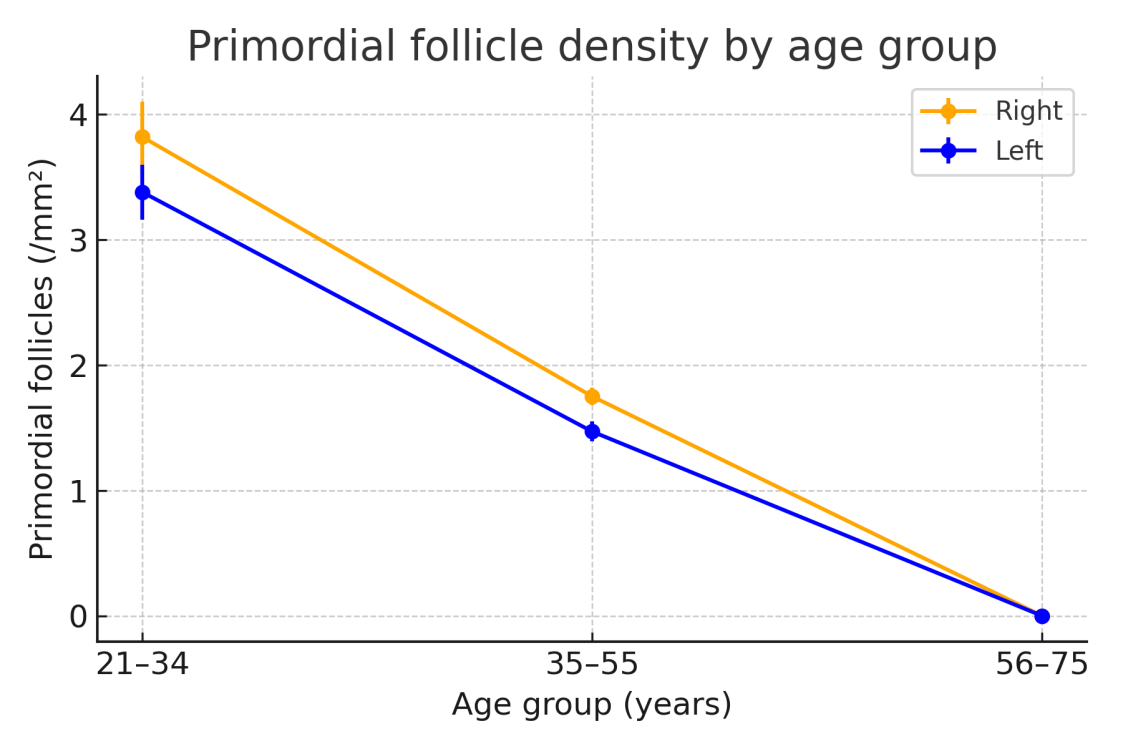
- The present study provides detailed morphological and morphometric evidence of age-related transformations in the human ovary, combining classical histology with quantitative measurements. A consistent pattern of tunica albuginea thickening and progressive cortical and medullary thinning was observed across all age groups, confirming the structural involution associated with reproductive aging. The decline in primordial follicle density with advancing age corresponds to the depletion of the ovarian reserve and reduced endocrine activity, reflecting a gradual transition from reproductive to postmenopausal physiology.Our data align with earlier reports by Karakasi et al. (2023) and Forabosco & Sforza (2007), who documented similar age-dependent morphological alterations and emphasized the role of stromal fibrosis and vascular remodeling in ovarian senescence. The observed thickening of the tunica albuginea likely reflects cumulative fibrotic deposition, while cortical thinning represents follicular exhaustion and loss of active parenchyma.An important finding of this study is the higher follicular density and larger dimensions of the right ovary, confirming the long-suspected hypothesis of right-sided functional dominance. Previous ultrasonographic and morphometric investigations have suggested that the right ovary receives a relatively richer blood supply from the abdominal aorta, which may account for its greater structural and functional resilience. This asymmetry may have subtle implications for ovulatory frequency and fertility potential, though further research is required to establish its physiological impact.
5. Limitations
- The main limitation of this work lies in the small sample size within the youngest age group (21–34 years), which limits comparative statistical power. Additionally, the retrospective and single-center design restricts generalizability to broader populations. Despite these limitations, the findings remain robust due to consistent trends across all measured parameters and appropriate statistical correction.
6. Future Directions
- Future studies integrating morphometric data with hormonal profiling, ultrasound, or MRI-based ovarian volumetry could help link structural changes to functional outcomes. Longitudinal or multiethnic studies may also clarify whether environmental, genetic, or lifestyle factors modulate the rate of ovarian aging.
7. Conclusions
- Ovarian morphology displays pronounced age-related variation influenced by both biological and anthropometric factors. The study demonstrated a significant thickening of the tunica albuginea, thinning of the cortex and medulla, and a marked decline in primordial follicle density with advancing age. Quantitative comparisons confirmed right–left asymmetry, with the right ovary showing consistently higher follicular counts and slightly larger structural parameters, supporting the hypothesis of functional dominance.These results not only reinforce known patterns of ovarian involution but also provide new reference data derived from a Central Asian cohort, enriching global understanding of ovarian aging. The morphometric benchmarks presented here may assist clinicians and researchers in interpreting histological, imaging, and endocrine findings related to ovarian reserve, fertility potential, and reproductive aging. Moreover, these values could serve as a foundation for developing predictive models of reproductive lifespan and guiding fertility preservation strategies in women of different age groups.Author Contributions: Firuza Nabieva – conceptualization, data collection, histological analysis, manuscript drafting.Funding: No external funding was received. Conflicts of Interest: The authors declare no conflicts of interest.
ACKNOWLEDGEMENTS
- The authors thank the Department of Morphology of the Bukhara Branch of RSSAPMCOaR for technical support.
Ethical Approval
- The study protocol was reviewed and approved by the Ethics Committee of the Bukhara State Medical Institute named after Abu Ali ibn Sino, Ministry of Health of the Republic of Uzbekistan (Approval Reference: EC/BSMI/2025/7812456; Date of Approval: 15 January 2024; Expiry: 14 January 2025).All ovarian tissue samples were obtained from surgical procedures performed for benign indications (uterine prolapse and myomas). Written informed consent was obtained from all participants prior to tissue collection, and all data were anonymized in accordance with institutional and international ethical standards.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML