-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4729-4740
doi:10.5923/j.ajmms.20251512.117
Received: Oct. 23, 2025; Accepted: Nov. 16, 2025; Published: Dec. 31, 2025

Intraoperative Ultrasound with Saline-Filled Silicone Applicators: An Innovative Echogenic Marker for Real-Time Neuronavigation
U. U. Altebayev , S. A. Sizdikhojaev , G. T. Achilova , R. R. Uzakova
Republican Scientific and Practical Medical Center of Neurosurgery, Tashkent State Medical University, Tashkent, Republic of Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Intraoperative ultrasound (ioUS) is a valuable real-time navigation tool in neurosurgery, yet its precision can be limited by the absence of stable echogenic landmarks. This study presents an innovative technique employing medical-grade silicone applicators or balloons filled with 0.9% saline solution as echogenic markers to enhance intraoperative orientation and navigation. Ten clinical cases involving supratentorial, brainstem, and spinal lesions were analyzed. The proposed markers demonstrated high echogenic contrast, stable visibility, and biocompatibility, enabling precise localization of lesions, safe definition of surgical trajectories, and verification of resection completeness. Compared with traditional cottonoid markers, saline-filled silicone applicators exhibited superior stability, adaptability to various anatomical conditions, and minimal tissue trauma. Their integration into ioUS workflows provided a cost-effective, easily reproducible, and dynamic alternative to neuronavigation and intraoperative MRI. The technique enhances surgical safety, minimizes injury to eloquent structures, and improves the radicality of tumor resections, making it a practical addition to modern neurosurgical armamentarium.
Keywords: Intraoperative ultrasound, Echogenic marker, Neuronavigation, Saline-filled silicone applicator, Brain tumor surgery, Real-time imaging, Neurosurgery, Intraoperative navigation, Brain shift compensation, Minimally invasive surgery
Cite this paper: U. U. Altebayev , S. A. Sizdikhojaev , G. T. Achilova , R. R. Uzakova , Intraoperative Ultrasound with Saline-Filled Silicone Applicators: An Innovative Echogenic Marker for Real-Time Neuronavigation, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4729-4740. doi: 10.5923/j.ajmms.20251512.117.
Article Outline
1. Introduction
- The technology of intraoperative ultrasonography (IOUS) was first presented by Kikuchi and colleagues (Japan) at the Second International Congress on Acoustics held in Cambridge, Massachusetts, USA, in 1956 [25]. Subsequent research confirmed its diagnostic value in localizing intraparenchymal pathological foci [5,8,42]. In 1978, Reid reported the first use of two-dimensional B-mode for intraoperative ultrasound mapping [36].Despite these publications, after the introduction of ultrasound into neurosurgical practice in the 1950s, the method did not gain widespread adoption and was not integrated into daily clinical routine. This was due to both the technological limitations of early ultrasound systems and the rapid development of alternative neuroimaging techniques. Specifically, the widespread introduction of computed tomography (CT) in the 1970s, followed by magnetic resonance imaging (MRI) in the 1980s, significantly diminished interest in the ultrasound method. However, despite the obvious advantages of CT and MRI, these technologies did not provide truly intraoperative, real-time navigation, which limited their applicability in dynamically changing intraoperative conditions.The efficacy of intraoperative ultrasonography (IOUS) has been confirmed in a wide range of clinical scenarios, including the localization of subcortical and deep-seated lesions, revision and replacement of ventriculoperitoneal shunts in patients with hydrocephalus, performance of stereotactic biopsies for deep-seated lesions, and diagnosis of intraventricular hemorrhages in newborns [4,7,9,11,16,26,43,45]. The use of IOUS allows for the reconstruction of spatial relationships between a pathological focus and anatomical landmarks, facilitating the selection of an optimal surgical approach, minimization of damage to healthy tissue, and, consequently, improvement of functional outcomes.However, even with the use of modern preoperative imaging modalities, such as 3 Tesla magnetic resonance imaging (MRI) with slice thicknesses of up to 1 mm, precise intraoperative localization of deep-seated or intraparenchymal lesions without a cortical representation remains a challenging task. An error of even a few millimeters can lead to unintentional resection of functionally significant areas, increasing the risk of postoperative complications and mortality.To address this problem, various materials capable of serving as echogenic markers for IOUS and providing real-time anatomical orientation have been proposed in the literature [14,46]. Despite the shared concept, none of these approaches gained widespread acceptance or were supported by systematic studies.In the present work, we propose a modified technique based on the use of medical silicone applicators or balloons filled with 0.9% NaCl solution (saline) as echogenic markers. Unlike the cottonoid patties used previously, such devices possess stable acoustic properties, reproducible echogenicity, and biocompatibility. An additional advantage is the ability to adjust the balloon's volume and shape, allowing adaptation to the specific features of the surgical cavity and reducing the risk of mechanical impact on brain tissue.This technique, integrated into clinical practice since the early 2000s, has proven its practical value and has been described in a number of publications [13,23,24,40,41]. Its application provides clearer visualization of intraoperative anatomy, assists the surgeon in determining the optimal surgical approach, enables precise trajectory planning towards the pathological focus, and increases the extent of resection while simultaneously reducing the risk of damage to functional structures.
2. Materials and Methods
- This study presents a modified technique based on the use of saline-filled silicone applicators as echogenic landmarks for intraoperative ultrasonography. The technique was illustrated by a series of clinical cases performed by a single surgeon. Ten patients who underwent surgical treatment using this technique starting from 2006 were retrospectively selected (Table 1): five patients with subcortical lesions, two patients operated on using an anterior transcallosal approach, and three patients with pathology of the brainstem and spinal cord.Intraoperative ultrasonography was performed using the Aloka Prosound α10 system (Hitachi Aloka Medical, Ltd., Tokyo, Japan) with three types of transducers: UST-536 ("hockey stick," linear, 4.44–13.33 MHz), UST-9120 (neonatal convex, 5.0–10.0 MHz), and UST-533 (microsurgical linear, 7.5–13.0 MHz). In routine clinical practice, the convex neonatal probe was predominantly used for supratentorial lesions. The "hockey stick" transducer was used for spinal cord procedures. The microsurgical probe was used in limited surgical approaches, including surgeries on the brainstem and medulla oblongata, as well as for small subcortical lesions and for surgeries performed in the sitting position.Flat silicone applicators of four sizes, made of medical-grade silicone and filled with 0.9% NaCl solution, were used as echogenic landmarks. This material is characterized by biocompatibility, resistance to deformation, and stable acoustic properties. Due to their high echogenicity, the silicone applicators were clearly visualized on ultrasound as hyperechoic structures. The use of various sizes allowed the technique to be adapted to specific anatomical conditions. Small applicators were used for brainstem and spinal cord procedures, as well as for the resection of small subcortical foci, while larger ones were used for deep-seated or extensive lesions. The size and shape of the applicators could be modified according to intraoperative needs.Unlike cottonoid patties, silicone applicators are easily distinguishable from surrounding structures with increased echogenicity, such as hemostatic materials or blood clots [2,31]. To enhance identification accuracy, a technique of displacing and returning the applicator using its attached thread was employed, allowing the surgeon to reliably distinguish it from surrounding tissues. This technique has been used in clinical practice by the senior author and proved particularly useful during the initial learning phase for young specialists.During intraoperative ultrasonography (IOUS), it is recommended to perform scanning in two anatomical planes simultaneously whenever possible. This approach allows for more accurate three-dimensional orientation of the pathological focus relative to the anatomical structures of the brain and spinal cord, significantly enhancing the precision of surgical navigation. The most informative planes for most supratentorial lesions are the coronal and sagittal planes, while axial scanning provides optimal orientation for pathology of the insular region. For lesions of the cerebellum, brainstem, and spinal cord, combinations of axial and sagittal planes are most valuable for spatial orientation.To improve orientation efficiency, it is crucial that the angles of the ultrasound scanning planes closely correspond to physiological anatomical landmarks and align with the planes of preoperative imaging. Discrepancies in angles can lead to disorientation, especially for surgeons using the technique for the first time. The introduction of echogenic markers in the form of medical silicone applicators or balloons filled with saline significantly simplifies this process. Due to their clear acoustic properties and precise positioning capability, these applicators produce stable echo signals that are easily correlated with preoperative MRI and 3D reconstruction results, thereby reducing the risk of errors in intraoperative navigation.The surgeon must be proficient in both traditional anatomical topography of the brain and its ultrasonic representation. The use of IOUS with silicone balloons as echogenic landmarks allows for more reliable identification of the surgical trajectory, minimizes damage to functionally significant tissues, and increases the accuracy of resection. High-quality additional imaging modalities also play an important role—preoperative magnetic resonance imaging (3 Tesla, slice thickness 1 mm), preoperative three-dimensional reconstructions (e.g., using OsiriX software), and intraoperative echo signals from silicone applicators—which together form a comprehensive orientation system.In practical work, planning of the surgical approach begins at the preoperative stage: 3D reconstruction is performed using OsiriX, correlating the pathological focus with cortical gyri, sulci, and major blood vessels. The construction of vascular network reconstructions is particularly important because, unlike parenchymal structures, superficial veins are not subject to shift due to cerebrospinal fluid loss and maintain stable topographic positions. This makes them reliable landmarks for IOUS and the use of silicone applicators.For optimal transmission of ultrasound waves, the surgical cavity must be completely filled with saline, ensuring maximum effective acoustic coupling between the transducer and the brain tissue. Silicone applicators, filled with the same solution, become part of a unified medium and produce a clear, reproducible echo signal. The patient's position is also important: it should be adjusted to maintain even distribution of the saline and continuous irrigation of the surgical site throughout the procedure.Thus, the combination of multiplanar ultrasonography, high-resolution preoperative reconstructions, and the use of silicone applicators as echogenic markers creates a reliable and dynamic intraoperative navigation system. This ensures precise definition of the surgical trajectory, reduces the risk of damage to healthy tissue, and significantly enhances the safety and radicalness of interventions for complex intraparenchymal lesions.Characteristics of Saline-Filled Silicone Applicators/Balloons:Medical-grade silicone is biocompatible and inert;Stable echogenicity due to filling with saline solution;Variety of shapes and sizes, allowing for individual adaptation;Clear visualization; easily displaced and identified by manipulating the attached thread;Minimal trauma; smooth surface reduces the risk of adhesion and mechanical damage;Versatile: applicable for supratentorial, brainstem, and spinal lesions.
3. Results
- This paper presents a technique developed by the authors, illustrated by clinical observations of patients with pathological formations of varying nature and different localizations within the cranial cavity. All cases were selected from a large series of surgical interventions performed by the lead author using intraoperative ultrasonography, where the key navigational element was the use of silicone applicators or balloons filled with saline.The presented clinical examples reflect both the basic principles of the method's application and its efficacy in solving various technical and navigational tasks. The silicone applicators served as echogenic landmarks, generating stable ultrasound signals and allowing the surgeon to correlate the intraoperative image with preoperative MRI data and three-dimensional reconstructions with high precision. Thus, the technique provided clear spatial orientation within the surgical field, even in the presence of significant shift or deformation of brain structures due to cerebrospinal fluid loss or partial tumor resection.Furthermore, the analysis of clinical cases demonstrated that the use of saline-filled silicone balloons minimizes the risk of errors in defining the boundaries of a pathological focus, particularly in areas of high functional significance—cortical zones adjacent to pathways, as well as near major vessels. In these scenarios, conventional ultrasonography may yield limited results due to uneven distribution of the acoustic signal, whereas the introduction of echogenic markers creates additional stable reference points.Thus, the presented observations demonstrate the practical value and broad capabilities of the described technique. It enhances the accuracy of intraoperative navigation, improves control over the extent of resection, reduces the likelihood of residual tumor mass retention, and increases the overall safety of the intervention. Systematic experience with its application confirms that this approach can be considered a reliable tool in the arsenal of modern neurosurgical technologies.Case 1A 9-year-old female patient was admitted with a two-month history of epileptic seizures. Magnetic resonance imaging of the brain revealed an intracerebral lesion located in the upper portions of the right precentral gyrus. Given the clinical manifestations and the topography of the lesion, a decision was made to proceed with surgical treatment.The surgery was performed with the patient in the supine position, undergoing a right frontoparietal craniotomy. To ensure precise intraoperative navigation and optimize ultrasound control, the technique utilizing saline-filled silicone applicators was employed. The applicators were placed within the surgical site in such a way as to create clear echogenic landmarks, allowing for highly reliable differentiation of the tumor boundaries from adjacent functionally significant areas of the cortex and subcortical structures. The use of this technique significantly improved the quality of visualization and reduced the risk of resection beyond the pathological focus.Radical tumor removal was performed, achieving gross total resection. Histopathological examination confirmed the diagnosis of a Grade II oligodendroglioma according to the World Health Organization (WHO) classification. Due to the favorable histological variant and complete tumor removal, the patient did not receive adjuvant therapy.The postoperative course was uneventful. Neurological status remained stable, and epileptic seizures ceased completely. Follow-up magnetic resonance imaging performed 3 months after the intervention showed no signs of residual or recurrent tumor, confirming the effectiveness of the applied technique (Fig. 1 a–f).
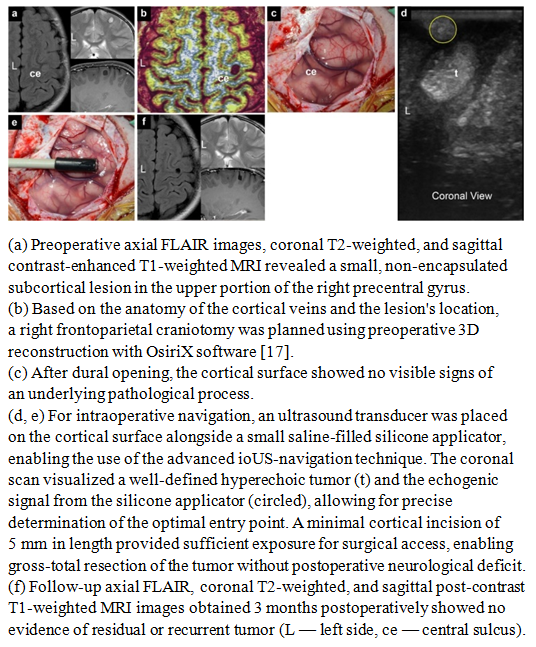 | Figure 1 |
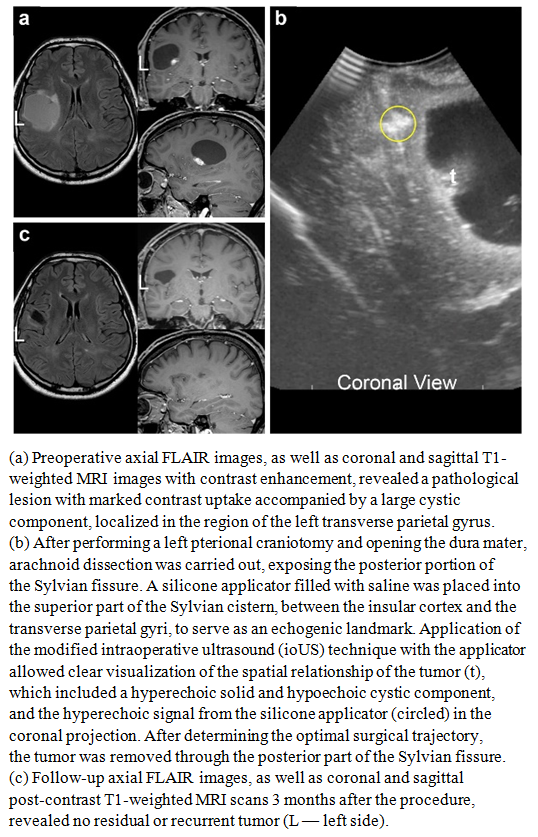 | Figure 2 |
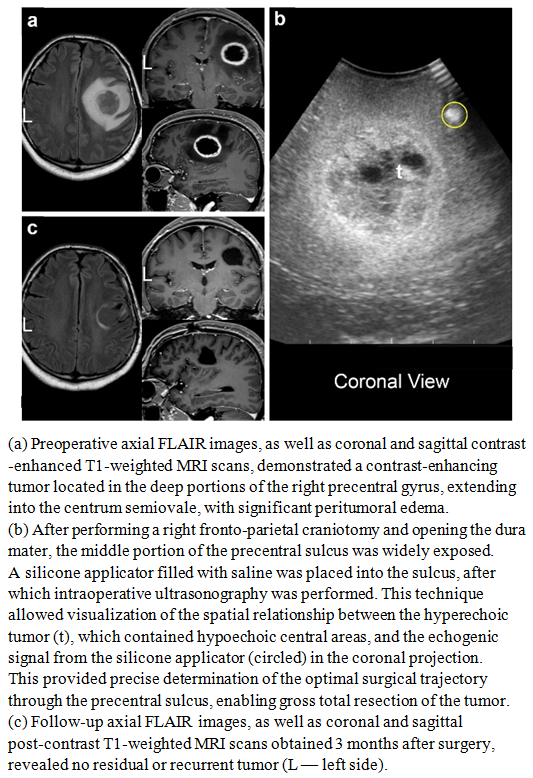 | Figure 3 |
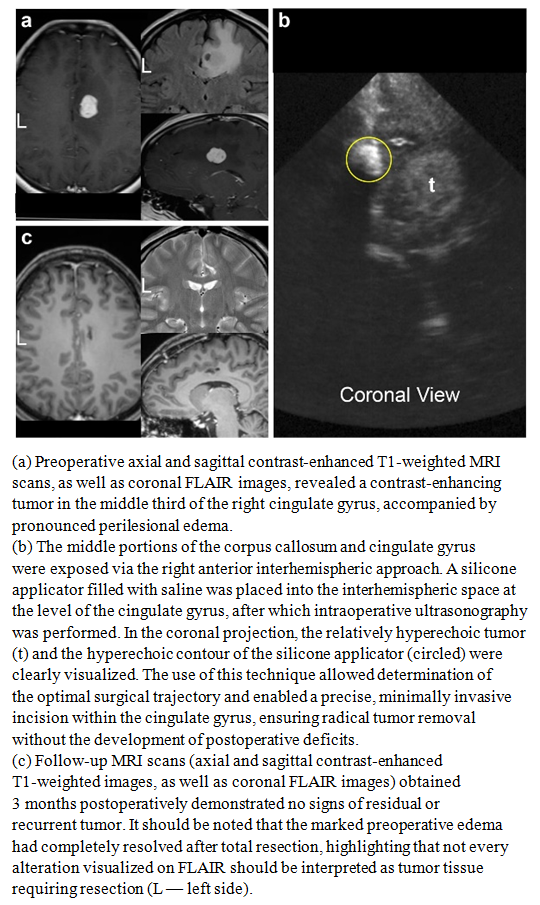 | Figure 4 |
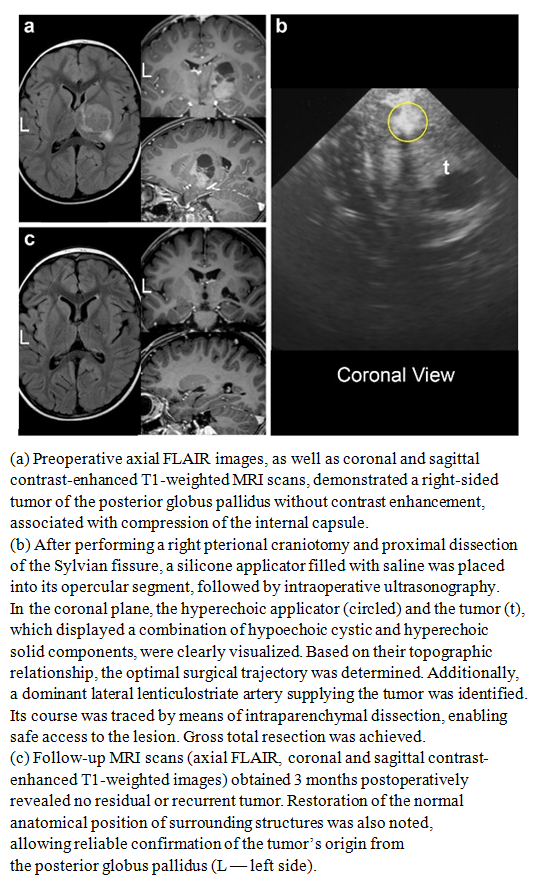 | Figure 5 |
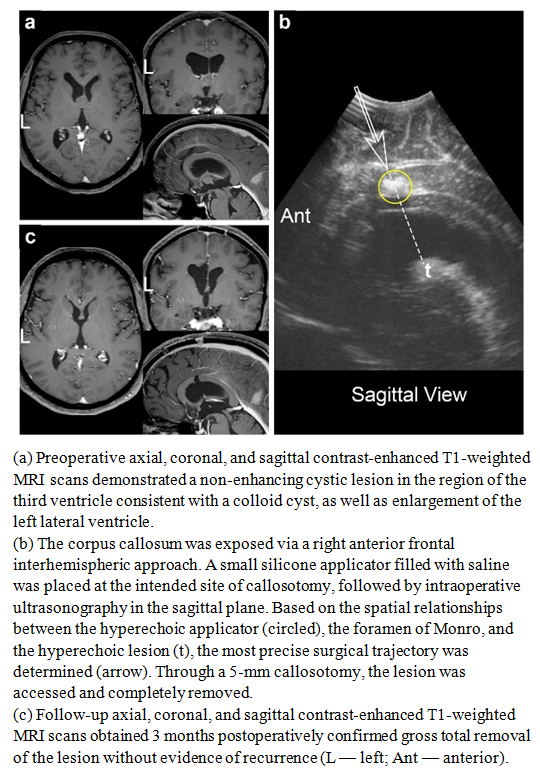 | Figure 6 |
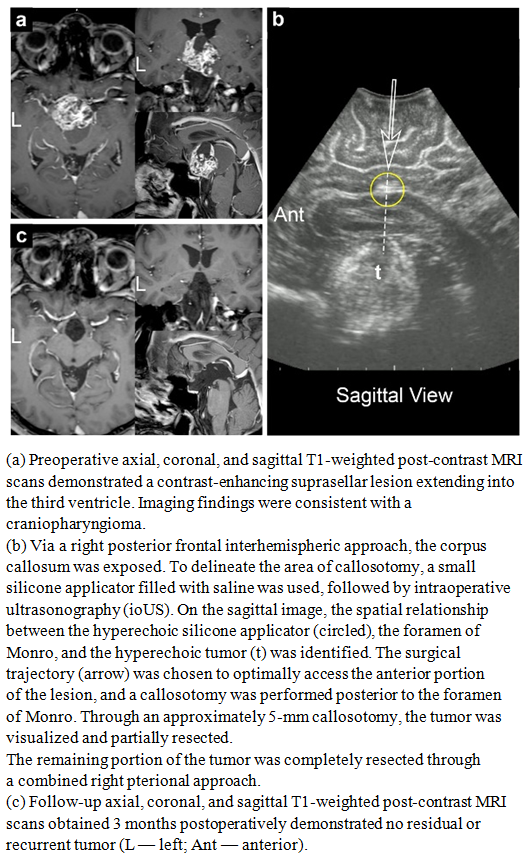 | Figure 7 |
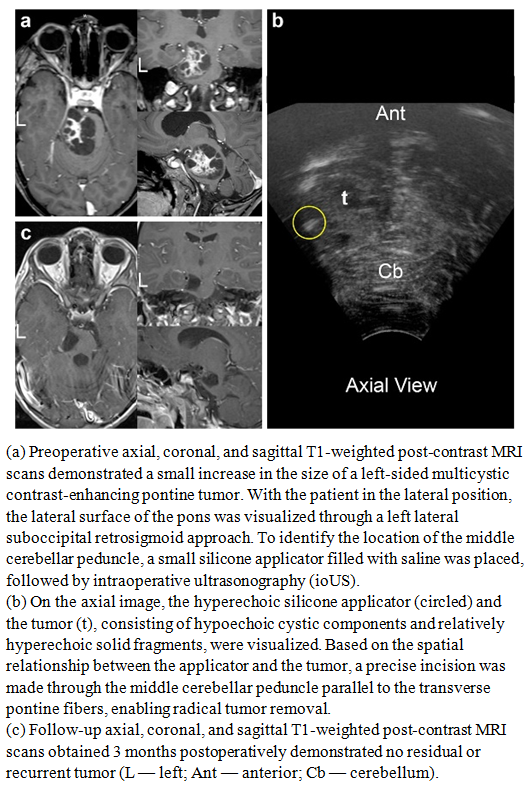 | Figure 8 |
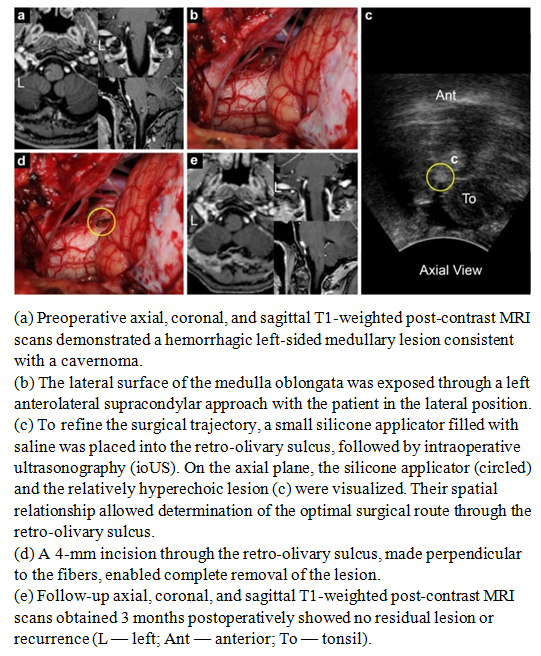 | Figure 9 |
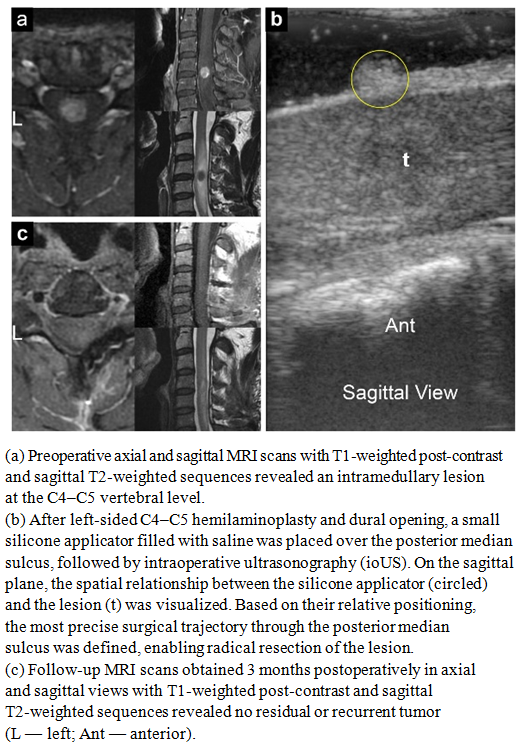 | Figure 10 |
4. Discussion
- The success of surgical intervention largely depends on the accurate determination of the anatomical location of the lesion and the planning of the optimal approach to it. The most appropriate site for craniotomy may be determined using preoperative imaging modalities, 3D reconstructions, neuronavigation systems, and analysis of the spatial relationship between topographic landmarks and the lesion [13,17,19,23,37,38,40,44]. Since the cortical exposure zone is limited, achieving precise anatomical orientation during surgery is not always a simple task. Gyri, sulci, and the cortical vascular network can serve as landmarks for defining the projection of superficial lesions onto the cortex [13,17,23,40].However, access to subcortical or deeply located lesions, which leave no trace on the cortical surface, remains a challenging task for neurosurgeons. Over the past two decades, a wide range of preoperative radiological imaging modalities (MRI, CT, DSA, PET, fMRI, etc.) have been increasingly applied in neurosurgical practice for intraoperative navigation [3,51]. These methods require topographic correlation of the patient’s position during surgery with preoperative imaging and provide anatomical orientation throughout the surgical procedure.When dealing with deeply located or subcortical lesions, intraoperative ultrasonography (ioUS) with guiding silicone applicators or balloons filled with saline has proven particularly useful, as it enables accurate determination of the optimal surgical trajectory and minimizes damage to normal tissue.During surgery, cerebrospinal fluid drainage, gravity, patient positioning, and anesthetic effects may cause brain shift and reduce the accuracy of neuronavigation systems [32,33,39]. Particularly in cases where modern neuronavigation systems lose precision, intraoperative ultrasonography (ioUS) can be easily and safely employed to generate a three-dimensional configuration of the lesion, providing superior anatomical orientation during surgery compared to other navigation methods. ioUS enables tumor visualization in multiple planes from different angles, which is not feasible with standard navigation technologies. Moreover, due to its dynamic nature, any intraoperative anatomical deformations can be promptly detected throughout the procedure.In 1983, Gooding and colleagues proposed the use of ioUS with hyperechoic surgical sponges soaked in saline to assess the course of dissection, the progress of resection, and the extent of tumor removal [14]. In the same year, Voorhies and colleagues described the use of ioUS with a small silicone applicator filled with saline to determine the spatial relationship between the tumor and surface topography, as well as to select the optimal surgical trajectory angle. They also suggested using a biopsy catheter under ultrasound guidance for tract marking and dissection guidance [47]. The following year, a paper was published describing the use of a silicone applicator in subcortical tumors to verify the surgical approach in cases where the tumor was not encountered at the expected depth [46].Despite these reports, the basic principles of applicator use and their primary purpose were limited merely to trajectory verification for locating subcortical tumors inaccessible during the initial surgical exploration [28]. Although the use of such applicators as acoustic markers had previously been described by Voorhies and colleagues [46], the method never became widespread and was gradually almost forgotten. We have revived and refined the use of silicone applicators as acoustic markers in routine neurosurgical practice, which allows for safe and precise orientation when dealing with deeply located or subcortical lesions.
5. Comparison of ioUS with Silicone Applicators, Neuronavigation, and Intraoperative MRI
- In our department, all modern intraoperative technologies are available, including intraoperative MRI (ioMRI), intraoperative ultrasonography (ioUS), ICG, 5-ALA, O-arm, neuronavigation, and neuromonitoring. We apply these tools during surgery, taking into account their capabilities and limitations, in order to achieve the best surgical outcomes.We have found that neuronavigation has certain limitations compared to ioUS. While ioUS provides true real-time imaging and is not affected by brain shift, neuronavigation relies on preoperative imaging and does not offer continuous real-time guidance. Thus, intraoperative brain shift remains a major issue when using neuronavigation, reducing its accuracy and reliability during surgery.To overcome this limitation, some groups have developed navigation systems integrated with ultrasound [6,10,15,20,27,28]. This is an effective technique for resolving problems related to intraoperative brain shift. However, the resolution of such systems was lower than that of our intraoperative ultrasound system. Since high resolution is critically important for ioUS, and our technique of intraoperative ultrasound with guiding silicone applicators provides full real-time visual support, the senior author preferred this method when working with intraparenchymal CNS lesions.Nevertheless, standard neuronavigation remains the method of choice for craniotomies and skull base surgeries, since most bony structures remain in place throughout the operation, providing stable anatomical landmarks.In our opinion, comparing intraoperative ultrasound (ioUS) with intraoperative MRI (ioMRI) is inappropriate for several reasons. The use of ioMRI requires additional infrastructure, MRI-compatible instruments, and specific patient positioning. One of the key advantages of our ioUS technique with guiding silicone applicators is that it requires no additional preparation or specialized instruments. Only silicone applicators, which are used in every case, and an ultrasound machine—available in almost any hospital—are necessary.Furthermore, the use of ioMRI for real-time navigation is not feasible. Typically, ioMRI is employed to assess residual tumor during surgery and provides higher accuracy compared to ioUS. However, even with all technological advancements, ioMRI cannot identify all residual tumor tissue or reliably differentiate edema from tumor. Repeated use of ioMRI is time-consuming and inconvenient, particularly when contrast agents are used for the first scan. In contrast, our technique can be repeated at any time at the surgeon’s discretion and provides the most valuable real-time navigation.If ioUS is compared to a car, ioMRI can be likened to an airplane. Both are necessary, but the car is used daily and is simple to operate, whereas the airplane is needed only occasionally and is more complex to use. There are certain “distances” that can only be covered by a car, only by an airplane, or by both. Similarly, we may use ioMRI alone, ioUS alone, or a combination of both methods in selected cases.Any neurosurgeon familiar with ultrasound imaging and able to integrate 3D MRI data with surgical anatomy can master the ioUS technique with guiding silicone applicators within a few minutes. For surgeons without prior ultrasound experience, assistance from colleagues in the radiology department may be helpful during the initial training phase. However, once the technique is mastered, we strongly recommend that intraoperative ultrasound examination be performed by the neurosurgeon conducting the operation.It is important to note that the use of ioUS does not increase surgical time, while it significantly improves both accuracy and safety. Nevertheless, ioUS has certain limitations, primarily related to the operator and technical execution, such as artifacts, use of inappropriate accessories, insufficient skills, or lack of experience [29,34]. In selected cases, the use of ioMRI or contrast-enhanced ultrasound may yield more accurate results, particularly for assessing residual tumor during surgery [10,30,35].Thanks to its ability to provide continuous real-time imaging and immunity to brain shift and intraoperative parenchymal changes, ioUS plays a key role in our practice as an intraoperative imaging tool. Moreover, it is relatively inexpensive and available in almost every hospital, making it easy to implement in neurosurgical operating rooms.As demonstrated in this study, the ioUS technique with guiding silicone applicators allows for safer and more precise surgery. It promotes maximal preservation of healthy parenchyma during intervention and, consequently, leads to better clinical outcomes with reduced morbidity. In addition to its ease of use and relatively low cost, we observed the high speed, reliability, and exceptional utility of this technique.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML