-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4702-4706
doi:10.5923/j.ajmms.20251512.112
Received: Dec. 3, 2025; Accepted: Dec. 27, 2025; Published: Dec. 29, 2025

The Influence of Experimental Metabolic Syndrome on Heart Morphology of Offenders
Sh. K. Bobokhonov1, S. M. Akhmedova2
1Tashkent State Medical University, Termiz Branch, Uzbekistan
2Tashkent State Medical University, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This article experimentally studied the effect of the development of metabolic syndrome in the mother's body on the morphology of the heart of the offspring. The experiment was conducted on the example of rats, and the structure and histological changes in the hearts of children from the control and metabolic syndrome groups were analyzed. During the study, lipid metabolism, glucose content, arterial vessel diameter, and the dynamics of dystrophic changes in myocardial tissue were assessed using morphometric and histological methods. According to the results, it was found that children born against the background of metabolic syndrome have a high risk of developing pathomorphological changes in the heart, such as disorganization of connective fibers in the walls of dystrophic arterioles and capillaries, as well as changes in the form of protein dystrophy of myocardial muscle fibers, myofibril fibrils.
Keywords: Metabolic syndrome, Heart morphology, Fetal development, Lipid metabolism, Myocardial dystrophy, Arterial vessels
Cite this paper: Sh. K. Bobokhonov, S. M. Akhmedova, The Influence of Experimental Metabolic Syndrome on Heart Morphology of Offenders, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4702-4706. doi: 10.5923/j.ajmms.20251512.112.
1. Introduction
- Metabolic syndrome has become a major global health problem. It is known to include several pathological conditions, such as obesity, hypertension, insulin resistance, hyperglycemia, and dyslipidemia. In particular, when this syndrome is observed in the mother, it has a significant impact not only on the health of the mother, but also on the development of the fetus and the early onset of cardiovascular diseases in the child's later life [1,9].Objectives: Studies have shown that metabolic disorders in the mother can affect the formation of the heart, vascular structure, and function of the fetus. This is associated with the concept of "fetal programming", in which the child's body undergoes certain epigenetic changes in the process of adaptation to pathologies in the mother's body. As a result, the child is predisposed to atherosclerosis, hypertension, insulin resistance, and other cardiovascular diseases [2,4,7].Hyperglycemia and hyperinsulinemia associated with metabolic syndrome in some women disrupt the normal development of the fetal cardiovascular system. This process increases the risk of congenital heart defects, tachycardia, arrhythmias, or subsequent cardiac dysfunction in the child [3,8]. Maternal obesity is also a high risk factor [4]. Obesity can lead to impaired lipid and glucose metabolism in the fetus, hypertrophy of the heart muscle tissue, and functional insufficiency [5].Therefore, early detection, correction, and preventive measures for metabolic syndrome in women of reproductive age are important not only for protecting maternal health, but also for the healthy development of the future generation and the prevention of cardiovascular diseases.Taking the above into account, we set ourselves the goal of studying the heart morphology of offspring born to mothers with metabolic syndrome.
2. Materials and Methods
- The studies were conducted from 2022 to 2025 in the vivarium of the Termez branch of the Tashkent Medical Academy. For the study, 15 white laboratory mother rats weighing 160-180 grams and 120 baby rats born from them were used. The white rats taken for the experiment were divided into 2 groups.The first group was the control group, consisting of 7 female rats weighing 180-220 grams, without clinical signs of somatic and infectious diseases. The rats in the control group were constantly fed a traditional diet, with free access to food and water. After the rats became pregnant and gave birth to their offspring, 1.0 ml of saline solution was injected into the stomach of the mother rats every morning during the lactation period. A transvaginal catheter was used as a probe.In the second group, we induced an experimental metabolic syndrome model in 8 female rats weighing 180-220 grams. After the rats were healthy, and signs of infectious and somatic diseases were excluded, they were fed a diet rich in fat and carbohydrates. The diet of the rats consisted of 60% laboratory food, 20% mutton fat, and 20% fructose. Drinking water was replaced with a 20% fructose solution. After the start of the experiment, male rats were added to the female rats until it was confirmed that metabolic syndrome had been induced, and during the period of pregnancy, the female rats were fed a full diet rich in fat and carbohydrates.Laboratory white rats in the control and experimental groups were kept under the same conditions of the vivarium. The number of rats in each cage did not exceed 6. Rat pups were euthanized under ether anesthesia on days 3, 7, 14, 21, and 28 after birth. The body weights of the control and experimental laboratory white rats were measured. Blood was also collected from the tail vein of the mother and pups, and the levels of glucose, triacylglycerol, total cholesterol, LDL (low-density lipoprotein), and HDL (high-density lipoprotein) were determined. Plasma glucose levels were determined using a glucometer (Accu-Chek Performa; Roche Diagnostics, Indianapolis, Indiana, USA). Total cholesterol, high-density lipoprotein (HDL), and total cholesterol were determined using an automatic biochemical analyzer (Mindray, BS-200, China). Tissues from the ventricular myocardium and interventricular septum were obtained for histological examination. Myocardial tissue was fixed in 10% formalin solution, processed in growing alcohol and prepared paraffin blocks. Histological preparations of 8-12 μm were prepared from the prepared paraffin blocks and stained with hematoxylin and eosin.Morphometric examinations were performed using the G.G. Avtandilov method and the NanoZoomer (REF C13140-21.S/N000198/HAMAMATSU PHOTONICS /431-3196 JAPAN) Hamamatsu (QuPath-0.4.0, NanoZoomer Digital Pathology Image) morphometric computer program. The obtained data were analyzed in the statistical section of Microsoft Excel 2010 to determine the arithmetic mean Mni, the average error of relative measurements m and the precision coefficient t. Micrographs of histological preparations were taken using a CX40 model OD400 camera microscope.Mother rats with metabolic syndrome in the experimental group decreased their food and water intake during the first 10 days of the experiment, and in the following days, they adapted to food, kept calm, and increased their food and water intake. During the experiment, obesity, hyperglycemia, hyperlipidemia and other metabolic changes were observed in rats fed a modified diet for 4–6 weeks. At the beginning of pregnancy, the body weight of rats of the experimental group was on average 10 g (4.2%) higher than that of the control group. This difference indicates the onset of body fat accumulation when experimental group rats were exposed to a metabolic syndrome-inducing diet (60% laboratory chow, 20% sheep fat, 20% fructose, and 20% fructose solution). By the 2nd week of pregnancy, the average weight of the rats in the experimental group was 320 g, which was 50 g (18.5%) higher than that of the control group. The largest difference was observed on the 21st day of pregnancy (3rd week): the rats in the experimental group weighed an average of 420 g, and in the control group - 300 g (Fig. 1). This was a difference of 120 g or 40% (p<0.05), indicating the aggravating effect of metabolic syndrome associated with obesity and hyperhydration.
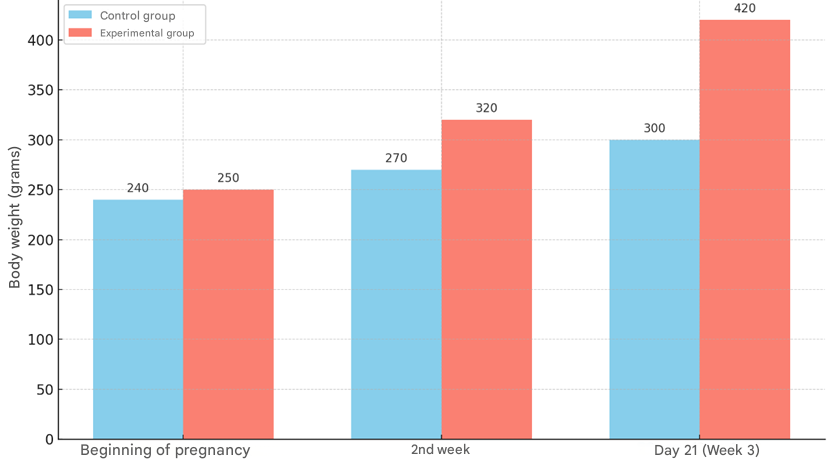 | Figure 1. Dynamics of body weight of pregnant rats in control and experimental groups during gestation |
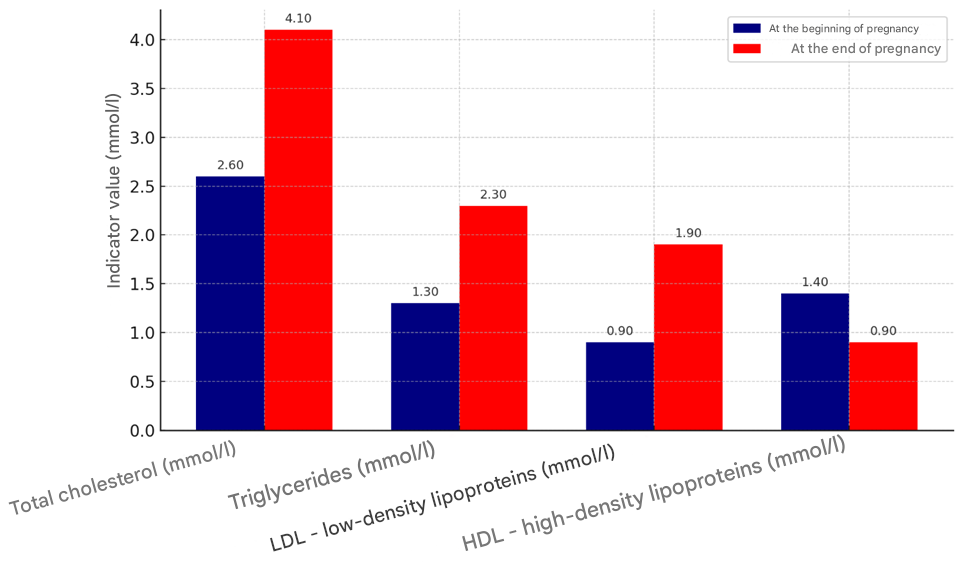 | Figure 2. Changes in lipid metabolism indicators in control and experimental pregnant rats (at 1 and 3 weeks of gestation) |
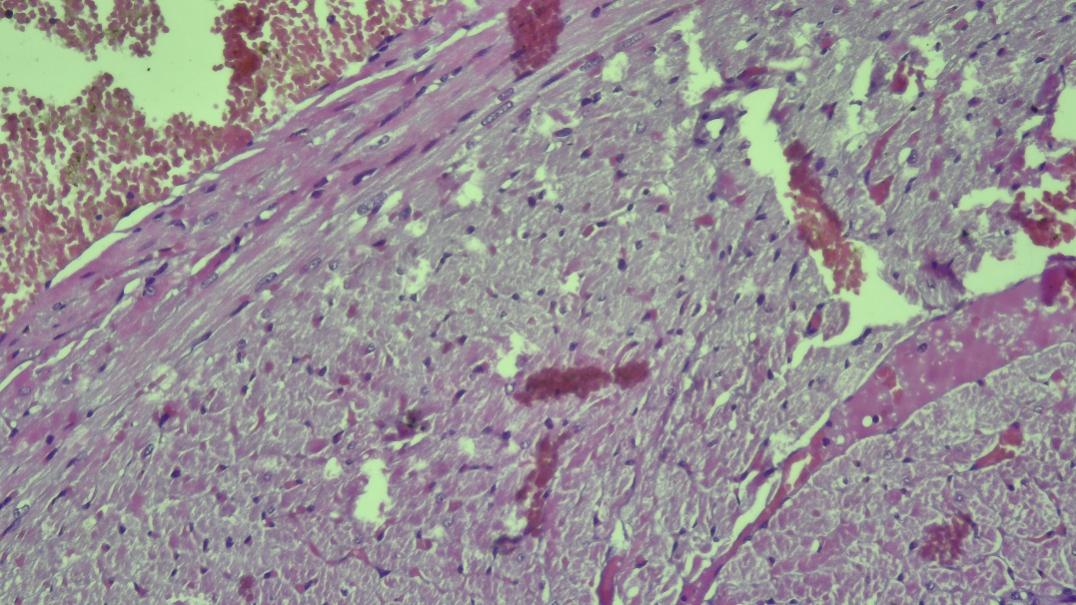 | Figure 3. In the subepicardial layer, cardiomyocytes are located in an unstable perpendicular direction |
3. Results and Discussion
- On the third day of life, the histological structure of the myocardium of rat pups born to mothers with metabolic syndrome did not differ from that of the control group. Cardiomyocytes formed elongated muscle fibers, with oval nuclei located in their central part and clearly expressed myofibrils.During the period of breastfeeding, on the seventh day of the experiment, the results of morphometric analysis showed a decrease in the thickness of the walls of all sections of the heart compared to the control group. In the small blood vessels of the heart, there were hemorrhages of a stasis and diapedesis nature, which were accompanied by perivascular edema, disorganization of the stroma and swelling of connective tissue elements. Also, dilated and full-blooded blood vessels were detected in the subepicardial zone. Initial edema was observed in the myocardial stroma.On the fourteenth day of the experiment, an increase in edema was observed in the myocardial stroma, mainly in the perivenular and pericapillary areas. Collagen fibers swelled, and superficial disorganization of the connective tissue structure began to develop. Blood vessels appeared rounded due to swelling of endothelial cells. In cardiomyocytes, cytoplasmic vacuoles, i.e., accumulation of clear fluid in the cytoplasm, were observed, and signs of hydropic dystrophy were noted. These swelling processes were focal, and dystrophic and unchanged cardiomyocytes were observed together. Lymphohistiocytic infiltration was also noted.On the 21st day of the experiment, changes in blood vessels in the myocardial tissue remained. Perivascular hemorrhages in the form of blood accumulation, stasis, and diapedesis were detected in the veins. The accumulation of fluid in the muscle tissue led to the friction of muscle fibers against each other. Small infiltrates consisting of lymphocytes, histiocytes, and fibroblasts were also detected in the myocardium. Signs of mucoid and fibrinoid staining were noted in the myocardial stroma. Protein hydropic dystrophy developed in cardiomyocytes, swellings and foci of plasmolysis were observed inside the cells. Plasmolysis foci appear under a microscope in a similar way to optical cavities. Cases of vacuolization are detected in the cytoplasm of cardiomyocytes. Thickened and swollen collagen fibers are noted in the endocardium. Initial changes of atherosclerosis were detected in the wall of arterioles. No changes were detected in the endothelial layer of the arterial wall, foam cells were detected in the subendothelial layer.On the 21st day of the experiment, a slight thickening of the basement membrane of the subendothelial layer, edema in the interstitial tissue and foci of fibrous structures, surrounded by a large amount of infiltration of macrophages and lymphocytes, was detected. Atrophic and focal hypertrophic changes were detected in the sparsely located muscle cells of the mediastinum.On the 28th day of the experiment, interstitial edema in the myocardium further increased and spread widely throughout the heart muscle tissue (Fig. 4).
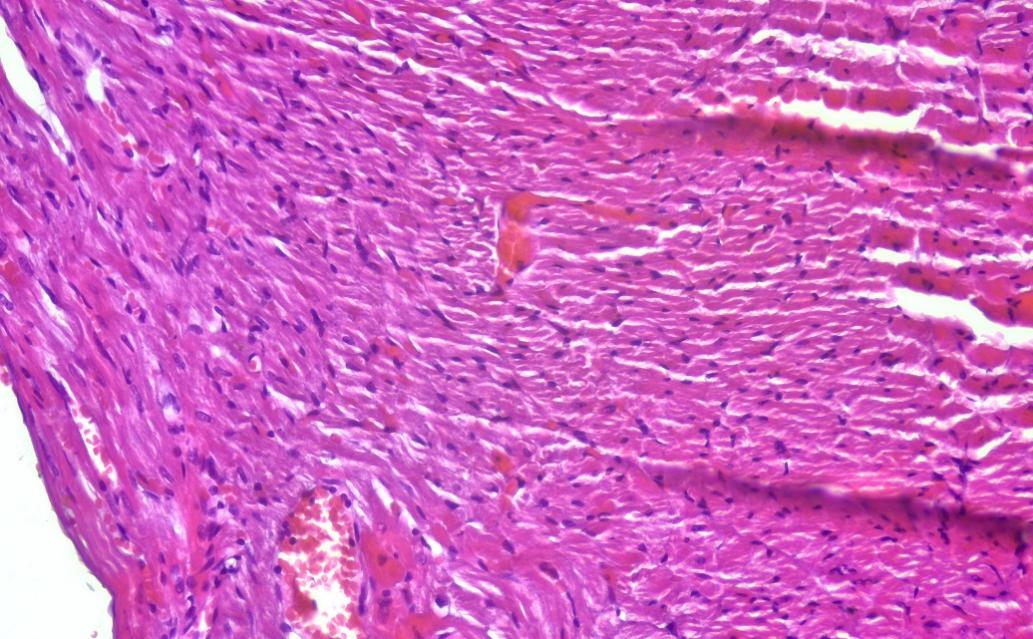 | Figure 4. On the 28th day of the experiment, interstitial edema spread in the subepicardial branch of the myocardium |
4. Conclusions
- In the experimental group, thickening of the arterial blood vessel walls in the heart wall of rat pups and a reduction in vessel diameter were noted. When compared with the control group, the results of morphometric analysis revealed a decrease in the internal diameter of the arterial vessels.Specifically: On day 3, the average arterial diameter was 49.8 ± 4.3 µm, showing no significant difference from the control group. By day 7, this indicator was 52.5 ± 5.6 µm, which is 8.8% lower than the control; on day 14 — 56.7 ± 6.3 µm, 11.2% smaller than the control; on day 21 — 61.3 ± 7.3 µm, 10.6% below normal; and on day 28 — 65.3 ± 2.3 µm, 15.6% less than the control group. It is particularly noteworthy that the arterial diameter normally increases with age. However, according to the experimental results, this growth was significantly delayed in the group exposed to mercazolil. Overall, it was found that during all study periods, the arterial vessel diameter remained consistently lower compared to the control group.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML