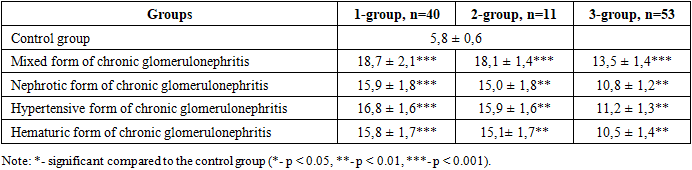-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(12): 4688-4691
doi:10.5923/j.ajmms.20251512.109
Received: Nov. 29, 2025; Accepted: Dec. 22, 2025; Published: Dec. 29, 2025

Results of Laboratory Indicators of Endothelial Changes in Chronic Glomerulonephritis
Kurbanova Zumrad Chutbayevna1, Tojiboyeva Dildora Abdujalilovna2, Sayfutdinova Zukhra Abdurashidovna3
1Professor, Head of the Department of Hematology, Transfusiology and Laboratory Work, Tashkent State Medical University, Uzbekistan
2Researcher, Department of Hematology, Transfusiology and Laboratory Work, Tashkent State Medical University, Uzbekistan
3PhD, Associate Professor, Department of Hematology, Transfusiology and Laboratory Work, Tashkent State Medical University, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Chronic kidney disease is a pathological condition characterized by morphofunctional disorders of the kidneys for three months or more, regardless of the nosological diagnosis, leading to negative consequences for the patient's health. The kidneys are an organ that ensures the excretion of various metabolites, osmotic homeostasis and water-electrolyte balance, and also has sufficient compensatory capabilities even in the event of the death of more than 50% of the total two million nephrons in the kidneys. The specific mechanisms of the development of chronic kidney disease are due to the direct characteristics of the disease, only its course should be determined in the early stages of chronic kidney disease. With a further decrease in the number of intact nephrons, a sequence of complex pathological processes begins: hyperfiltration, hypercoagulation, and impaired renal protein transport, changes in the excretion of damaged cell mediators, which in most cases ends with the formation of nephrofibrosis, even if the cause of the initial damage to the nephrons is eliminated. This situation is characterized by the fact that in all kidney diseases, the pathogenetic mechanisms of kidney tissue damage are diverse and consist of many symptom complexes, ultimately manifested by disruption of the functioning of all organs and systems.
Keywords: Endothelial dysfunction, Endothelin-1, von Willebrand factor, S-Icam
Cite this paper: Kurbanova Zumrad Chutbayevna, Tojiboyeva Dildora Abdujalilovna, Sayfutdinova Zukhra Abdurashidovna, Results of Laboratory Indicators of Endothelial Changes in Chronic Glomerulonephritis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 12, 2025, pp. 4688-4691. doi: 10.5923/j.ajmms.20251512.109.
Article Outline
1. Introduction
- The incidence of acute kidney injury (AKI) has increased over the past few decades, and it has recently been recognized as a significant cause of chronic kidney disease (CKD). CKD, in turn, can progress to end-stage renal failure [5]. Glomerulonephritis ranks first among the most dangerous forms of autoimmune pathology [4]. According to the modern interpretation of the pathogenesis of chronic glomerulonephritis, the interaction of damaging factors with protective mechanisms or anti-inflammatory reactions plays an important role in the development of the disease [7]. In chronic glomerulonephritis, many diseases also initially cause changes in the vascular endothelium. Endothelial cell damage may contribute to the development of various glomerular diseases [2]. Endocapillary proliferation is a process that occurs with the proliferation of local cells within the glomerulus (e.g., mesangial and endothelial cells) and the influx of leukocytes into the glomerulus, resulting in narrowing or complete occlusion of the glomerular capillary lumen [1]. Glomerulonephritis can present with a variety of clinical and laboratory manifestations [3]. CTGF is normally expressed at very low levels in the kidneys, but its expression increases dramatically as fibrosis progresses [6]. In chronic glomerulonephritis, markers of endothelial dysfunction, such as soluble cell adhesion molecule (HEAM), endothelin-1 (End-1) and von Willebrand factor (WF), and S-Icam, were examined. The results showed that endothelial dysfunction was detected in all patients with chronic glomerulonephritis membranous nephropathy. The average HEAM level in group 1 was 17.4 ng/ml, in group 2 this figure was 15.5 ng/ml, and in group 3 it was 12.1 (Figure 1). In the control group, this figure was 5.8 ng/ml, and the changes in HEAM levels among different clinical manifestations of chronic glomerulonephritis are presented in Table 1.
 | Figure 1. Changes in soluble cell adhesion molecule in the main groups, ng/ml |
|
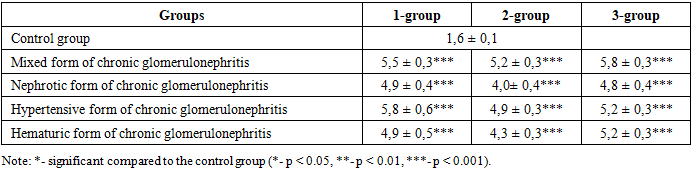
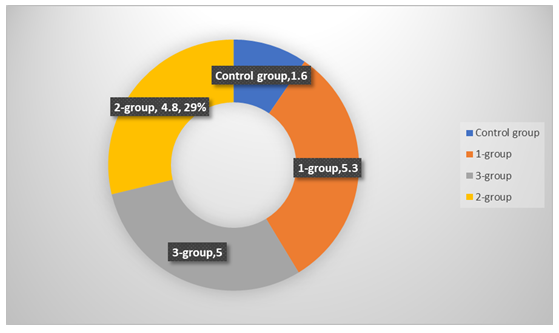 | Figure 2. Endothelin-1 index in the main groups, ng/ml |
|
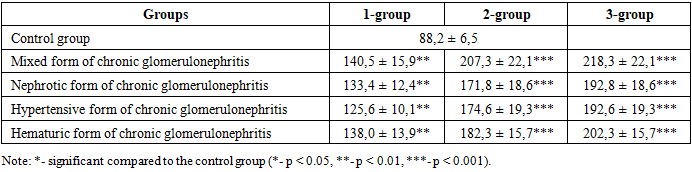
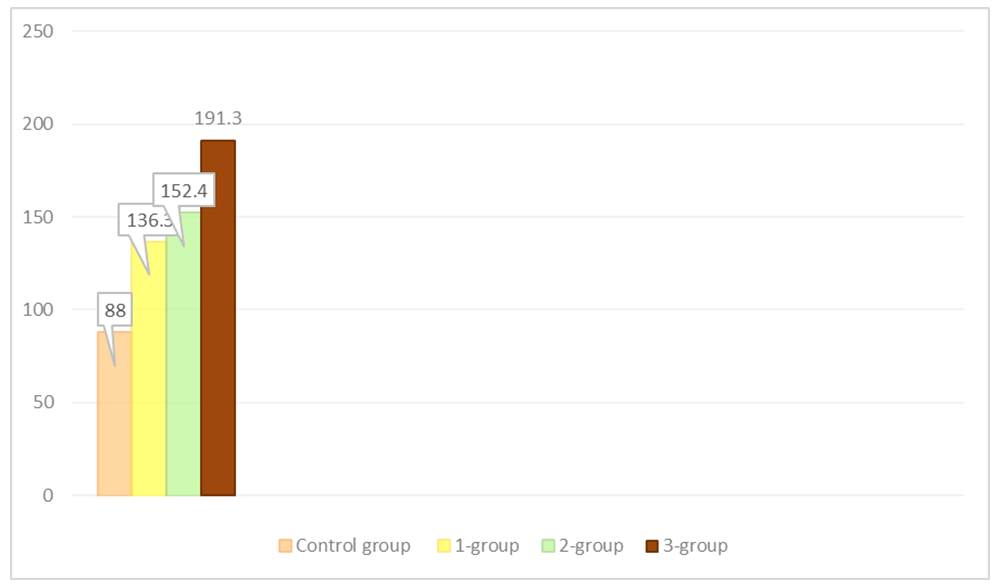 | Figure 3. Willebrand factor in the main groups, ng% |
|
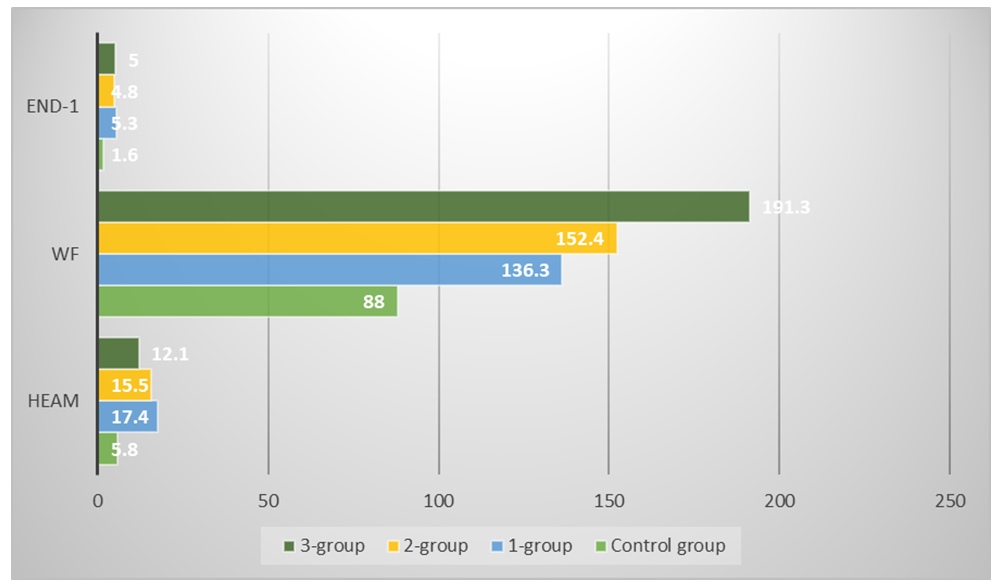 | Figure 4. Endothelial dysfunction scores in the main groups |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML