Turaeva Dilafruz Kholmuradovna1, Garifulina Lilya Maratovna2
1Assistant of Samarkand State Medical University, Samarkand, Uzbekistan
2Professor, Doctor of Science of Samarkand State Medical University, Samarkand, Uzbekistan
Correspondence to: Garifulina Lilya Maratovna, Professor, Doctor of Science of Samarkand State Medical University, Samarkand, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Introduction. The prevalence of non-alcoholic fatty liver disease is increasing in line with the global obesity epidemic and is diagnosed at an younger age, as for, it is pathogenetically associated with lipid metabolism pathology. Material and methods: Therewere examined 88 children with exogenous-constitutional obesity, who were divided into two groups: group 1st with a uniform type of obesity and group 2nd with an abdominal type of obesity, as well as 40 conditionally healthy children with normal body weight. Lipid metabolism was studied, and an ultrasound examination of the liver was performed. Results: It was found that in children with obesity, NAFLD is diagnosed in 50.0% of children, and in children with visceral obesity in 67.5%. Most often, fatty infiltration of hepatocytes is detected in children aged 16-18 years with abdominal obesity (100%). It was found that in obese boys, steatosis develops earlier than in girls and occurs against the background of more expressed lipid metabolism disorders. Structural changes in the liver parenchyma in NAFLD are characterized by the development of a diffuse-focal process with the simultaneous presence of areas of varying sizes with changes such as fatty degeneration and steatohepatitis against the background of unchanged parenchyma. Conclusion. Children with various forms of obesity require timely implementation of a set of diagnostic and therapeutic and preventive measures aimed at identifying disorders and improving the morphofunctional state of the liver in order to reduce the risk of developing diseases of the hepatobiliary system and metabolic-inflammatory progression of hormonal-metabolic disorders.
Keywords:
Obesity, Children, Lipid metabolism, Non-alcoholic fatty liver disease
Cite this paper: Turaeva Dilafruz Kholmuradovna, Garifulina Lilya Maratovna, Features of Lipid Metabolism and Pathology of the Hepatobiliary System in Children with Obesity, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 4089-4094. doi: 10.5923/j.ajmms.20251511.75.
1. Relevance of the Problem
World statistics show that the most common chronic disease of the hepatobiliary system in children and adolescents is non-alcoholic fatty liver disease (NAFLD), which in the vast majority of cases occurs due to the increasing incidence of overweight and obesity [1,2]. Even in publications of the last century, there is data that 25%-55% of children with obesity have non-alcoholic fatty liver disease, in 2/3 of these children the disease progresses, often complicated by fibrosis of the liver parenchyma and even cirrhosis of the liver [3,4]. On the research works of K. Nadeau and M. Rashid, this statement was shown and proven through a study of a large cohort of children. The lack of uniform standard protocols for diagnosing non-alcoholic fatty liver disease creates difficulties for statistically reliable determination of the prevalence of this condition in all countries of the world [5,6]. Currently, according to a systematic review and meta-analysis, the prevalence of NAFLD in the pediatric population is 7.6%, and among obese children – 34.2% [7]. The mechanisms of development and progression of non-alcoholic fatty liver disease continue to be the subject of study by many researchers around the world. The formation of NAFLD is based on metabolic disorders of all types of metabolism against the background of obesity [8]. The trigger mechanism for fat deposition in liver cells with subsequent alteration of hepatocytes is an excess concentration of insulin in the patient’s blood serum, the formation of insulin resistance, and hypertriglyceridemia [9,10]. This is a complex of pathological conditions occurring with lipid metabolism disorders, leading to destruction of the liver parenchyma of non-alcoholic genesis. It is the presence of these changes that allows non-alcoholic fatty liver disease to be classified as one of the conditions of metabolic syndrome. Scientists are deciding on the inclusion of the diagnosis of NAFLD in the classification of metabolic syndrome [11].A kind of beginning of the vicious circle of liver steatosis is insulin resistance, which, in turn, leads to an imbalance between the synthesis of triglycerides and the production of liver lipoproteins, and this acts as a trigger for the deposition of free fatty acids in the liver parenchyma [12,13]. The deposition of free fatty acids leads to “fatty degeneration” of the mitochondria of liver cells, this disrupts β-oxidation of fatty acids and inhibits ATP production, this stimulates oxidative processes in hepatocytes, as a result of which hepatotoxic active substances are synthesized forms of oxygen, which leads to "oxidative stress", as well as the production of lipid peroxidation products, which cause the death of hepatocytes. Literature data on the frequency of NAFLD in patients with lipid metabolism disorders are contradictory, but increasingly authors agree on the relationship between these conditions [14,15]. In light of the presented data, we were interested in studying the state of lipid metabolism in children with obesity, with the determination of the characteristics of the development of non-alcoholic fatty liver disease.
2. Material and Methods
There were observed 84 children with exogenous-constitutional obesity in the Family Clinics in Samarkand (Uzbekistan). The average age of the children was 12.12±0.31 years. The control group consisted of 40 children of the same age (12.15±0.27) with body weight indicators within the normal range.The anthropometric data were obtained using standard methods. Height-weight parameters, waist and hip circumference of children and adolescents were measured. The child's physical development was analyzed by comparing the obtained data with the WHO cumulative centile tables (age, gender, height-weight aspect) [16]. The body mass index (BMI) was also calculated.The obtained anthropometric parameters were assessed in accordance with the standard deviations of the body mass index (SDS) based on WHO recommendations [16]. The basis for establishing the diagnosis of obesity was the determination of the intersection point of age and BMI above +2.0 SDS BMI [17].A sample of 84 children with exogenous-constitutional obesity, who made up the main group, had a BMI of +2.6 to ≥+3 SDS, i.e., children had a BMI characterizing obesity from II-III degree, the average BMI was 32.74±0.47 kg/m2, the average SDS of BMI was in the range of 2.88±0.11, in the control group, the BMI had a range from +1.0 to -1 SDS, while the average BMI was 19.28±0.22 kg/m2 with a standard deviation of SDS BMI of 0.91±0.05 (p0.001 compared to the main group).All children in the main sample had their waist size (WS) and hip volume (HV) determined, with subsequent determination of the WS/HV ratio, which served as an objective indicator of the presence or absence of abdominal obesity. WS was correlated with the WC percentile tables data for gender and age, and the diagnosis of obesity was made according to The abdominal type was defined as WS values equal to the 90th percentile and above for the corresponding age gradation and gender [18]. Provided that the adolescent’s age is 16 years or above, the criterion was WS ≥ 94 cm for boys and ≥ 80 cm for girls. According to the WS indices and the WS/HV ratio, all children with exogenous-constitutional obesity were divided into 2 groups. Group 1 (abdominal obesity- АО) - 40 children had a BMI of +2.0 to ≥+3 SDS, i.e. children had a BMI characterizing obesity from I-III degree, the waist WS in this group exceeded the 90th percentile for the corresponding age and gender. The average BMI in children of this group (32.14±0.51 kg/m2) significantly exceeded the indices of the control group (p<0.001). Group 2 (uniform obesity- UO)– 44 children, included patients with BMI +2.0 to ≥+3 SDS, which was corresponding to I-III obesity groups, but with WS within the normal range according to percentile deviations according to age and gender, on average, BMI was 32.71±0.46 kg/m2. The study of the lipid spectrum of blood serum (total triglycerides (ТG), high density lipoprotein cholesterol (HDLC), low density lipoprotein cholesterol (LDLC) was carried out by the absorption photometry method on the automated test system of the biochemical analyzer Cobas Integra 400 plus (Roche, France).Statistical processing of the obtained data was carried out on a personal computer using the Statistica 10 program. Parametric statistics methods were used for indicators with a normal distributionand and calculated on the arithmetic mean (M), the averge square deviation (σ), the standard error of the mean (m), and relative values (frequency, %). For indicators with a distribution different from normal, nonparametric statistics methods were used with the calculation of the median Me and the interquartile range. The statistical significance of differences in the obtained data was calculated using the Student’s criterion (t) with subsequent calculation of the probability of error (R). Comparison of the frequency feature was carried out using Pearson's chi-square; for values of the expected phenomenon less than 10, Fisher's exact criterion was used.
3. Results of the Study and Discussion
When studying the frequency of dyslipidemia in children with abdominal obesity and in the comparative characteristics of children with a uniform type of obesity, it was revealed that the most common pathology in children with AO was hypertriglyceridemia (47.5%, χ 2 = 9.784, p = 0.02, OR = 2.714, 95% CI 1.727-13.245), which was 2.9 times more common than in the group with UO (15.9%). and the frequency of low HDLC (45.0%, χ2=5.788, p=0.01, OR=3.182, 95%CI 1.216-8.323), which was 2 times higher compared to the group with a uniform type of obesity (20.4%). No difference in the frequency of hypercholesterolemia and HDLC was observed.Table 1. Comparative analysis of the incidence of lipid metabolism pathology in the observation groups (n (%)
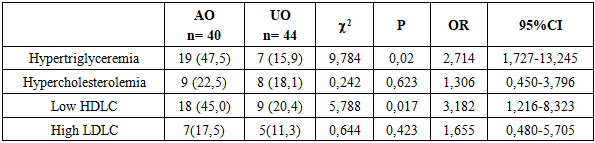 |
| |
|
The average total cholesterol level in children with AO showed a high statistical increase (4.50±0.15 mmol/L) compared to other groups (3.96±0.15 mmol/L, P2<0,02 in the UO group and 2.94±0.06 mmol/L, P<0,001 in the control group) despite the reference values in all three study groups. The highest frequency of hypertriglyceridemia in children with AO was accompanied by the highest average level of TG in children of this group 1.38±0.04 mmol/l, i.e. the level of TG in children with visceral obesity exceeded the recommended level of TG for diagnosing MS in children according to the recommendations (IDF), as well as a high level of TG for adolescents presented in NCEP (USA). At the same time, in children with a uniform type of obesity, it was statistically lower (1.11±0.04 mmol/l, p<0,001).Table 2. Average lipid metabolism indication in the observation groups (M±m)
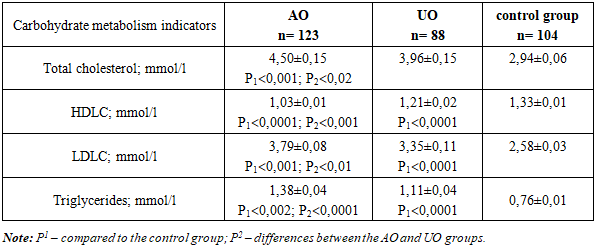 |
| |
|
In children with abdominal obesity, the level of HDLC was significantly lower compared to children with the uniform type and was 1.03±0.01 mmol/l (1.21±0.02 mmol/l in children with UO, p<0,001 and 1.33±0.01 mmol/l, p<0,0001 in children in the control group).In the study of the main carrier of endogenous cholesterol in the blood, LDLC, it was revealed that its average level in children with AO (3.79±0.08 mmol/l) exceeded the indicators of children with RO (3.35±0.11 mmol/l, p<0,001 ), which, according to the recommendations of the NCEP (USA), is an indicator of high cardiovascular risk, and its reduction, along with with a decrease in total cholesterol leads to a reliable reduction in the risk of coronary heart disease, as well as overall mortality and is the main strategy for their prevention and treatment.When conducting a comparative analysis of lipid metabolism parameters in children with abdominal obesity depending on age, it was found that the level of total cholesterol did not have significant differences between age groups; the same picture was observed in relation to LDL cholesterol, where the lipid level was almost at the same borderline level in all age groups of children.Children aged 7-9 years had the lowest TG level (1.13±0.10 mmol/l, p<0,01compared to the control and p<0,05 compared to the group with RO) and the highest HDLC (1.14±0.05 mmol/l, p<0,02 compared to the control and p<0,02 compared to the group with UO, which indicates a possible compensatory mechanism in children of this age category, with a decrease in this mechanism as the child grows older.In children with a uniform type of obesity, no reliable difference was observed between age groups, despite the increase in the pathological level of all lipid metabolism indicators. Only in relation to the level of triglycerides, the increase of which is the main marker of cardiovascular risk, a statistical difference was observed, while in children aged 7-9 years, the TG level was the lowest (0.89±0.07 mmol/l).Table 3. Comparative characteristics of lipid metabolism indicators depending on gender in children of the compared groups
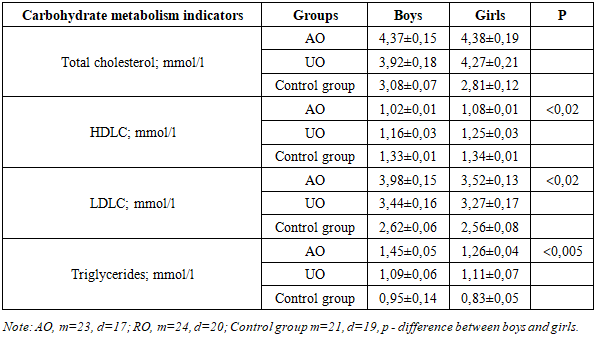 |
| |
|
When comparing the characteristics by gender, it was found that boys with AO had statistically higher pathological indicators for both HDLC (1.02±0.01 mmol/l in boys and 1.08±0.01 mmol/l in girls; p<0,02) and LDL C (3.98±0.15 mmol/l in boys and 3.52±0.13 mmol/l in girls; p<0,02) and by triglyceride level (1.45±0.05 mmol/l in boys and 1.26±0.04 mmol/l in girls; p<0,005).It should be noted that no gender differences in the level of lipid metabolism were found in the group with a uniform type of obesity.When determining the relationship between lipid metabolism pathology depending on the progression of obesity, it was found that in the group with an abdominal type of obesity, a progressive decrease in HDLC was noted with a disease duration of 2-3 years to 7 years or more (with a disease duration of 2-3 years -1.18±0.02 mmol/l, 4-6 years - 1.09±0.02 mmol/l, 7 years and more 1.02±0.01; p<0,001 compared to 2-3 years of experience), while the differences were statistically significant. The same pattern was noted in relation to the increase in the pathological level of TG (with 2-3 years of experience -1.14±0.08 mmol/l, 4-6 years - 1.27±0.11 mmol/l, 7 years and more 1.48±0.06; p<0,01 compared to 2-3 years of experience).The results of the study show that children with 7 years or more of experience had levels of HDLC (<1,0 mmol/l) and TG (>1.3 mmol/l) that are risk factors for the development of comorbid pathology, i.e. a dependence of the degree of lipid metabolism disorder on the duration of the disease was noted.With a uniform type of obesity, a dependence of the level of lipid metabolism indicators on the duration of the disease was also noted, but there were no reliable differences between the subgroups depending on the duration of the disease, and in all groups, including those with a duration of obesity of 7 years or more, lipid metabolism indicators were included in reference value limits (total cholesterol - 4.67±0.39 mmol/l; HDLC - 1.11±0.03 mmol/l; LDLC - 3.87±0.21 mmol/l; TG - 1.16±0.12 mmol/l).The diagnosis of NAFLD was made based on ultrasound examination of the liver, which showed that 27 (67.5%) children with AO and 15 (34.1%) children with uniform obesity had an increase in the transverse size of the liver. Diffuse increase in parenchyma echogenicity was detected in 28 (70.0%) children with abdominal obesity and 19 (43.1%) children with uniform obesity.It should be noted that NAFLD in children with obesity had the form of a diffuse focal process in the form of the simultaneous presence of various sized areas with changes such as fatty degeneration and steatohepatitis against the background of unchanged parenchyma. Thus, based on the ultrasound examination of NAFLD was diagnosed in children of both groups, and in the group with AO it was significantly more frequent compared to the uniform type of obesity (67.5% and 34.1%, respectively, χ2=9.355, p=0,003, OR=4.015, 95% CI 1.618-9.966). Thus, the chance of detecting NAFLD in children with visceral obesity was 4.0 times higher compared to children with uniform obesity. Of the total sample of children with exogenous-constitutional obesity, the frequency of NAFLD was 50.0%. Of interest was the analysis of the age composition of children with NAFLD. Thus, in children with abdominal obesity, the disease was diagnosed in 16 of 19 (66.6%) children aged 10-15 years and in all children aged 16-18 years (11 children (100%). In the group of children with a uniform type of obesity, signs of NAFLD were found in only 8 of 23 children (34.7%) aged 10-15 years, and 7 of 14 (50.0%) children aged 16-18 years.The obtained data have an important prognostic value, since the high frequency of NAFLD diagnosis in children aged 10-15 years may indicate that pathological processes in the liver begin at school age long before the clinical manifestation of metabolic syndrome.The gender distribution of NAFLD showed that there were more boys with this disease than girls, and these indicators concerned both children with the abdominal type and the uniform type of obesity (18 boys out of 27 children (66.6%) with AO and 9 boys out of 15 children (60.0%) with RO), which is consistent with the data from literary sources, indicating a predominance of males among patients with NAFLD [19]. At the same time, the results showed a predominance of this pathology in children with abdominal obesity, which may be evidence of a relationship between the development of NAFLD and abdominal obesity. It is known that the main components of hepatocellular lipids are TG, and the substrates for their synthesis are fatty acids and glycerophosphate. In the case when the production of TG exceeds the synthesis of lipoproteins and their secretion from the hepatocyte in the form of very low density lipoprotein cholesterol, lipids accumulate in the hepatocyte, which leads to the development of fatty hepatosis [20]. HDLC is also synthesized in the liver and, to a lesser extent, in the mucosa of the small intestine and performs two important functions: it is a source of apoproteins for chylomicrons and very low density lipoprotein cholesterol and mediates reverse cholesterol transport [21]. In this regard, it is of interest to analyze the level of serum lipids depending on the state of the liver.We analyzed the frequency of diagnostics of lipid metabolism pathology in children with obesity depending on the pathology in the liver. Thus, an increase in the level of triglycerides in children with NAFLD in the group of children with AO was diagnosed more often (59.2%) than in children without it (23.0%), which had a statistical difference (χ2 = 0.04, p < 0.05). Also, all pathological changes in the field of lipid metabolism were predominant in children with NAFLD against the background of visceral obesity, while confidence limits between frequency differences were not identified.Table 4. Comparative analysis of the incidence of lipid metabolism pathology in children with NAFLD and obesity (n (%)
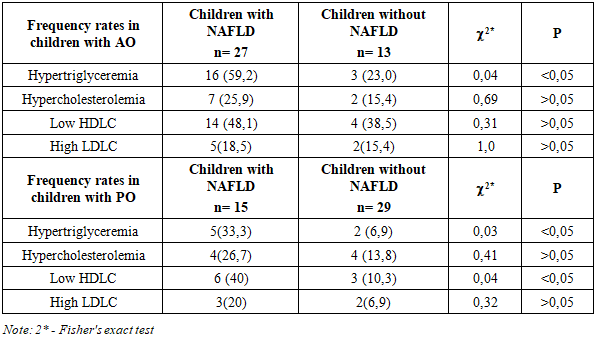 |
| |
|
In children with a uniform type of obesity, a predominance of the frequency of lipid metabolism pathology in children with NAFLD was also noted, while the difference in frequencies with respect to an increase in triglyceride levels (χ2 = 0.03, p< 0.05) and a decrease in HDLC (χ2= 0.04, p < 0.05) was statistically significant.Serum lipid levels were analyzed depending on the liver condition. As can be seen from the presented Table No. 5, statistically significant differences between the average lipid levels of the studied subgroups were found in children in the AO and RO groups.in relation to HDL-C, so in children with NAFLD its lower value was noted compared to children without NAFLD (p<0,001). In relation to other indicators, statistically significant differences were not revealed, with a more pathological level in children with NAFLD. Also, a deeper pathology of lipid metabolism was noted in children with AO and NAFLD compared to children with RO and NAFLD (Table 5).Table 5. Average lipid metabolism parameters in children with NAFLD and obesity (M±m)
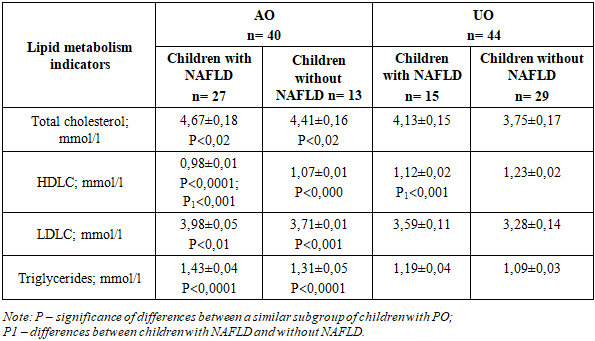 |
| |
|
4. Conclusions
Thus, in children with obesity, lipid metabolism pathology was more pronounced in children with visceral distribution of adipose tissue, with the highest frequency of increased blood triglycerides and decreased HDLC, with a predominance of pathology in boys aged 16-18 years. It should be noted that the presence of pathological levels of lipid metabolism, which are an indicator of high cardiovascular risk in children of this category. Based on the data of the study conducted in children with obesity, it was found that NAFLD is diagnosed in 50.0% of children with exogenous-constitutional obesity, and in 67.5% of children with visceral obesity. Most often, fatty infiltration of hepatocytes is detected in children aged 16-18 years with abdominal obesity (100%), However, according to our data, signs of NAFLD are also diagnosed in 1/3 of children aged 10-15 years with a uniform type of fat distribution. This fact allows us to conclude that the development of the pathological process in the liver in children begins at school age, long before the manifestation of obesity complications, and NAFLD is not only one of the early clinical criteria of complicated obesity in children, but also an important etiopathogenetic factor in the development of the symptom complex of metabolic syndrome in childhood. The data obtained indicate that steatosis develops earlier in obese boys than in girls and occurs against the background of more pronounced lipid metabolism disorders. Structural changes in the liver parenchyma in NAFLD are characterized by the development of a diffuse-focal process with the simultaneous presence of variously sized areas with changes such as fatty degeneration and steatohepatitis against the background of unchanged parenchyma. All of the above indicates that children with obesity, regardless of the form (abdominal type or uniform type), require timely implementation of a set of diagnostic and therapeutic and preventive measures aimed at identifying disorders and improving the morphofunctional state of the liver in order to reduce the risk of developing diseases of the hepatobiliary system and metabolic-inflammatory progression of hormonal-metabolic disorders.
References
| [1] | Golovanova E.V., Lazebnik L.B. Review of international and domestic clinical guidelines for the diagnosis and treatment of non-alcoholic fatty liver disease. Experimental Clinical Gastroenterology- 2016; 135(11): 76–83. |
| [2] | Dana Goldner, Joel E. Lavine, Nonalcoholic Fatty Liver Disease in Children: Unique Considerations and Challenges, Gastroenterology, Volume 158, Issue 7, 2020. |
| [3] | Ivashkin V.T., Drapkina O.M., Maev I.V. et al. Prevalence of non-alcoholic fatty liver disease in outpatients in the Russian Federation: results of the DIREG 2 study // RZHGGK. 2015. No. 6. P. 31–41. |
| [4] | Pavlovskaya E.V., Strokova T.V., Surkov A.G., et al. Obesity in preschool children: metabolic features // Russian Bulletin of Perinatology and Pediatrics. - 2013. - No. 6. – P. 91-96. |
| [5] | Nadeau K. Case report: nonalcoholic steatohepatitis in a teenage girl with type 2 diabetes / K. Nadeau, G. Klingensmith, R. J. Sokol // Curr. Opin. Pediatr. - 2003. -Vol. 15. – P. 127–131. |
| [6] | Rashid M. Nonalcoholic steatohepatitis in children / M. Rashid, E. A. Roberts // J. Pediatr. Gastroenterol. Nutr. - 2000. – Vol. 30. – P. 48–53. |
| [7] | Anderson E. L., Howe L. D., Jones H. E., Higgins J. P. T., Lawlor D. A. (2015). Th e Prevalence of Non-Alcoholic Fatty Liver Disease in Children and Adolescents: A Systematic Review and Meta-Analysis. PLoS ONE10: e0140908. doi: 10.1371/journal.pone.0140908. |
| [8] | Abruzzese G.A., Motta A.B. Nonalcoholic fatty liver disease in children and adolescents. CurrPharmDes. 2015; 21 (35): 5144–5150. |
| [9] | Mishina E.E., Mayorov A. Yu., Bogomolov P.O., et al. Nonalcoholic fatty liver disease: cause or consequence of insulin resistance? // Diabetes mellitus. 2017. Vol. 20. No. 5. P. 335–343. |
| [10] | Dowla S., Aslibekyan S., Goss A. et al. Dyslipidemia is associated with pediatric nonalcoholic fatty liver disease. J Clin Lipidol. 2018; 12 (4): 981–987. DOI: 10.1016/j.jacl.2018.03.089. |
| [11] | Agbim U., Asrani S. Non-invasive assessment of liver fibrosis and prognosis: an update on serum and elastography markers. Expert Rev Gastroenterol Hepatol. 2019; 13(4): 361–374. DOI: 10.1080/17474124.2019.1579641. |
| [12] | Buzzetti E., Pinzani M., Tsochatzis E. The multiple-hit pathogenesis of non-alcoholic fatty liver disease (NAFLD) // Metabolism. 2016. Vol. 65 (8). P. 1038–1048. |
| [13] | Caro-Sabido E.A., Larrosa-Haro A. Efficacy of dietary intervention and physical activity in children and adolescents with nonalcoholic fatty liver disease associated with obesity: A scoping review. Rev Gastroenterol Mex. 2019; 84(2): 185–194. DOI: 10.1016/j.rgmx.2019.02.001. |
| [14] | Kwon Y.D., Ko K.O., Lim J.W. et al. Usefulness of Transient Elastography for Non-Invasive Diagnosis of Liver Fibrosis in Pediatric Non-Alcoholic Steatohepatitis. JKoreanMedSci. 2019; 34(23): e165. DOI: 10.3346/jkms.2019.34.e165. |
| [15] | Medrano M., Arenaza L., Migueles J.H. et al. Associations of physical activity and fitness with hepatic steatosis, liver enzymes, and insulin resistance in children with over-weight/obesity. Pediatr Diabetes. 2020; 21(4): 565–574. DOI: 10.1111/pedi.13011. |
| [16] | WHO. Obesity and overweight. 2022. https://www.who.int/ru/news-room/fact-sheets/detail/obesity-and-overweight. |
| [17] | Peterkova A.V., Bezlepkina O.B., Vasyukova O.V. et al. Obesity in children. Clinical guidelines. Moscow: Ministry of Health of the Russian Federation; 2021. 77 p. |
| [18] | National clinical guidelines. All-Russian Scientific Society of Cardiologists. Moscow 2019. p. 528. |
| [19] | Zakharova I. N. Non-alcoholic fatty liver disease in children: modern possibilities of pathogenetic therapy / I. N. Zakharova, L. A. Zvenigorodskaya, S. V. Yablochkova // Effective pharmacotherapy. - 2022. - No. 2. P. 32-36. |
| [20] | Polunina T. E. Non-alcoholic steatosis of the liver in the practice of an internist / T. E. Polunina, I. V. Maev // Effective pharmacotherapy in gastroenterology. - 2019. - № 1. - P. 14-19. |
| [21] | N.V. Bolotova, K.A. Cherednikova, N.Yu. Filina, L.M. Garifulina, O.А. Logacheva. Modern view on the role of the intestinal microbiota in the development of metabolic disorders. Pediatria n.a. G.N. Speransky. 2023; 102 (4): 140-146. – DOI: 10.24110/0031-403X-2023-102-4-140-146. |



 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML



