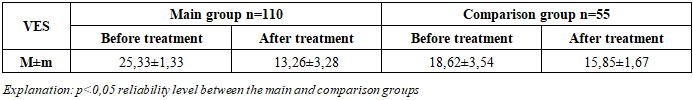-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 4016-4019
doi:10.5923/j.ajmms.20251511.60
Received: Oct. 17, 2025; Accepted: Nov. 11, 2025; Published: Nov. 17, 2025

Characteristics of Combination Therapy Approaches in Vitiligo Patients with Torch Infections
Baxshilloyeva Rushana Ermat qizi, Latipov Ixtiyor Ikromovich
Bukhara State Medical Institute, Bukhara, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Depigmentation is a significant clinical feature of pigmentary disorders, and mostly occurs in vitiligo. Traditional treatments often focus on phototherapy methods; however, alternative therapies, such as regenerating melanocytes or stabilizing the destruction of cells, have recently gained attention due to advances in medical science technologies. The primary objective of this article is to systematically review and synthesize current evidence on the diagnosis and management of ToRCH infections in vitiligo patients, with particular emphasis on clinical features, risk factors, and the effectiveness of antiviral and antibiotic therapies. The review examines specific methods for identifying ToRCH infections relevant to the clinical course of vitiligo and investigates clinical changes in patients with secondary immunodeficiency resulting from pathogen activity. A scientific analysis was conducted on the frequency of lesions, their instability, and resistance to traditional treatments in this group. The article further assesses the value of clinical and laboratory parameters as risk factors in ToRCH infection diagnosis associated with depigmentation.
Keywords: Vitiligo, ToRCH infections, Humoral immunity, Acrofacial, Herpes simplex viruses type I and II, Cytomegalovirus
Cite this paper: Baxshilloyeva Rushana Ermat qizi, Latipov Ixtiyor Ikromovich, Characteristics of Combination Therapy Approaches in Vitiligo Patients with Torch Infections, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 4016-4019. doi: 10.5923/j.ajmms.20251511.60.
1. Introduction
- Despite the medical advances made in the current era of development, the attitude of the population towards vitiligo remains unsatisfactory. The understanding of the disease still repeats the views of several years ago. In most cases, specialists in the field and the general public do not view the diagnosis of vitiligo as a disease, but as a cosmetic defect. Among some ethnic groups, vitiligo is contagious and incurable has a negative impact on the socio-psychological lifestyle of patients. According to WHO, from 0.2 to 2.2% of the world's population (about 40 million people) suffers from vitiligo [1]. Vitiligo accounts for 8% of all skin diseases and 32.2% of all diseases accompanied by skin pigmentation disorders [2,3,6]. The relevance of studying vitiligo is due, on the one hand, to its widespread distribution among patients of any age and gender, its resistance to traditional and non-traditional treatment methods, and the peculiarities of the course of the disease, on the other hand, to the fact that foci of white spots (especially on open areas of the body, on the hands, feet, face and other areas of the body) as an external defect significantly affect the psychoemotional state of the patient. In the clinical course of the disease, more than 5% of the skin lesion area causes mental disorders in 58.7% of patients [4].Recent studies have focused on the analysis of immunohistochemical changes in the pathological focus in vitiligo. The absence of KIT receptor proteins involved in the initial differentiation of melanocytes in the diseased tissue and the variation in the amount of keratinocyte-derived cytokines, basal cell factor (SCF; KIT ligand), tumor necrosis factor (TNF-α), and interleukin (IL)-1 indicate that there are several issues that need to be studied immunohistochemically [11,12]. In addition, the activation of melanocytes in the production of IFN-α and IFN-γ as a result of stimulation of interferons, which are the main part of humoral and tissue immunity, and their apoptosis as a result of their participation in the immune process have prompted there-substantiation of viral and infectious theories [9]. The origin of vitiligo in autoimmune hepatitis with chronic hepatitis C virus (HCV) seropositivity in chronic hepatitis B (HBV) vitiligo was relatively low in the study by Akcan et al. Other types of viruses, such as Epstein-Barr virus, hepatitis E and C viruses, and HIV, in addition to herpes viruses, herpes zoster virus, and CMV, indicate a crucial role of viruses in the pathogenesis of vitiligo by modulating the immune system. Many studies have been conducted on the role of herpes simplex virus, cytomegalovirus, and Epstein-Barr virus in causing such diseases [7,9,10]. The clinical features of vitiligo in association with human immunodeficiency virus (HIV) also confirm that changes directly related to immunological and infectious processes play an important role in the etiopathogenesis of the disease [3,5] Therefore, in the course of scientific research, the relationship between the activity of immune markers and cellular antigens of the ToRCH infections in the clinical course of vitiligo and the use of specific diagnostic methods were studied. Because not enough scientific research has been conducted on the co-occurrence of the disease with infectious agents that cause immunological changes.
2. Materials and Methods
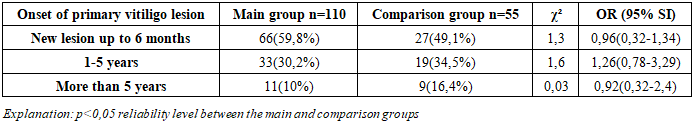
- The review followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. A search of 145 patients in the dermatology department identified 110 patients who underwent PCR screening for Herpes simplex and cytomegalovirus DNA. Of these, 59 patients who tested positive for HSV or CMV were included in randomized controlled trials (RCTs) comparing antiviral treatment plus traditional therapy to traditional therapy alone in vitiligo patients; 35 vitiligo patients who tested negative served as the control group. The 59 positive patients were subdivided into three main groups: the first group included patients with vitiligo who had markers of several ToRCH infections; the second group included patients with vitiligo and markers of herpes pathogens; the third group consisted of patients with vitiligo and markers of cytomegalovirus pathogens. The comparison group consisted of vitiligo patients without detectable ToRCH pathogen markers. Results from all groups were statistically analyzed together with outcomes from a healthy control group. Exclusion criteria included pregnant women, those with thyroid diseases, children under 5 years of age, patients with active infectiuos diseases, and patients over 65 years. All material and data were obtained from patients based on informed consent. Melanin concentration and erythema were measured with noninvasion methods using Mexameter MX 18 equipment. Analysis result was correlated before and after treatment respectively. To identify markers of ToRCH infections as concomitant diseases in the blood analysis of vitiligo patients, immunoglobulin G levels were measured using the immunoenzyme (IFA) method with a Microplate reader I class B type spectrophotometer. Diagnosis was based on patient questionnaire data and clinical signs. DNA fragments of herpes simplex virus and cytomegalovirus in blood plasma from the main and control groups were qualitatively determined using PCR with the Genter 96R (Tianlong) amplifier. Statistical analysis utilized patient outpatient records and medical histories, including questionnaire data and final diagnostic conclusions. Results were processed using MS Excel (2010) and Statistica for Windows 8.0. Group differences were assessed with the nonparametric Mann-Whitney test. The chi-square (χ²) test was applied for percentage comparisons. Differences were considered significant at p<0.05.
3. Results
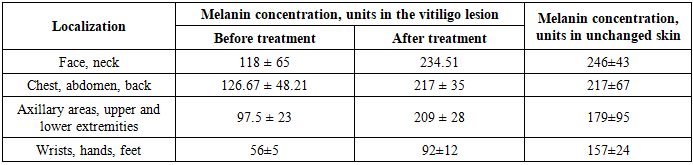
- Melanin concentration and erythema were measured at multiple time points before and after irradiation during combined antiviral and antibacterial treatment. Erythema values in vitiligo lesions increased during therapy to 215.2 ± 56.7 conventional units, representing a 57% increase compared to values in normal skin and depigmented lesions prior to therapy. In newly repigmented lesions, melanin concentration reached 126.67 ± 48.21, closely approximating the melanin content of unaffected skin. As melanin values increased, erythema in vitiligo lesions decreased to 178.7 ± 23.6 conventional units and subsequently stabilized, while melanin values continued to rise. After the course of therapy, total melanin was 234.51 ± 42.08 and erythema was 189.3 ± 34.7 conventional units. (Table 1).
|
|
|
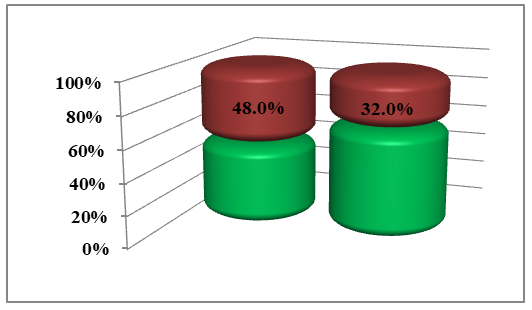 | Picture 1. Detection of HSV and CMV in vitiligo patients (according to PCR test) red –negative; green-positive n=110 |
|
4. Conclusions
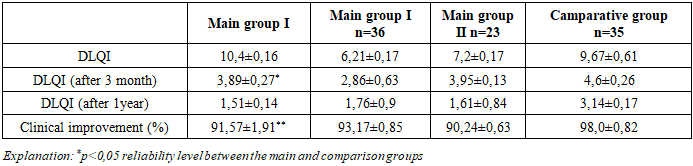
- In younger adults, early diagnosis and treatment of ToRCH infections may contribute to improved repigmentation and stabilization of vitiligo. It was found that the serum levels of ToRCH causative agents in patients with vitiligo were significantly higher, and the disease progressed to acrofacial, vulgar, leukotrichia, and Koebner phenomena compared to patients in the comparison group, and were higher than those in healthy donors in the control group. It was found that the negative effects of herpes simplex (HSV I, II) and cytomegalovirus (CMV) play the main role in the pathogenesis and development of the disease, as well as the clinical course of patients with vitiligo, and the tolerance of clinical forms of unstable vitiligo to traditional treatment. In order to detect the treatment tactics and the severity of the pathological process in patients with vitiligo, it is recommended to use as a risk factor, early detection of clinical signs of ToRCH infections, mainly herpes simplex (HSV I, II) and cytomegalovirus (CMV), and to determine them through laboratory diagnostic methods. In patients with identified pathogens, adequate complex treatment is recommended.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML