-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 4013-4015
doi:10.5923/j.ajmms.20251511.59
Received: Oct. 10, 2025; Accepted: Nov. 8, 2025; Published: Nov. 17, 2025

Influence of Antibiotics as an Exogenous Factor on Morphometric Changes and Lactase Activity in the Postnatal Development of Rats
Hoshimov Azizbek Alijonovich
Department of Normal Physiology, Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: Hoshimov Azizbek Alijonovich, Department of Normal Physiology, Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The study investigates the impact of the broad-spectrum antibiotic ampicillin on lactase activity and morphometric parameters of the small intestinal mucosa in rats during postnatal development. Experimental animals received ampicillin orally at a dose of 50 mg/kg for 10 days from the 5th to the 15th day of life. Histological and morphometric analyses of the jejunum were performed on days 15 and 30 of postnatal development. The results demonstrated a significant reduction in the height of intestinal villi, the depth of crypts, and a marked decrease in lactase enzymatic activity compared with control animals. These findings indicate that antibiotic exposure during early postnatal development disrupts the normal maturation of intestinal epithelium and suppresses lactase expression, which may lead to transient maldigestion and delayed functional adaptation of the intestine.
Keywords: Lactase activity, Ampicillin, Intestinal morphology, Morphometry, Postnatal development, Rats
Cite this paper: Hoshimov Azizbek Alijonovich, Influence of Antibiotics as an Exogenous Factor on Morphometric Changes and Lactase Activity in the Postnatal Development of Rats, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 4013-4015. doi: 10.5923/j.ajmms.20251511.59.
1. Introduction
- Lactase (β-galactosidase, EC 3.2.1.23) is a key brush-border enzyme of enterocytes responsible for hydrolyzing lactose into glucose and galactose. Its activity reaches a peak during the milk-feeding period and gradually declines after weaning in most mammals [1,2]. The level of lactase expression is tightly regulated by both endogenous factors (such as hormonal and genetic mechanisms) and exogenous factors, including the composition of diet, intestinal microbiota, and exposure to antibiotics [3]. Early postnatal life is a critical period for intestinal development, during which external influences can significantly alter enzyme expression and morphofunctional maturation of the mucosa. Antibiotic therapy, while widely used in neonatal and pediatric practice, can profoundly disturb the intestinal microbiome and epithelial differentiation [4]. Broad-spectrum antibiotics, such as ampicillin, suppress not only pathogenic but also commensal bacteria, leading to microbial imbalance (dysbiosis) and impaired nutrient absorption. Previous studies have shown that antibiotic-induced dysbiosis affects villus structure, epithelial renewal, and enzymatic activity, but detailed morphometric evaluation in the context of lactase regulation remains limited. Therefore, the present study was designed to evaluate the influence of ampicillin on the morphometric parameters of the jejunal mucosa and the activity of lactase in rats during postnatal development [5].
2. Materials and Methods
- Experimental design.The study was conducted on 24 white male Wistar rats aged 21–25 days, with an average body weight of 60–80 g. The animals were housed under standard vivarium conditions (temperature 22 ± 2°C, 12 h light/dark cycle, humidity 50–60%) and had free access to water and a standard laboratory diet. All experimental procedures complied with the international ethical standards for animal research (EU Directive 2010/63/EU).Experimental groups.The animals were randomly divided into two groups:1. Control group (n = 12): received distilled water orally.2. Experimental group (n = 12): received ampicillin (Sigma-Aldrich, USA) at a dose of 50 mg/kg body weight once daily for 10 consecutive days via oral gavage.Sampling.Animals from each group were sacrificed on days 5, 10, and 20 after the start of the experiment (n = 4 per time point). The small intestine was dissected, and 2–3 cm segments of the jejunum were fixed in 10% neutral buffered formalin for histological and morphometric studies.Histological examination.Tissue samples were dehydrated in graded ethanol, cleared in xylene, and embedded in paraffin. Sections (5 μm thick) were stained with hematoxylin and eosin (H&E) for general morphology and with periodic acid–Schiff (PAS) reaction to visualize lactase-positive enterocytes.Morphometric analysis.Using a Leica DM500 light microscope equipped with an image analysis system (ImageJ software), the following parameters were measured:• villus height (μm),• crypt depth (μm),• villus-to-crypt ratio,• epithelial cell height (μm),• percentage of lactase-positive enterocytes (%).For each parameter, at least 10 villi and 10 crypts were analyzed per animal.Statistical analysis.Data were expressed as mean ± standard deviation (SD). Statistical significance between groups was assessed using Student’s t-test, with p < 0.05 considered significant.
3. Results
- Macroscopic observations. In the control group, rats demonstrated normal behavior, active feeding, and stable body weight gain. In contrast, ampicillin-treated rats exhibited mild lethargy and decreased appetite during the treatment period, with a slight delay in weight gain that normalized after withdrawal of the antibiotic.Histological findings Histological examination of the jejunum in the control rats revealed normal villus architecture with well-developed enterocytes and intact epithelial lining. The crypts were of moderate depth, and the lamina propria contained sparse lymphoid elements.In contrast, the ampicillin-treated rats displayed pronounced morphological changes: shortening and deformation of villi, desquamation of epithelial cells, vacuolization of enterocytes, and mild inflammatory infiltration in the lamina propria. These alterations were most evident on day 10 of treatment. By day 20, partial regeneration of the intestinal mucosa was observed: villi length increased, epithelial continuity was restored, and the number of lymphoid cells decreased, indicating compensatory recovery processes.Morphometric results
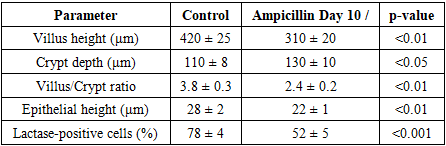 Statistical analysis revealed a significant decrease in villus height, epithelial cell height, and lactase-positive enterocytes, along with a relative increase in crypt depth in the ampicillin group compared to control. These changes reflect temporary suppression of lactase enzyme expression and compensatory hyperplasia of crypt cells in response to epithelial damage.
Statistical analysis revealed a significant decrease in villus height, epithelial cell height, and lactase-positive enterocytes, along with a relative increase in crypt depth in the ampicillin group compared to control. These changes reflect temporary suppression of lactase enzyme expression and compensatory hyperplasia of crypt cells in response to epithelial damage.4. Discussion
- The results of this experimental study demonstrate that exogenous administration of the antibiotic ampicillin during early ontogenesis induces significant structural and functional alterations in the intestinal mucosa of rats. These changes were manifested by reduction of villus height, epithelial cell atrophy, and decrease in the proportion of lactase-positive enterocytes, reflecting impaired enzymatic activity and delayed maturation of the intestinal epithelium. The suppression of lactase activity may be attributed to the disruption of intestinal microbiota balance under the influence of the antibiotic. It is well established that the symbiotic microbiota contributes to the regulation of intestinal enzyme expression through microbial metabolites and trophic factors. Ampicillin, being a broad-spectrum antibiotic, reduces microbial diversity, which leads to secondary dysbiosis, resulting in decreased stimulation of enterocyte differentiation and enzyme synthesis. The observed crypt hyperplasia and partial regeneration by day 20 indicate the activation of compensatory mechanisms in the intestinal epithelium. These mechanisms likely involve the proliferation of stem cells within the crypts and the gradual restoration of microbial balance after cessation of antibiotic exposure.The obtained data are consistent with previous reports (e.g., Dai et al., 2021; Bäckhed et al., 2015) showing that antibiotic-induced dysbiosis during early life alters intestinal morphology, reduces digestive enzyme activity, and affects nutrient absorption. Thus, the morphometric and histological alterations observed in this experiment reflect the sensitivity of the developing intestine to exogenous factors such as antibiotics. This finding emphasizes the importance of rational antibiotic use during early ontogenesis to prevent long-term disturbances in intestinal function and metabolic homeostasis.
5. Conclusions
- The present experimental study provides evidence that exogenous administration of the antibiotic ampicillin during early ontogenesis leads to transient suppression of intestinal lactase activity, accompanied by morphometric and structural alterations in the jejunal mucosa of rats. The observed decrease in villus height, epithelial cell size, and the proportion of lactase-positive enterocytes reflects impaired absorptive and enzymatic function of the intestinal epithelium. Partial restoration of intestinal structure by day 20 suggests the presence of compensatory regenerative mechanisms, likely mediated by epithelial stem cells and microbiota recovery following cessation of antibiotic exposure. These findings highlight the sensitivity of the developing gastrointestinal tract to exogenous chemical agents and underscore the importance of judicious antibiotic use during early postnatal development to prevent long-term metabolic and digestive dysfunctions.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML