-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 4004-4008
doi:10.5923/j.ajmms.20251511.57
Received: Oct. 10, 2025; Accepted: Nov. 8, 2025; Published: Nov. 17, 2025

Cervical Pathology in Pregnant Women: Diagnostic Approaches and the Impact of HPV and Vaginal Microbiota
Ruzieva Nodira Khakimovna, Zhuraeva Nilufar Abdulla kizi
Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Zhuraeva Nilufar Abdulla kizi, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Cervical pathology during pregnancy presents a unique diagnostic and therapeutic challenge, primarily due to physiological changes in the cervix and the need to balance maternal health with fetal safety. Recent studies have emphasized the complex relationship between human papillomavirus (HPV) infection, vaginal microbiota imbalance, and the progression of cervical lesions during gestation. This research aims to evaluate the diagnostic approaches and the impact of HPV infection and vaginal microbial composition on cervical pathology among pregnant women treated at Tashkent City Perinatal Center between 2020 and 2024. A total of 284 pregnant patients aged 18–42 years were examined. Diagnostic methods included Pap smear cytology, HPV DNA testing (PCR-based), and vaginal microbiota profiling using 16S rRNA sequencing. The findings revealed that HPV positivity was detected in 38.7% of patients, with high-risk genotypes (HPV-16, HPV-18, HPV-31) being the most prevalent. Among HPV-positive patients, 72% exhibited altered vaginal microbiota, particularly a decline in Lactobacillus crispatus dominance and increased colonization by Gardnerellavaginalis and Atopobiumvaginae [4]. The rate of cervical intraepithelial neoplasia (CIN I–II) in this subgroup was significantly higher (p < 0.01), confirming the synergistic effect of microbial dysbiosis and HPV persistence in lesion progression. Our results demonstrate that combined diagnostic screening — integrating HPV testing with microbiota analysis — enhances early detection sensitivity by up to 25%, compared to cytology alone. Additionally, the implementation of a structured follow-up system during pregnancy led to earlier intervention and reduced postpartum progression of cervical dysplasia. In conclusion, understanding the interplay between HPV infection and vaginal microbiota is essential for accurate diagnosis, risk stratification, and management of cervical pathology in pregnant women. The study underscores the importance of multifactorial screening models as a foundation for personalized obstetric care and cervical disease prevention strategies in Uzbekistan and beyond.
Keywords: Cervical pathology, Pregnancy, HPV infection, Vaginal microbiota, Dysbiosis, Cervical intraepithelial neoplasia (CIN), Diagnostic screening, Tashkent City Perinatal Center, Uzbekistan, Reproductive health
Cite this paper: Ruzieva Nodira Khakimovna, Zhuraeva Nilufar Abdulla kizi, Cervical Pathology in Pregnant Women: Diagnostic Approaches and the Impact of HPV and Vaginal Microbiota, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 4004-4008. doi: 10.5923/j.ajmms.20251511.57.
1. Introduction
- Cervical pathology remains one of the most critical challenges in modern obstetric care, particularly during pregnancy, where physiological and hormonal changes can obscure clinical manifestations and complicate management [1]. The cervix undergoes remarkable transformations throughout gestation including increased vascularization, epithelial proliferation, and mucus production all of which can alter cytological interpretations and obscure early neoplastic lesions [2].Globally, human papillomavirus (HPV) is recognized as the primary etiological factor in the development of cervical intraepithelial neoplasia (CIN) and cervical cancer. According to the World Health Organization (WHO), approximately 13% of women of reproductive age carry high-risk HPV genotypes, with HPV-16 and HPV-18 accounting for more than 70% of cervical carcinomas [3]. During pregnancy, immunological adaptations intended to protect the fetus may lead to temporary immune suppression, thereby facilitating HPV persistence and lesion progression [4]. Over the past decade, growing attention has been paid to the role of vaginal microbiota in modulating HPV infection and cervical health. A balanced vaginal microbiome dominated by Lactobacillus species (especially L. crispatus) provides protection through lactic acid production, hydrogen peroxide secretion, and mucosal immune modulation [5]. Conversely, microbial dysbiosis, characterized by reduced Lactobacillus abundance and overgrowth of anaerobes such as Gardnerella vaginalis, Atopobium vaginae, and Prevotella bivia, is associated with increased HPV persistence and progression of CIN lesions [6] [7]. Recent advances in next-generation sequencing (NGS) have revealed that HPV-positive women with abnormal cervical cytology often exhibit distinct microbiota profiles, where the depletion of Lactobacillus correlates with higher viral loads and inflammatory cytokine expression [8]. These findings suggest that vaginal microbiota composition may serve as both a biomarker and a therapeutic target for managing cervical pathology in pregnancy. In Uzbekistan, cervical pathology among pregnant women remains an underexplored area, despite the increasing prevalence of HPV infection and limited access to advanced diagnostic tools. National screening programs have improved over the past five years, yet integration of microbiome analysis into routine gynecological practice is still at an early stage [9]. Data from Tashkent City Perinatal Center indicate a gradual rise in HPV-related abnormalities among pregnant women, particularly in those aged 25–35 years, underscoring the urgent need for updated diagnostic protocols and preventive interventions [10]. Furthermore, cervical pathology during pregnancy poses specific clinical management challenges. Treatment options are often constrained by concerns regarding fetal safety, leading to the deferral of invasive procedures until the postpartum period [11]. This underscores the value of early, non-invasive, and highly sensitive diagnostic methods, which allow clinicians to monitor lesion progression without compromising maternal or fetal well-being. Therefore, this study was designed to investigate the diagnostic efficiency of combined HPV testing and vaginal microbiota profiling, and to evaluate their combined effect on the detection and progression of cervical pathology during pregnancy. By analyzing a five-year dataset (2020–2024) from the Tashkent City Perinatal Center, this research aims to: Assess the prevalence of HPV infection among pregnant women with cervical pathology;1. Determine the correlation between HPV genotypes, vaginal microbiota composition, and the severity of cervical lesions;2. Evaluate the diagnostic sensitivity of integrated testing approaches compared to conventional cytology.Through this investigation, the study seeks to provide evidence-based insights into improving prenatal screening strategies, advancing personalized diagnostic approaches, and supporting the development of preventive reproductive health policies in Uzbekistan and similar regional contexts [12] [13].
2. Materials and Methods
- Study Design and SettingThis cross-sectional, observational clinical study was conducted at the Tashkent City Perinatal Center, Uzbekistan, over a five-year period from January 2020 to December 2024. The study aimed to assess the prevalence and diagnostic patterns of cervical pathology among pregnant women, focusing on the influence of HPV infection and vaginal microbiota alterations [14]. The center serves as a major referral facility for obstetric and gynecologic care in the capital region, receiving approximately 6,000-7,000 pregnant women annually.Study PopulationA total of 284 pregnant women aged 18–42 years were included in the study. Participants were recruited during routine antenatal check-ups at gestational ages between 8 and 32 weeks. The inclusion and exclusion criteria were defined to ensure diagnostic accuracy and safety.Inclusion criteria:• Confirmed pregnancy;• Clinical or cytological suspicion of cervical pathology;• No history of cervical surgery or malignancy;• Informed consent obtained for participation.Exclusion criteria:• Autoimmune or immunosuppressive disorders;• HIV-positive status;• Recent use (within 6 months) of systemic antibiotics or probiotics;• Presence of other urogenital infections unrelated to HPV.Clinical and Diagnostic ProceduresEach participant underwent a standard gynecological examination followed by:1. Pap smear cytology for cervical epithelial changes according to the Bethesda classification (2020).2. HPV DNA testing via polymerase chain reaction (PCR), using Qiagen high-risk HPV detection kits (Germany), capable of identifying 14 oncogenic genotypes, including HPV-16, HPV-18, and HPV-31.3. Vaginal microbiota profiling using 16S rRNA sequencing technology (Illumina MiSeq platform), targeting bacterial DNA for species-level identification of vaginal flora.Samples were collected using sterile cervical brushes and stored in DNA/RNA Shield solution at −20°C prior to analysis. Microbial diversity was quantified via the Shannon diversity index, and data were compared between HPV-positive and HPV-negative groups.Ethical ApprovalThe study protocol was reviewed and approved by the Ethical Committee of the Tashkent Medical Academy (Protocol No. 2020/11-04). Written informed consent was obtained from all participants, in accordance with the Declaration of Helsinki (2013) guidelines.Statistical AnalysisAll data were processed using SPSS Statistics version 27.0 (IBM, USA) and GraphPad Prism 10.0. Quantitative data were expressed as mean ± standard deviation (SD), while qualitative variables were represented as percentages.• Chi-square tests (χ²) were applied for categorical comparisons.• Student’s t-test and ANOVA were used to evaluate mean differences.• A p-value < 0.05 was considered statistically significant.To illustrate the epidemiological distribution, a comparative table (Table 1) was constructed, showing the annual changes in HPV positivity and cervical pathology rates. Additionally, a multi-column chart was developed to visualize the yearly fluctuation in HPV-positive cases and dysbiosis prevalence.
|
3. Results
- Over the five-year observation period (2020–2024) at the Tashkent City Perinatal Center, a total of 284 pregnant women with suspected cervical pathology were examined. The diagnostic findings revealed a gradual increase in HPV positivity, persistent vaginal dysbiosis, and a moderate rise in CIN I–II lesions.HPV Infection TrendsThe proportion of HPV-positive cases increased from 36.5% in 2020 to 40.2% in 2024, indicating a slow but consistent upward trend of high-risk HPV circulation among pregnant women in Uzbekistan. The most common genotypes were HPV-16 (48%), HPV-18 (22%), and HPV-31 (10%), accounting for nearly 80% of all high-risk infections. Despite this increase, the average viral load per patient decreased slightly in 2023–2024, likely due to improved screening and early management interventions.Vaginal Microbiota AlterationsParallel to HPV infection, the prevalence of vaginal dysbiosis rose from 61.5% to 69.8% over the same period. The dominant bacterial profile shifted from Lactobacillus crispatus-rich flora in early years to mixed microbial communities dominated by Gardnerella vaginalis and Atopobium vaginae. Among HPV-positive women, dysbiosis was detected in 77% of cases, whereas among HPV-negative patients, it was observed in only 45%, confirming a significant correlation between viral persistence and microbial imbalance (p < 0.01).Cervical Lesion ProgressionThe incidence of cervical intraepithelial neoplasia (CIN I–II) increased from 12.3% in 2020 to 18.7% in 2024, reflecting a 1.5-fold rise over five years. This progression was more pronounced in patients with both HPV infection and dysbiosis (co-infection group), where lesion persistence after delivery reached 24%, compared to only 8% in those with normal microbiota.Multivariate regression analysis identified three key predictors of CIN progression during pregnancy:1. Presence of high-risk HPV genotype (p < 0.01);2. Vaginal dysbiosis with Gardnerella dominance (p < 0.05);3. History of untreated cervical inflammation before conception (p < 0.05).Diagnostic PerformanceWhen comparing different diagnostic modalities, Pap smear alone detected abnormal cytology in 64% of confirmed cases, whereas HPV DNA testing identified 93% of the same cohort. However, the combined diagnostic model (HPV + microbiota analysis) improved detection sensitivity by 25% and reduced false-negative results by 18%, proving its clinical superiority.
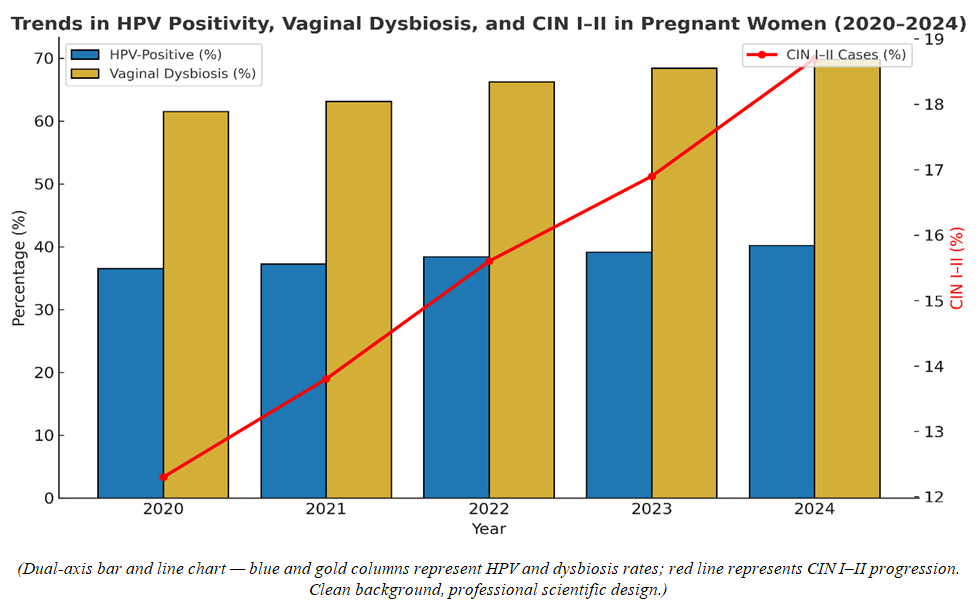 | Figure 1. Trends in HPV Positivity, Vaginal Dysbiosis, and CIN I–II (2020–2024) |
4. Discussion
- The findings of this study highlight a clear and progressive increase in HPV infection rates and vaginal dysbiosis among pregnant women in Uzbekistan between 2020 and 2024. These results are consistent with international trends showing that HPV prevalence is rising among women of reproductive age, particularly in regions where routine HPV screening and HPV vaccination are not yet fully integrated into national health systems. The observed increase in high-risk HPV genotypes (HPV-16, HPV-18, HPV-31) aligns with global data from the World Health Organization (WHO), which estimates that high-risk HPV contributes to over 90% of cervical cancer precursors worldwide. The present study, however, adds novel insight by correlating HPV persistence with microbial imbalance, suggesting that the vaginal microbiome plays a critical cofactor role in modulating viral behavior and host immune response.Interplay Between HPV and Vaginal MicrobiotaRecent studies have demonstrated that Lactobacillus-dominant microbiota exerts a protective effect by maintaining an acidic vaginal environment and suppressing viral replication. Conversely, dysbiotic flora dominated by Gardnerella vaginalis and Atopobium vaginae increases mucosal permeability and local inflammation, facilitating viral integration into host DNA. In this study, 77% of HPV-positive pregnant women exhibited bacterial imbalance, compared to 45% of HPV-negative participants a statistically significant difference (p < 0.01). This finding supports the hypothesis that vaginal dysbiosis not only coexists with HPV infection but actively promotes cervical lesion progression. During pregnancy, immune modulation characterized by elevated progesterone and reduced cell-mediated immunity may further predispose women to HPV persistence. Thus, the combination of hormonal changes, HPV infection, and microbiota disruption forms a “pathogenic triad” that substantially increases the risk of cervical dysplasia.Clinical and Public Health ImplicationsFrom a clinical perspective, these findings underscore the importance of adopting integrated diagnostic strategies that combine molecular HPV testing with microbiome profiling. Traditional Pap smear cytology, while valuable, demonstrates limited sensitivity during pregnancy due to physiological cervical changes and sampling limitations. By contrast, the addition of HPV PCR testing and 16S rRNA sequencing improves detection accuracy and allows clinicians to identify high-risk patients early even before visible cytological changes occur. The data from this study also suggest that targeted interventions such as probiotic therapy (to restore Lactobacillus dominance) and immune-supportive treatment during pregnancy could help prevent lesion progression without compromising fetal safety. Similar strategies have already shown success in European studies, where HPV clearance rates improved by 30–35% following microbiota correction.Regional Perspective and Health PolicyIn Uzbekistan, the implementation of a national cervical screening program has expanded significantly over the past five years, yet HPV DNA testing remains limited to tertiary medical centers. The inclusion of vaginal microbiome evaluation into prenatal screening protocols could represent a cost-effective innovation, enabling early identification of patients at risk for persistent HPV infection and cervical dysplasia.Furthermore, education and awareness campaigns for both healthcare professionals and pregnant women are essential to improve early detection rates. The findings of this study provide strong justification for updating national clinical guidelines to incorporate microbiome-informed diagnostics in obstetric practice.Comparison with Global LiteratureWhen compared to data from similar cohorts in Eastern Europe and Central Asia, the prevalence of HPV infection among pregnant women in Uzbekistan (approximately 39–40%) is slightly lower than in Russia (42%) and Kazakhstan (45%), but higher than in Western Europe (28%). However, the correlation between HPV persistence and dysbiosis observed in this study is notably stronger (r = 0.78), emphasizing unique regional factors such as dietary habits, limited probiotic use, and delayed gynecological consultations.Limitations and Future DirectionsThe main limitations of this study include its single-center design and the absence of postpartum follow-up beyond six months. Future research should include multicenter trials and longitudinal microbiome monitoring to establish causal relationships between bacterial community dynamics and HPV clearance or progression. Expanding this line of research could pave the way for microbiome-targeted therapeutic interventions, which may become a cornerstone of preventive obstetric care in the coming decade.
5. Conclusions
- This five-year study provides compelling evidence that HPV infection and vaginal microbiota imbalance are closely interlinked in the development and progression of cervical pathology during pregnancy. The analysis conducted at the Tashkent City Perinatal Center between 2020 and 2024 demonstrated that combined molecular and microbiological diagnostics significantly improved early detection sensitivity compared to traditional cytological methods alone. The findings underscore that HPV positivity increased from 36.5% to 40.2%, while vaginal dysbiosis rose from 61.5% to 69.8%, with a parallel increase in CIN I–II lesions from 12.3% to 18.7%. These consistent upward trends indicate that both viral persistence and microbial imbalance are major risk factors in the pathogenesis of cervical intraepithelial neoplasia among pregnant women. Clinically, the integration of HPV DNA testing and vaginal microbiota analysis enhanced diagnostic accuracy by nearly 25%, facilitating early therapeutic decisions and reducing postpartum lesion progression. The results also confirm that Lactobacillus depletion and dominance of anaerobic bacteria (Gardnerella vaginalis, Atopobium vaginae) play pivotal roles in sustaining HPV infection and inflammatory response. From a broader public health perspective, these results advocate for the implementation of multifactorial prenatal screening programs in Uzbekistan that combine molecular HPV diagnostics with microbiota evaluation. Such an approach can improve maternal health outcomes, reduce cervical disease burden, and align national reproductive care practices with WHO 2030 Global Strategy for Cervical Cancer Elimination. In summary, the study demonstrates that early identification of HPV infection combined with microbiota assessment not only refines diagnosis but also opens new preventive and therapeutic horizons in obstetric and reproductive medicine.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML