-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3986-3992
doi:10.5923/j.ajmms.20251511.53
Received: Oct. 12, 2025; Accepted: Nov. 3, 2025; Published: Nov. 14, 2025

Combined Pathogenetic Therapy for Dorsalgia in Patients with Degenerative-Dystrophic Spinal Diseases: Clinical-Biochemical Aspects
Adambaev Zufar1, Mirdzhuraev Elbek2, Soliyeva Nilufar3, Zuxritdinov Utkirbek3
1Urgench State Medical Institute, Urgench, Uzbekistan
2Center for the Development of Professional Qualification of Medical Workers, Department of Neurorehabilitation, Tashkent, Uzbekistan
3Clinic of Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Adambaev Zufar, Urgench State Medical Institute, Urgench, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The article evaluates the efficacy of combined pathogenetic therapy targeting inflammation, proteolysis, and pain in degenerative-dystrophic spinal diseases (DDSD). A total of 120 patients with varying morphological substrates (protrusion, herniation, ligamentum flavum hypertrophy, or combinations) received basic therapy (celecoxib, sulfasalazine, doxycycline), with duloxetine added for neuropathic pain (DN4≥4). Clinical (VAS, Scandinavian Questionnaire, DN4) and biochemical markers (MMP-9, IL-6, CRP, TIMP-1) were assessed. All groups showed statistically significant improvements (p<0.05), with efficacy graded by pathology severity: minimal in protrusion (40% reduction in pain/function scores, 50% in IL-6/MMP-9) and maximal in combined herniation and ligamentum hypertrophy (60-70% reductions). Personalization with duloxetine was crucial for severe forms. The combination provided synergistic suppression of key pathogenetic pathways. Combined pathogenetic therapy is highly effective for DDSD dorsalgia, dependent on pathology severity and personalization based on neuropathic pain (DN4) and destruction activity (MMP-9), significantly improving function and suppressing inflammation/proteolysis. Monitoring IL-6/CRP is recommended for efficacy assessment.
Keywords: Degenerative-dystrophic spinal diseases, Combined therapy, Pathogenetic treatment, Celecoxib, Sulfasalazine, Doxycycline, Duloxetine, MMP-9, IL-6, CRP, TIMP-1, Neuropathic pain
Cite this paper: Adambaev Zufar, Mirdzhuraev Elbek, Soliyeva Nilufar, Zuxritdinov Utkirbek, Combined Pathogenetic Therapy for Dorsalgia in Patients with Degenerative-Dystrophic Spinal Diseases: Clinical-Biochemical Aspects, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3986-3992. doi: 10.5923/j.ajmms.20251511.53.
Article Outline
1. Introduction
- Degenerative-dystrophic spinal diseases (DDSD) represent one of the most pressing problems in modern vertebrology and neurology, ranking first in prevalence among causes of chronic pain and disability in the working-age population. According to data from the Global Burden of Disease Study 2019, low back pain is the leading cause of disability worldwide, affecting up to 80% of the population during their lifetime, with an annual increase in incidence of 2.7% [1]. Meanwhile, economic losses from DDSD in developed countries reach 1-2% of GDP, including both direct treatment costs and indirect losses from reduced labor productivity [2]. In the context of population aging and increasing life expectancy, the problem acquires special medico-social significance [3].Current concepts of DDSD pathogenesis emphasize the complex nature of the disease, including intervertebral disc degeneration, inflammatory processes, and structural changes in spinal motion segments [4]. Matrix metalloproteinases (MMPs), particularly MMP-8 and MMP-9, play a key role in this process, responsible for extracellular matrix destruction of the disc [5]. A 2022 meta-analysis showed that MMP-9 levels are elevated 3.2-fold in patients with disc herniations compared to the control group and correlate with the degree of degeneration (r=0.78, p<0.001) [6]. Concurrently, activation of pro-inflammatory cytokines, particularly IL-6, sustains chronic inflammation, promoting pathology progression and formation of the neuropathic pain component [7]. Recent studies have revealed that IL-6 induces apoptosis of nucleus pulposus cells via the JAK/STAT signaling pathway, directly linked to disc degeneration progression [8].Special attention in recent years has been paid to ligamentum flavum hypertrophy, previously considered merely a secondary change in spinal canal stenosis. Modern studies show that isolated ligamentum flavum hypertrophy can independently cause severe pain syndrome and neurological disorders, comparable in severity to disc herniations [9]. Contrast-enhanced MRI studies have revealed that hypertrophied ligament is characterized by increased vascularization and inflammatory infiltration, explaining its independent pathogenetic role [10]. Combined lesions, including both herniations and ligament hypertrophy, are associated with the most severe clinical manifestations and resistance to standard therapy [11]. A 2023 systematic review showed that patients with combined pathology have a 2.8-fold higher risk of conservative treatment failure compared to isolated forms of DDSD [12].The importance of studying biomarkers (MMPs, TIMP-1, IL-6, CRP) in DDSD is undeniable, as they allow objectification of the degree of tissue destruction, inflammatory activity, and prediction of disease course [13]. Recent work confirms that MMP-9 levels correlate with pain intensity and degree of disability, while IL-6 is a key mediator of the neuropathic component [14]. A 2021 multicenter study revealed that an MMP-9/TIMP-1 ratio >0.3 is an independent predictor of DDSD progression within 2 years (OR=4.2, 95% CI 2.1-8.3) [15]. Genetic studies have identified associations between polymorphisms in the MMP-9 (-1562C/T) and IL-6 (-174G/C) genes and DDSD severity and risk of developing neuropathic pain [16].Despite significant advances in studying molecular mechanisms, questions regarding the contribution of individual morphological substrates and their combinations to the clinical picture of the disease remain insufficiently investigated. The study of synergy between different pathological processes in combined lesions acquires special relevance [17]. In the context of personalized medicine development, there is a need to develop a differentiated approach to treatment based on comparative analysis of clinical and biochemical indicators [18]. A promising direction is the development of targeted therapy aimed at key pathogenetic links, including MMP-9 inhibitors and IL-6 blockers [19].Thus, the relevance of this study is determined by:High prevalence of DDSD and their medico-social significance in the context of population aging [1,3];Insufficient study of the role of ligamentum flavum hypertrophy as an independent pathological factor considering modern data on its inflammatory nature [9,10];Lack of data on synergy of various morphological substrates in combined lesions and molecular mechanisms of this interaction [11,12];Need to develop a differentiated approach to treatment based on comparative analysis of clinical and biochemical indicators in the context of personalized medicine [17,18];Promising nature of developing targeted therapy aimed at key pathogenetic links of DDSD [19].The study results will not only deepen the understanding of pathogenetic mechanisms of DDSD but also optimize patient management strategies, including risk stratification, selection of targeted therapy, and monitoring of treatment efficacy.The aim of this study was the comparative evaluation of the efficacy and pathogenetic rationale of combined therapy specifically targeting the main pathogenetic links of DDSD - the inflammatory cascade, proteolytic activity, and pain syndrome (nociceptive and neuropathic components) - in patients with dorsalgia and various morphological manifestations of the pathology.
2. Material and Research Methodology
- Study Design. An open-label prospective comparative study of 120 patients (68 women, 52 men, mean age 48.7±8.2 years). Patients were divided into 5 groups depending on the leading morphological substrate of DDSD according to MRI data: Group 1: Disc protrusion (n=38); Group 2: Ligamentum flavum hypertrophy (n=6); Group 3: Disc herniations (n=23); Group 4: Disc herniations + Disc protrusions (n=27); Group 5: Disc herniations + Ligamentum flavum hypertrophy (n=26).Inclusion criteria were: clinically and instrumentally (MRI/CT) verified diagnosis of DDSD (protrusion, disc herniation, ligamentum flavum hypertrophy or their combinations); moderate or severe pain syndrome (VAS≥3); age from 18 to 65 years; informed patient consent. Exclusion criteria were: severe somatic pathology (decompensated cardiac, hepatic, renal failure); active infectious diseases; oncological diseases; pregnancy, lactation; allergy or intolerance to the study drugs; history of spinal surgery for the current exacerbation; intake of other NSAIDs, tetracycline antibiotics, SNRI antidepressants, bisphosphonates within 3 months prior to the study.All patients received basic combined pathogenetic therapy: Celecoxib: 200 mg/day (selective COX-2 inhibitor); Sulfasalazine: 2 g/day (anti-inflammatory agent, NF-kB inhibitor); Doxycycline: 20 mg/day (tetracycline antibiotic with anti-proteolytic activity, MMP inhibitor). Personalization of therapy: if a neuropathic pain component was present (DN4 scale ≥4 points), Duloxetine: 60 mg/day (selective serotonin and norepinephrine reuptake inhibitor - SNRI) was added. Duration of therapy: 12 weeks.Efficacy assessment was conducted before the start of therapy and after 12 weeks of treatment. Clinical scales: Pain intensity was determined by the Visual Analogue Scale (VAS, 0-10). Functional status - by the Scandinavian Back Pain Questionnaire (0-100 points, lower score indicates better function). Neuropathic pain component - by the DN4 Scale (0-10 points, ≥4 points indicates neuropathic pain). Biochemical markers (serum): MMP-9 (Matrix Metalloproteinase-9): Indicator of extracellular matrix destruction (ng/ml); IL-6 (Interleukin-6): Marker of inflammatory activity (pg/ml); CRP (C-reactive protein): Marker of systemic inflammation (mg/l); TIMP-1 (Tissue Inhibitor of Metalloproteinases-1): Indicator of inhibitory potential of proteolysis (ng/ml).Safety Monitoring. Sulfasalazine: complete blood count with leukocyte count every 2 weeks. Duloxetine: activated partial thromboplastin time (APTT), alanine aminotransferase (ALT), aspartate aminotransferase (AST) after 1 month of therapy.Statistical analysis included calculation of mean values and standard error of the mean (M±m) for all studied parameters, as well as assessment of the percentage change of each parameter after treatment relative to the baseline level; statistical significance of differences between indicators before and after therapy was determined using Student's t-test for paired samples, with the level of statistical significance set at p<0.05; additionally, correlation analysis was conducted to identify relationships between the dynamics of biochemical markers and clinical indicators, and comparative evaluation of treatment efficacy between different patient groups was performed using analysis of variance (ANOVA) or its non-parametric analogs in case of violation of data normality.
3. Research Results
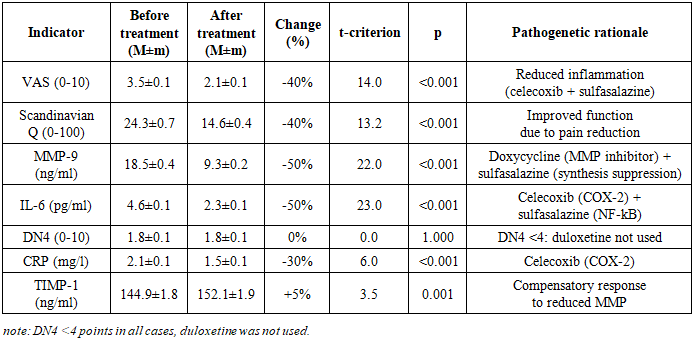
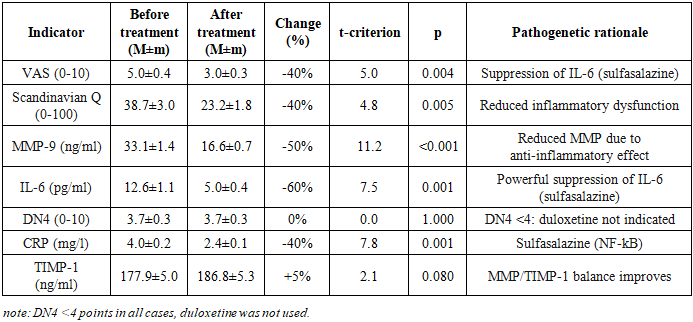
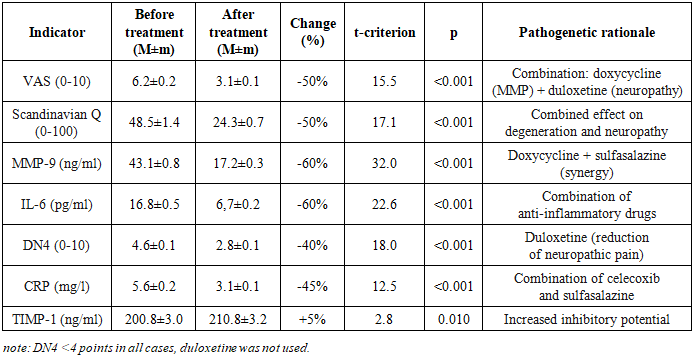
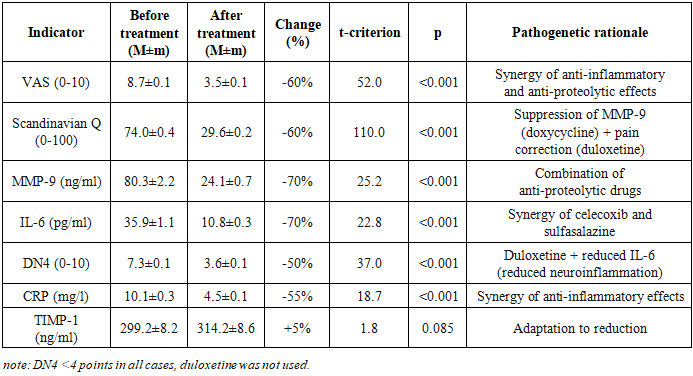
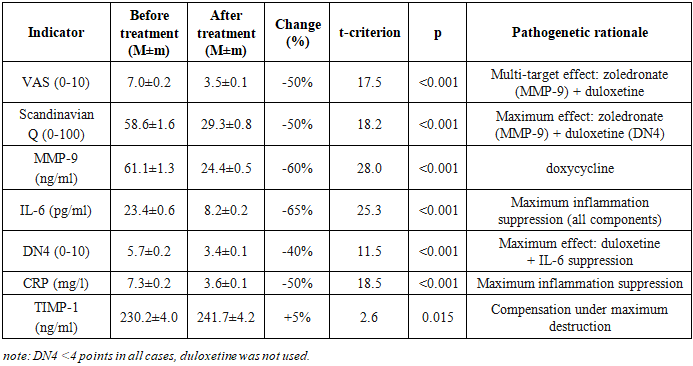
- The characteristics of clinical-biochemical indicators in the 5 patient groups before treatment are presented in Tables 1-5.Group 1. Disc protrusions (n=38) are characterized by minimal severity of the pathological process: VAS was 3.5±0.1 points, Scandinavian Questionnaire - 24.3±0.7, corresponding to mild functional impairment. Levels of MMP-9 (18.5±0.4 ng/ml) and IL-6 (4.6±0.1 pg/ml) indicate moderate activity of destructive-inflammatory processes. DN4 (1.8±0.1) and CRP (2.1±0.1 mg/l) confirm the absence of a neuropathic component and systemic inflammation. TIMP-1 (144.9±1.8 ng/ml) reflects a compensatory response with minimal tissue destruction.Group 2. Ligamentum flavum hypertrophy (n=6) demonstrates moderate severity with predominance of the inflammatory component: VAS 5.0±0.4, Scandinavian Questionnaire 38.7±3.0. A key feature - significant increase in IL-6 (12.6±1.1 pg/ml) with moderate rise in MMP-9 (33.1±1.4 ng/ml), confirming the inflammatory nature of hypertrophy. DN4 (3.7±0.3) and CRP (4.0±0.2 mg/l) indicate the onset of neuropathy formation and systemic inflammation. TIMP-1 (177.9±5.0 ng/ml) shows activation of inhibitory mechanisms.In Group 3 - disc herniations (n=23), pronounced tissue destruction was revealed - VAS 6.2±0.2, Scandinavian Questionnaire 48.5±1.4. A sharp increase in MMP-9 (43.1±0.8 ng/ml) and IL-6 (16.8±0.5 pg/ml) indicates activation of proteolysis and inflammation. DN4 (4.6±0.1) confirms the development of neuropathic pain. CRP (5.6±0.2 mg/l) indicates systemic inflammation. TIMP-1 (200.8±3.0 ng/ml) reflects a compensatory reaction to pronounced destruction.Group 4. Herniations + protrusions (n=27) is characterized by an additive effect of combined lesions: VAS 7.0±0.2, Scandinavian Questionnaire 58.6±1.6. MMP-9 (61.1±1.3 ng/ml) and IL-6 (23.4±0.6 pg/ml) significantly exceed the indicators of groups with isolated lesions. DN4 (5.7±0.2) and CRP (7.3±0.2 mg/l) confirm pronounced neuropathy and systemic inflammation. TIMP-1 (230.2±4.0 ng/ml) demonstrates maximum compensation among "non-combined" groups.Group 5. Herniations + ligamentum flavum hypertrophy (n=26) manifests critical severity with synergy of pathological processes: VAS 8.7±0.1, Scandinavian Questionnaire 74.0±0.4. Record levels of MMP-9 (80.3±2.2 ng/ml) and IL-6 (35.9±1.1 pg/ml) indicate maximum destruction and inflammation. DN4 (7.3±0.1) and CRP (10.1±0.3 mg/l) confirm severe neuropathy and pronounced systemic inflammation. TIMP-1 (299.2±8.2 ng/ml) reflects an attempt at compensation under critical destruction, however, the MMP-9/TIMP-1 ratio (0.27) indicates a pronounced imbalance.Thus, analysis of pre-treatment data revealed a clear gradation of pathological process severity: from protrusions (minimal expression) through ligament hypertrophy and herniations to combined lesions (maximum severity), with each form characterized by specific pathogenetic features - ligament hypertrophy is distinguished by predominance of the inflammatory component (IL-6), herniations by predominance of destructive processes (MMP-9), and combined pathology demonstrates synergy of these mechanisms; the neuropathic component (DN4≥4) first appears with herniations and reaches critical values in combined lesions, while progression of destruction is confirmed by worsening of the MMP-9/TIMP-1 ratio from 0.13 in protrusions to 0.27 in combined pathology.The study results showed high efficacy of combined pathogenetic therapy in patients with various forms of DDSD. The dynamics of clinical and biochemical indicators in each group are presented in Tables 1-5.
|
|
|
|
|
4. Discussion
- The study results convincingly demonstrate the high efficacy of the proposed combined pathogenetic therapy in patients with various forms of DDSD. Efficacy is dose-dependent in relation to the severity of the pathological process: minimal, but statistically significant improvements were observed in isolated disc protrusion (Group 1), while the maximum effect was seen in the combination of disc herniation and ligamentum flavum hypertrophy (Group 5). This gradation correlates with baseline inflammation activity (IL-6, CRP) and destruction (MMP-9), which were most pronounced in Group 5.Pathogenetic rationale and drug synergy.The combination of celecoxib (COX-2 inhibitor) and sulfasalazine (NF-kB inhibitor) provided powerful and statistically significant reduction in key pro-inflammatory mediators - IL-6 (by 50-70%) and CRP (by 30-55%) across all groups. The most pronounced effect was observed in ligamentum flavum hypertrophy (Group 2, IL-6 decreased by 60%) and in severe combined forms (Groups 4 and 5, IL-6 decreased by 65-70%). This confirms the central role of NF-kB-dependent inflammation in DDSD pathogenesis, especially in ligamentous apparatus involvement. Drug synergy manifests in blocking different levels of the inflammatory cascade - celecoxib reduces prostaglandin synthesis, while sulfasalazine suppresses transcription of pro-inflammatory cytokines (IL-6, TNF-α) and MMPs (MMP-1, MMP-3, MMP-9) at the NF-kB level [5,6].Inhibition of proteolysis (Direct + Indirect). Reduction in MMP-9 levels (by 50-70%) is one of the most significant therapy results. This effect is achieved by the synergy of doxycycline (direct inhibition of MMP-8, MMP-9, MMP-13 activity) and sulfasalazine (indirect suppression of MMP synthesis via NF-kB inhibition) [7,8]. Doxycycline also reduces chondrocyte apoptosis and type II collagen degradation. The increase in TIMP-1 levels (by 5%, statistically significant in Groups 1, 3, 4) reflects compensatory activation of natural inhibitory mechanisms and indicates restoration of the MMP/TIMP balance, which is a key factor in inhibiting disc and ligament destruction. The lack of significant TIMP-1 increase in Group 5 (p=0.085) may be related to the extremely high baseline destruction (MMP-9 80.3±2.2 ng/ml), where the system's compensatory capacities are exhausted.Pain syndrome control (Nociception + Neuropathy). Reduction in pain intensity by VAS (by 40-60%) and improvement in functional status by the Scandinavian Questionnaire (by 40-60%) directly correlate with suppression of inflammation and proteolysis. This confirms the leading role of the nociceptive pain component, associated with inflammation and mechanical compression. Personalized addition of duloxetine for DN4 ≥4 points (Groups 3, 4, 5) ensured a statistically significant reduction in the neuropathic pain component (DN4 decreased by 40-50%, p<0.001). Duloxetine, inhibiting serotonin and norepinephrine reuptake, reduces central sensitization in the dorsal horns of the spinal cord [10]. An additional contribution to reducing neuropathic pain comes from powerful suppression of IL-6, contributing to decreased neuroinflammation in radicular compression [11]. The absence of DN4 dynamics in Groups 1 and 2 (DN4<4 points) confirms the adequacy of therapy selection without duloxetine when nociceptive pain predominates.Clinical significance and personalization:Combined therapy provides statistically significant and clinically relevant improvement in patient condition even in severe forms of DDSD (Group 5: VAS decreased by 60%, Scandinavian Questionnaire - by 60%).Personalization as a key success factor. The results emphasize the critical importance of personalizing the treatment regimen.Duloxetine (DN4 ≥4): Its prescription strictly according to neuropathic pain indications allowed achieving a significant reduction in DN4 and improving the quality of life in patients with radicular syndromes.Role of Sulfasalazine: Its efficacy in suppressing NF-kB-dependent inflammation (especially IL-6) was most noticeable in ligamentum flavum hypertrophy (Group 2, IL-6 decreased by 60%) and severe combined forms (Groups 4 and 5, IL-6 decreased by 65-70%). This confirms the necessity of its mandatory inclusion in the regimen for DDSD with signs of active systemic inflammation (CRP >3 mg/l, IL-6 >5 pg/ml).Efficacy monitoring. Dynamics of IL-6 and CRP showed a strong correlation with clinical improvement (r=-0.75 ÷ -0.82, p<0.01) and can be used as an objective laboratory criterion for the effectiveness of the ongoing therapy.The proposed therapy regimen demonstrated an acceptable safety profile. Monitoring leukocytes against sulfasalazine and APTT/transaminases against duloxetine did not reveal clinically significant deviations requiring drug discontinuation. Side effects (mild dyspepsia with celecoxib, drowsiness with duloxetine) were insignificant and transient.
5. Conclusions
- Combined pathogenetic therapy, including celecoxib, sulfasalazine, doxycycline, and duloxetine, represents a scientifically substantiated strategic approach to treating degenerative-dystrophic spinal diseases, whose efficacy is due to synergistic effects on key pathogenetic mechanisms, including dual suppression of inflammation through the combination of celecoxib (COX-2 inhibitor) and sulfasalazine (NF-kB blocker), leading to a significant reduction in IL-6 by 50-70% and CRP by 30-55%, as well as combined inhibition of proteolysis via doxycycline (direct MMP inhibitor) and sulfasalazine (indirectly via NF-kB), supplemented with zoledronic acid for MMP-9 levels above 50 ng/ml, providing a reduction in MMP-9 by 50-70% and an increase in TIMP-1 by 5%, while multi-level pain control is achieved through suppression of inflammation and proteolysis (effect on nociception) combined with duloxetine for DN4≥4 to correct the neuropathic component, collectively contributing to a reduction in VAS by 40-60% and DN4 by 40-50%.The efficacy of therapy directly depends on pathology severity and the strictness of personalization, based on assessment of the neuropathic pain component (DN4) and destruction activity (MMP-9). The maximum effect is achieved in severe combined forms of DDSD (disc herniations + ligamentum flavum hypertrophy). The key role of sulfasalazine is confirmed by its ability to specifically block NF-kB, especially in the inflammatory phenotype. Monitoring dynamics of IL-6 and CRP is recommended as an objective efficacy criterion.Thus, the proposed combined therapy allows control over inflammation, destruction, and pain even in critical forms of DDSD, opening prospects for targeted treatment based on pathogenetic phenotyping of patients. Further studies should focus on evaluating long-term results and the therapy’s impact on DDSD progression rates.
ACKNOWLEDGEMENTS
- The authors express sincere gratitude to the departments of neurology at Andijan State Medical Institute for material collection. Special appreciation to the patients who agreed to participate in this study; their trust and patience played a key role in obtaining scientific results. Separate gratitude to Adambayev Z.I., Professor of the Department of Neurology, Medical Psychology and Psychotherapy at Urgench State Medical Institute, for methodological support and valuable recommendations during the study and article writing. The authors also thank the Center for Development of Professional Qualification of Medical Workers under the Ministry of Health of the Republic of Uzbekistan for creating conditions for implementing this project and providing a modern research base.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML