-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3980-3985
doi:10.5923/j.ajmms.20251511.52
Received: Oct. 15, 2025; Accepted: Nov. 6, 2025; Published: Nov. 14, 2025

Trend in the Occurrence of CHD and Some Risk Factors Among People with Impaired Glucose Tolerance
U. K. Kayumov1, N. G. Ashurova2
1Center for the Development of Professional Qualifications of Medical Professionals, Uzbekistan
2Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: U. K. Kayumov, Center for the Development of Professional Qualifications of Medical Professionals, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Metabolic syndrome (MS) is a combination of interrelated metabolic disorders such as obesity, hyperglycemia, hyperlipidemia, and hypertension. These disorders significantly increase the risk of developing a number of comorbid and polymorbid diseases, including atherosclerosis, coronary heart disease( CHD), cerebrovascular disorders (CVD), arrhythmias, heart failure, liver, kidney, bone, joint and muscle diseases, psychiatric disorders, gout, chronic kidney disease (CKD), hemostasis disorders, neurosis and depression.
Keywords: Metabolic syndrome, Impaired glucose tolerance, Insulin resistance
Cite this paper: U. K. Kayumov, N. G. Ashurova, Trend in the Occurrence of CHD and Some Risk Factors Among People with Impaired Glucose Tolerance, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3980-3985. doi: 10.5923/j.ajmms.20251511.52.
1. Introduction
- DM and IGT have a significant impact on the risk factors for various diseases. At the same time, they themselves are among the most common metabolic disorders that significantly increase the risk of developing other diseases. According to the International Diabetes Federation (IDF), the number of people with diabetes exceeded 537 million in 2021, and this figure may increase to 783 million by 2045 [56]. These conditions are associated with a variety of risk factors, such as hypertension, obesity, dyslipidemia, hyperuricemia, intestinal microflora disorders, micronutrient deficiencies, and psychiatric disorders, including depression and neurosis (3, 10).. These risk factors not only worsen the course of the underlying disease, but also contribute to the development of cardiovascular diseases, chronic kidney failure, cancer, and other complications. Therefore, the study of the influence of DM and IGT on these risk factors, with an emphasis on their prevalence, pathogenesis and clinical significance, is important for understanding the mechanisms of formation of comorbid diseases [1,7,52]. Thus, the metabolic syndrome has a significant impact on the development of many comorbid and polymorbid diseases. The high prevalence of conditions such as atherosclerosis, CHD, NMC, liver and kidney diseases highlights the need for early detection and comprehensive treatment of MS [2,19,53]. Special attention should be paid to the prevention and modification of risk factors, which can significantly reduce the burden on the health system and improve the quality of life of patients.The modern concept of prevention of internal diseases is based on risk factors. In order to correctly assess the pathogenesis of the disease, it is necessary to have information about its causes, about the factors that cause this disease, about the factors that worsen the course of this disease, as well as about comorbid conditions [5,16,50].Based on this, we made an attempt to study the main components of the metabolic syndrome. It should be noted that the main components of the metabolic syndrome are very important and serious risk factors for cardiovascular diseases. A very important point in studying various risk factors is to identify them [3,12,42].Objective: To study the dynamicsin the prevalence of CHD and some risk factors among individuals with impaired glucose tolerance.
2. Material and Methods
- A total of 2,394 people were examined. Of these, 894 were examined in Tashkent under the population research program, and 67 patients with metabolic syndrome underwent an in-depth study. The survey was also conducted among the unorganized population of the city of Bukhara. At the same time, to study the dynamics of the prevalence and levels of the main components of MS, 2 groups of individuals were examined. The first group - 797 people were examined in 2006 (materials of the population study of Bukhara residents). After 15 years, 703 more people were examined under the same program (with additions). Both times, the research program provided for the identification of the main components of MS [4,13,51].Research methods: the following methods were used in the work: epidemiological, questionnaire, biochemical and instrumental. Epidemiological methods included generally accepted methods for population studies: selection of a representative sample, achievement of a sufficient population response of at least 75%, application of standardized and unified methods, all methods used represent the WHO list for population work [8,12,18,55]. Biochemical methods of research: - the state of glycemia was studied according to the results of a standard glucose tolerance test (TSH). Along with the usual tests-fasting glucose and 2 hours after sugar load, glycemia was also determined after 1 hour. The glucose content was determined in capillary blood using an automatic glucose analyzer. The indicators were evaluated according to the WHO criteria (WHO, 1999): IGT-fasting hyperglycemia (fasting glucose level >5.6 mmol/l and <6.1 mmol/L, with glycemic levels 2 hours after glucose loading <7.8 mmol/l; hyperglycemia 2 hours after glucose loading (glucose level >7.8 mmol/l and <11.1 mmol/ l at normal fasting blood glucose levels. In order to study the sympathoadrenal phase of the glycemic curve, in addition to the WHO criteria, cases of IGT associated with hyperglycemia were detected 1 hour after glucose loading (glucose level >8.88 mmol/ l). Diabetes mellitus - fasting hyperglycemia >6.1 mmol/l and 2 hours after glucose loading >11.1 mmol/l.- blood lipids: the level of cholesterol (CH) and triglycerides (TG) was determined in venous blood. > 6,1 mmol/L Hypercholesterolemia (HC) was recorded at a cholesterol level > 6.1 mmol/L Hypercholesterolemia, and TG > 1.7,7 mmol/L was taken for hypertriglyceridemia (IGT). - glycosylated hemoglobin (HbA1c) was studied in venous blood by colorimetric method. HbA1c levels below 5.7% were considered normal, values from 5.7% to 6.4% were assessed as IGT, and when HbA1c levels exceeded 6.4%, diabetes mellitus was diagnosed.Instrumental research methods: - when assessing body weight, the criteria of the WHO International Group on Obesity (IOTF WHO, 1997) were used, based onthe Ketle index levels: weight(kg)/height(m)2. Cases of a Ketle index level from 25 to 29.9 were attributed to BMI, and if the IQ level was ≥30, it was recorded as obesity. Abdominal obesity (AO) was detected in accordance with the recommendations of the International Diabetes Federation [1,3,27].- The ECG was recorded at rest in 12 standard leads, followed by evaluation according to the Minnesota Code criteria (Rose, and Blackborn, 1968) [16,17,51].- blood pressure (BP) was measured according to the standard method, taking into account the average values of two measurements, blood pressure indicators were classified according to WHO recommendations (WHO, 1999). Due to the population-based nature of the study, the analysis was performed in 2 groups: group 1 - individuals with normal blood pressure: SBP < 139 mmHg.рт.ст and / or DBP < 89 mmHg.рт.ст; Group 2 - patients with hypertension-SBP >140 mmHg; DBP > 90 mmHg. 90 мм.рт.ст.
3. Results and Discussions
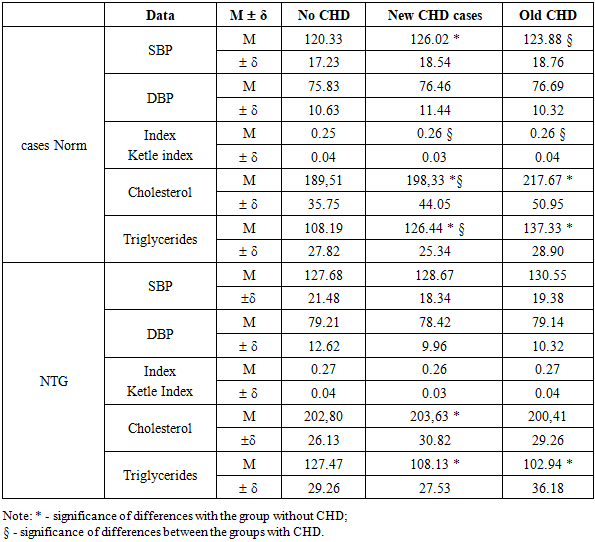
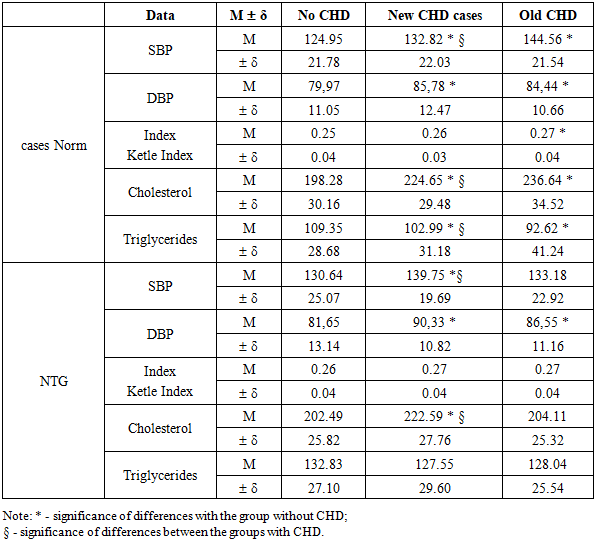
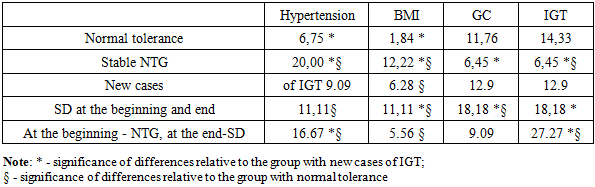
- Impaired glucose tolerance (IGT) may play a role in the pathogenesis of coronary heart disease (CHD). To date, the analysis has been made of cases of CHD identified both at the start of the study and after its completion. At the same time, the group with CHD at the end of follow-up included both patients who already had CHD at the start and people who developed the disease within 12 years after the initial screening [15,27,42].At the same time, the question of the relationship between the development of new cases of CHD and the presence of GTT is of interest. In this regard, the frequency of new cases of CHD was estimated among two categories: those with GTT detected at the start and those who developed GTT during the next 12 years of follow-up [7,2,22,40].Analysis of the obtained data indicates that there is a definite relationship between the incidence of "new" cases of coronary heart disease (CHD) and impaired glucose tolerance (IGT). Overall, the incidence of CHD almost doubled over the 12-year follow-up period. However, the nature of these dynamics differed depending on changes in the glycemic status [9,13,19,48].In the group of individuals who maintained normal glucose tolerance throughout the entire period, "new" cases of CHD were recorded in 18.40% of cases. In those who were diagnosed with HTH at the start of the study, but who experienced normalization of carbohydrate metabolism within 12 years, the incidence of new cases of CHD was 14.06%.The highest rates of CHD were observed in the group where IGT developed during follow — up-36.36% of new cases. Among individuals with stable HTH that persisted throughout the entire period (from the moment of detection to the end of follow-up), "new" cases of CHD were registered in 23.26% of cases [6,23,47].It should also be noted that almost every fifth patient with diabetes mellitus (DM), as well as those who had CHD at the start and subsequently developed DM, revealed "new" cases of CHD [2,7,33].Further, the average blood pressure, Ketle index, and lipid levels were studied among individuals with different glucose tolerance who had CHD at the start of the study and among those who developed this disease within 12 years after the start of the study [4,17,41].Analysis of the obtained data (tab.1) showed that the initial mean values of blood pressure (BP) in individuals without impaired glucose tolerance (IGT) in groups with "new" and "old" cases of coronary heart disease (CHD) did not differ statistically significantly [3,15,26]. At the same time, the level of systolic blood pressure (SBP) in the group with "new" cases of CHD was significantly higher compared to the group without CHD. In addition, lipid concentrations in the group with "old" cases of CHD were statistically significantly higher than in the group with "new" cases. Lipid levels in individuals without CHD were significantly lower compared to both groups - both with "new" and " old " cases of CHD [4,9,23,50].At the beginning of the study, the mean BP values in the analyzed groups did not differ statistically significantly in individuals with IGT, although the group with "old" cases of CHD tended to have higher values [9,23,37,54]. At the same time, the total cholesterol and triglyceride levels in individuals without CHD were significantly higher compared to patients with both "new" and "old" cases of CHD [6,33,47,51]. It should also be noted that the average BP values and Ketle index in individuals with IGT in all study groups exceeded the corresponding values in individuals with normal glucose tolerance.
|
|
|
4. Conclusions
- Thus, impaired glucose tolerance impacts the main components of metabolic syndrome differently. The association between IGT and elevated blood pressure, particularly diastolic, increases the risk of coronary heart disease in individuals with IGT. The association of cholesterol and triglyceride levels with IGT among individuals with "new" and "old" cases of CHD, as well as among individuals without CHD, is quite complex and unstable [32,47,53,51].The formation of new cases of risk factors is closely associated with the presence of IGT, as well as with its transition to manifest diabetes mellitus (DM). This is especially pronounced among patients with newly diagnosed IGT and stable IGT.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML