-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3964-3966
doi:10.5923/j.ajmms.20251511.48
Received: Oct. 7, 2025; Accepted: Nov. 5, 2025; Published: Nov. 14, 2025

Autologous Reconstruction of Central Anterior Vaginal Wall Defects: A Mesh-Free Surgical Technique
Yuldashev Sanjar Keldiyarovich
PhD, Republican Specialized Scientific and Practical Medical Center for Maternal and Child Health, Tashkent, Uzbekistan
Correspondence to: Yuldashev Sanjar Keldiyarovich, PhD, Republican Specialized Scientific and Practical Medical Center for Maternal and Child Health, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This article presents a surgical technique for autologous reconstruction of central anterior vaginal wall defects without the use of synthetic mesh implants. The method involves mobilization and reinforcement of native fascial and muscular tissues to restore anatomical integrity. A prospective cohort of patients with stage II–III pelvic organ prolapse underwent mesh-free anterior colporrhaphy. Clinical outcomes were assessed using POP-Q staging, symptom questionnaires, and postoperative complication rates. The approach demonstrated effective anatomical correction, significant improvement in pelvic symptoms, and low recurrence over a 12-month follow-up. No mesh-related adverse events were observed, and vaginal elasticity was preserved. The technique offers a biologically compatible alternative to synthetic implants, minimizing foreign body reactions. Autologous repair may be considered a safe and effective option in selected patients seeking mesh-free prolapse surgery.
Keywords: Pelvic organ prolapse, Anterior vaginal wall, Autologous reconstruction, Mesh-free surgery, Native tissue repair, Colporrhaphy, Surgical technique
Cite this paper: Yuldashev Sanjar Keldiyarovich, Autologous Reconstruction of Central Anterior Vaginal Wall Defects: A Mesh-Free Surgical Technique, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3964-3966. doi: 10.5923/j.ajmms.20251511.48.
1. Introduction
- Pelvic organ prolapse (POP) remains one of the most prevalent and socially significant conditions in gynecology, exerting a substantial impact on women's quality of life worldwide. It is characterized by the descent of pelvic organs such as the bladder, uterus, and rectum beyond their normal anatomical positions due to weakening of the pelvic floor support structures [1,2]. Clinical manifestations of POP range from mild discomfort to severe forms accompanied by urinary, defecatory, sexual, and physical dysfunction. Epidemiological studies report a wide prevalence range from 2.9% to 53% depending on diagnostic criteria and patient age. In the United States, the rate of clinically significant prolapse reaches 25%, while in Japan it approaches 46%. The true prevalence may be even higher, as mild cases often remain asymptomatic and undetected during routine examinations [3,4,5]. According to projections by the World Health Organization and demographic models, the number of women affected by POP is expected to increase by 50–70% by 2050 due to global population aging. Age is a major risk factor, with a marked rise in prolapse incidence among women over 60, attributed to hormonal changes, reduced connective tissue elasticity, and diminished muscle strength. However, up to 47% of cases occur in women of working age, underscoring the socioeconomic relevance of the condition beyond geriatric populations. POP contributes to reduced productivity, limited physical activity, and impaired psychosocial well-being, imposing a significant burden on healthcare systems [6,7]. The economic impact includes direct costs for diagnosis and treatment, as well as indirect losses from temporary or permanent disability. Surgical interventions for prolapse account for up to 28% of gynecological procedures, with cystocele and uterine prolapse corrections being the most common. Despite the availability of various surgical techniques, including synthetic mesh implants, recurrence and mesh-related complications remain pressing concerns, prompting the search for safer and more physiological reconstructive approaches [8,9,10]. In recent years, organ-preserving procedures using autologous tissues particularly vascularized flaps have gained attention for their ability to maintain blood supply, reduce rejection and infection risks, and enhance the durability and functionality of reconstructed structures. In light of the growing demand for effective and safe POP treatments, the development and implementation of such techniques represent a critical direction in modern reconstructive gynecology. Therefore, evaluating the efficacy and safety of vascularized autologous flap repair for central anterior vaginal wall defects holds significant clinical and social relevance. The findings may contribute to optimizing surgical strategies, reducing recurrence rates, and improving patient quality of life in alignment with global trends toward personalized and minimally invasive medicine [11,12].Objective of the study: to evaluate the clinical efficacy and safety of using a vascularized autologous flap in the surgical correction of central anterior vaginal wall defects.
2. Materials and Methods
- This prospective clinical study included a total of 130 female patients diagnosed with pelvic organ prolapse (POP), who sought medical care at the Republican Specialized Scientific and Practical Medical Center for Maternal and Child Health in Tashkent, Uzbekistan, between 2022 and 2024. All participants were evaluated and treated within the framework of a standardized institutional protocol approved by the local ethics committee. Inclusion criteria comprised women aged 35 to 78 years with stage II or higher POP according to the Pelvic Organ Prolapse Quantification (POP-Q) system, presenting with symptomatic anterior compartment defects. Exclusion criteria included prior pelvic reconstructive surgery with mesh, active pelvic infection, malignancy, or contraindications to surgical intervention. Preoperative assessment involved detailed gynecological examination, POP-Q staging, urodynamic testing when indicated, and completion of validated quality-of-life questionnaires. All surgical procedures were performed by experienced urogynecologic surgeons using a mesh-free technique based on autologous tissue reconstruction. Patients were followed postoperatively at regular intervals to assess anatomical outcomes, symptom resolution, complication rates, and patient satisfaction.
3. Result and Discussion
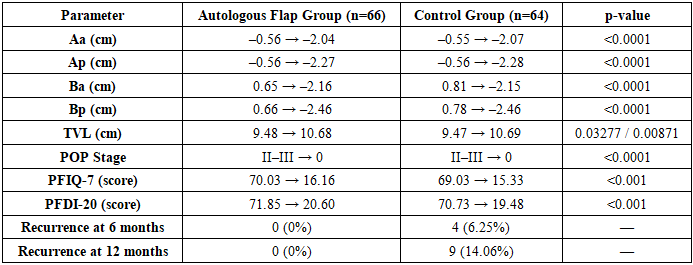
- Significant postoperative improvements were observed in both study groups based on POP-Q measurements, subjective symptom scores, and recurrence rates. At 6 months post-surgery, none of the patients in either group exhibited clinical signs of pelvic organ prolapse (POP), indicating successful anatomical restoration. Comparative analysis of POP-Q point localization before and after surgery revealed statistically significant changes across all measured parameters (p < 0.05), confirming the efficacy of both surgical approaches.In the autologous flap group (AFG, n = 66), the mean values of anterior compartment points (Aa, Ba) improved from –0.56 ± 0.25 cm and 0.65 ± 0.17 cm preoperatively to –2.04 ± 0.11 cm and –2.16 ± 0.12 cm postoperatively, respectively (p < 0.0001). Similar improvements were noted in the control group (CG, n = 64), with Aa and Ba shifting from –0.55 ± 0.22 cm and 0.81 ± 0.31 cm to –2.07 ± 0.10 cm and –2.15 ± 0.11 cm, respectively (p < 0.0001). Posterior points (Ap, Bp) and total vaginal length (tvl) also showed significant enhancement in both groups. The average prolapse stage decreased from grade II–III to 0 in all patients postoperatively.Subjective quality-of-life assessments using PFIQ-7 and PFDI-20 questionnaires demonstrated marked symptom relief. In the AFG, PFIQ-7 scores decreased from 70.03 ± 6.58 to 16.16 ± 6.65, and PFDI-20 scores from 71.85 ± 4.84 to 20.60 ± 5.53 (p < 0.001). The CG showed similar reductions: PFIQ-7 from 69.03 ± 6.70 to 15.33 ± 6.56, and PFDI-20 from 70.73 ± 5.00 to 19.48 ± 5.95 (p < 0.001). The mean score differences were statistically significant in both groups (p = 0.000001), with average reductions of 53.88 ± 2.12 and 51.26 ± 3.55 points in the AFG, and 53.71 ± 1.79 and 51.25 ± 3.64 points in the CG for PFIQ-7 and PFDI-20, respectively.During follow-up at 6 and 12 months, no cases of POP recurrence were recorded in the autologous flap group. In contrast, the control group exhibited 4 cases (6.25%) of recurrence at 6 months and 9 cases (14.06%) at 12 months, indicating a statistically and clinically relevant difference in long-term outcomes.
|
4. Conclusions
- This study confirms that autologous reconstruction using vascularized native tissue flaps is a clinically effective and biologically safe alternative to synthetic implant-based techniques for the correction of central anterior vaginal wall defects in patients with pelvic organ prolapse. The mesh-free surgical approach demonstrated excellent anatomical restoration, with statistically significant improvements in POP-Q parameters and complete resolution of prolapse in all patients at 6 months postoperatively. Importantly, the autologous flap group showed superior long-term durability, with no recurrence observed at 12 months, in contrast to the control group, which exhibited a recurrence rate of 14.06%.The use of vascularized autologous tissue offers distinct advantages over synthetic mesh implants, including preservation of native tissue elasticity, reduced risk of foreign body reaction, and elimination of mesh-related complications such as erosion, infection, and chronic pain. These benefits translate into improved functional outcomes and enhanced patient-reported quality of life, as evidenced by significant reductions in PFIQ-7 and PFDI-20 scores.In the context of growing concerns regarding mesh safety and regulatory restrictions in many countries, the findings of this study support the adoption of mesh-free, organ-preserving techniques as a preferred strategy for anterior compartment prolapse repair. Autologous flap reconstruction not only aligns with principles of personalized and minimally invasive pelvic surgery but also addresses the need for sustainable, complication-free treatment options in both geriatric and working-age populations. Further research and long-term follow-up are warranted to validate these outcomes and refine surgical protocols for broader clinical application.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML