Kurbanov Jalalkhan Bakhromovich1, Mamajonov Bokhadirjon Solijanovich2
1Fergana Public Health Medical Institute, Fergana, Uzbekistan
2Andijan State Medical Institute, Fergana, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Thromboembolic complications after surgery are important in determining the molecular substrates underlying damage to the pulmonary arteries in thromboembolism. In particular, detection of blood clotting factors in vascular endothelium using SD 31 and SD 63 markers is an important link in the assessment of gene expression.
Keywords:
Thromboembolism, Immunohistochemical examination, Pathomorphology, Thrombosis
Cite this paper: Kurbanov Jalalkhan Bakhromovich, Mamajonov Bokhadirjon Solijanovich, Immunohistochemical Characteristics of Vessels in Pulmonary Artery Thromboembolic Developed After Surgery, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3958-3963. doi: 10.5923/j.ajmms.20251511.47.
1. Introduction
Pulmonary embolism (PE) is the final stage of venous thromboembolism (VTE) and is the third leading cause of death among circulatory diseases worldwide, after myocardial infarction and stroke, with an estimated 0.1% of the world's population, or 8.5 million people, dying from this disease each year [1,2]. In the United States, approximately 600,000 patients are hospitalized with PE each year, 33% of whom die in critical condition [3,4]. PE remains one of the leading causes of death in surgical hospitals. It has been reported that it occurs in 4.1% of patients in vascular surgery, 14.3% in abdominal surgery, 33.3% in surgical surgery, 30.7% in neurosurgery, and 40% in proctology. Thromboembolic complications remain one of the leading causes of sudden death, particularly in the elderly. Despite significant progress in understanding coagulation and vascular-thrombotic mechanisms, the pathogenetic features of age-related changes in the vascular wall and endothelial response during thromboembolism remain insufficiently studied. One of the key links in the pathogenesis of thrombogenesis is the interaction between endothelial cells, platelets, and their activation markers, among which CD63 - a tetraspanin protein localized in the granules of platelets and endothelial cells, including Weibe-Palade bodies [5,6,7] - plays a major role. It is involved in the processes of degranulation, adhesion, and secretion of biologically active substances, including von Willebrand factor and P-selectin, which regulate the adhesive and aggregative properties of the vascular wall. However, in clinical and morphological practice, the dynamics of CD63 expression in blood vessels and thrombi depending on patient age and stage of the thromboembolic process remain poorly understood. In particular, there is a lack of data on: the morphofunctional characteristics of endothelial cells and their secretory activity in thromboembolic events among different age groups; changes in CD63 expression as a marker of platelet and endothelial cell degranulation depending on the severity and localization of thrombosis; the relationship between age-related vascular involution and decreased expression of adhesion and secretion markers such as CD63, von Willebrand factor, and P-selectin; pathomorphological signs of “aging endothelium” in the context of increased thromboembolic susceptibility. With advancing age, a decline in the metabolic and secretory activity of the endothelium is observed, along with a reduction in the number of Weibel–Palade bodies, degeneration of platelet granular structures, and disruption of intercellular interactions, which collectively increase the risk of unregulated thrombus formation [8,9,10,11]. These processes form the basis of age-dependent vascular involution, yet their morphological and immunohistochemical characteristics in thromboembolic complications have been only fragmentarily described [12,13,14]. Thus, the relevance of the present study is determined by: the need for an in-depth investigation of the molecular and morphological mechanisms of vascular wall aging and their role in the development of thromboembolic complications; the absence of systematized data on CD63 expression and other endothelial markers across various age groups in acute and chronic thromboembolism; the limited diagnostic value of conventional histological techniques for identifying age-related endothelial changes and the need for immunohistochemical markers (CD63, vWF, P-selectin) to assess the degree of vascular aging; the practical importance of defining age-related expression patterns of CD63, which may contribute to clarifying the morphogenesis of thromboembolism and predicting thrombotic risk in both clinical and forensic pathology. Taken together, these factors emphasize the necessity of a comprehensive morphological and immunohistochemical analysis of vascular–platelet interactions in thromboembolic complications, taking into account age-related vascular alterations.Aim of the StudyThe aim of this study is to investigate the age-related immunohistochemical features of CD63 expression in vascular endothelium and thrombi in cases of thromboembolic complications, in order to identify the molecular and morphological mechanisms of endothelial aging and its role in the pathogenesis of thrombus formation and vascular damage.
2. Materials and Methods
The study used morphological, morphometric, immunohistochemical, and statistical methods to improve the assessment of age-related pathomorphological changes in pulmonary arteries and lung tissue in patients with pulmonary thromboembolism that developed after surgery. The material was pulmonary arteries and lung tissue from autopsies of 78 women who died of pulmonary embolism.
3. Results and Discussion
Paraffin-embedded biopsy materials were subjected to immunohistochemical examination using monoclonal antibodies using standard methods. The CD31 PECAM marker is a marker indicating the level of platelet-1, which adheres to the endothelium. The glycoprotein protein is mainly expressed on vascular endothelial cells, platelets, granulocytes, monocytes, and some T lymphocyte coreceptors, indicating the development of new vessels, and if it is present on the inner surface of the vessel, then thromboplastin is formed and the damage process on the inner surface of the vessel is acute.This glycoprotein is a transmembrane protein, and normally it does not react with the CD 31 PECAM marker and is not masked. Since the free location of these glycoproteins indicates the absence of intercellular contact, in our study, the complication of thromboembolism indicates the degree of vascular damage.This also means that this glycoprotein in the perivascular areas accelerates the process of angiogenesis by connecting other types of mesenchymal cells, and if the yellow-golden expression is detected on the inner surface of the vessels, it proves that the process of injury and thrombogenesis is taking place in these vessels.If, upon examination, it is detected in a scattered and irregular appearance in the stroma of the prevascular and intramural tissue, this means that the process of transformation of endothelial cells from mesenchymal cells and neoangiogenesis is taking place.In our study, there were 3 groups, and the lung tissue and small-caliber vessels of 18-44, 45-59, 60-74 years old were stained with the CD 31 PECAM marker. The following immunohistochemical analysis showed a high positive expression of the CD 31 PECAM marker in 6 out of 26 patients aged 18-44 years, which clinically and morphologically indicates a predominance of vascular damage at this age, and this indicates an acceleration of the process of thrombogenesis due to damage to the inner surface of small-caliber blood vessels of the lungs. The systemic appearance of leukotrienes and cytokines released from damaged tissues in the period after any type of surgical intervention indicates a predominance of the ability of the body to cause a systemic vascular response, mainly in 18-44 years, which also indicates a predominance of the body's age-related hyperergic response and the fact that in the period after thromboembolic complications, small-caliber vessels of the lungs are damaged mainly from small-caliber vessels.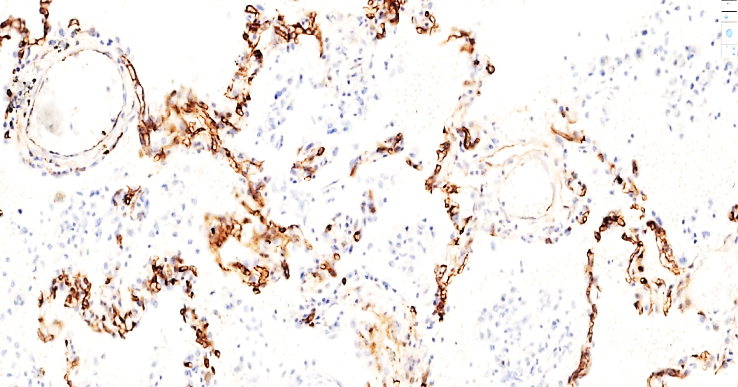 | Figure 1. A 41-year-old patient died of pulmonary embolism on the 4th day after surgery. Moderately positive expression of the CD 31 PECAM marker is detected in the intima of the alveolar walls and small-caliber vessels of the lung tissue. Staining is Dab chromogen. Size 20x10 |
 | Figure 2. A 40-year-old patient died of pulmonary embolism on the 3rd day after surgery. Moderately positive expression of the CD 31 PECAM marker is detected in the intima of the alveolar walls and small vessels of the lung tissue. Staining is Dab chromogenic. Size 20x10 |
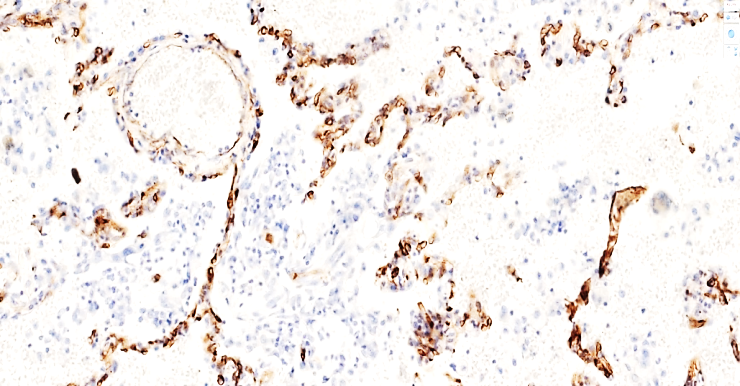
This made up 23% of the 18-44 year olds in our study. In 20 out of 26, moderate positive expression of the CD 31 PECAM marker was detected, which was also reflected in the fact that the vascular damage indicator was mainly expressed on the inner surface of medium and small caliber vessels. This, in turn, leads to the conclusion that these 18-44 year olds had moderate positive expression overall.In the next 2nd group, moderate positive expression of the CD31 PECAM marker was detected in 3 out of 26 in the 45-59 year olds, which was 11.54%. Low positive expression of CD31 PECAM was detected in 6 out of 26, which was 23%. A negative reaction was detected in 17 cases, mainly due to the fact that the rate of vascular damage after any surgical procedure in patients aged 45-59 years is lower than in patients aged 18-44 years, which is explained by the advantage of morphological adaptation and age-related tolerance, which is also explained by a decrease in the biological resistance of the vessels.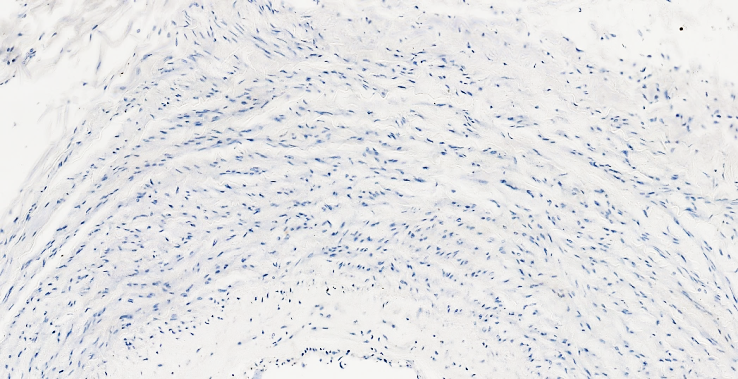 | Figure 3. A 58-year-old patient died of pulmonary embolism on the 3rd day after surgery. Lung tissue shows a negative reaction of the CD 31 PECAM marker in the pulmonary trunk and the vessel wall. Staining is Dab chromogenic. Size 20x10 |
 | Figure 4. A 72-year-old patient died of pulmonary embolism on the 4th day after surgery. Lung tissue shows a negative reaction of the CD 31 PECAM marker in the pulmonary trunk and the vessel wall. Staining is Dab chromogenic. Size 20x10 |
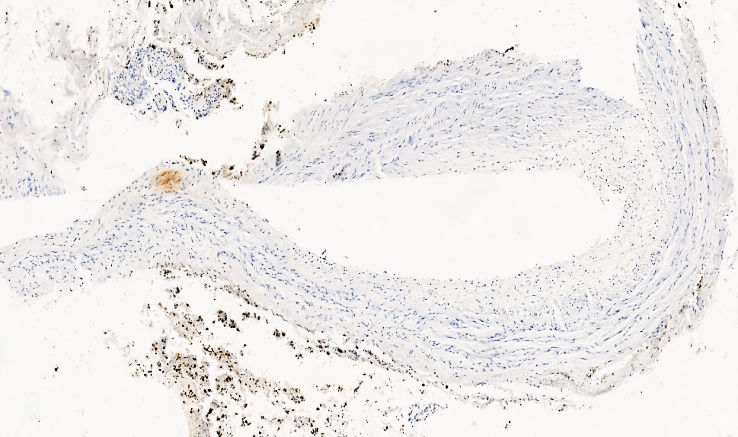
This confirms the low rate of damage to the lower vessels after the pulmonary veins were sutured with a thrombus or embolus in the postoperative period, as mentioned in the morphological studies.Our group 3, in the postoperative period in 60-74 year olds, the development of hypercoagulable syndrome mainly proceeds with the predominance of the thrombogenesis process in the vessels, and in clinical morphological terms, the increase in the biological tolerance index of the vessels, the fragility of the vessels and the very low content of the glycoprotein adsorbing protein component confirm the very low rate of angiogenesis and vascular damage. Biological aging of the vessels in 60-74 year olds and the absence of the adhesive glycoprotein that connects the endothelial cells to each other also mean that the endothelium responds very poorly to any influences. Of the 26 cases in group 3, 5 had low positive expression, while 21 had a predominance of negative reaction. Those that gave a positive reaction were mainly detected in tissues from the primary lung branch to the front, and were almost not detected in the 2nd and 3rd lung branches.Our next marker, CD 63, is also a membrane adhesive protein belonging to the tetraspanin family, and is a glycoprotein. The functional characteristics of this protein are characterized by staining Weibel-Palade bodies, which are mainly present in the blood and endothelial cells of the blood vessel wall. These bodies are mainly found in the endothelial cells of all vessels and contain the von Willebrand factor and the P-selectin protein. This factor is mainly due to hypercoagulability indicators in blood coagulation, adhesion of platelets in the tissue to the vascular wall, and since P-selectin is detected only in active platelets, they are collectively called Weibel-Palade bodies. Normally, these bodies are expressed by a negative reaction to the CD 63 marker, which means that the vessels are at rest.Now, in the period after any surgical operation in the body, mainly within 3-5 days, prostaglandins and cytokines produced in damaged tissues have a damaging effect on the vascular walls, which also depends on the type of surgical operation, its invasiveness, trauma, etc., and if the operation occurs with an average of 500-700 ml of bleeding, this is manifested by a decrease in positive reactions of this factor. For example, in polytraumas, bleeding of more than 1.2 liters gives a negative reaction to the CD 63 marker, which is explained by a sharp decrease in platelets, endotheliosis, desquamation of endothelial cells, and collapse of the vascular lumen. In group 1 of our study, in terms of the expression of the CD 63 marker, this indicator was expressed in 21 out of 26 subjects aged 18-44 years, which was also reflected in the above-mentioned age-related vascular response and adhesion of platelets to the endothelial layer and the presence of Weibel-Palade bodies in the cytoplasm of endothelial cells. The presence of Weibel-Palade bodies in the form of golden yellow granular inclusions in endothelial cells, confirming the presence of von Willebrand factor and P-selectin protein, is also explained by the fact that in the thromboembolic complications that occur in the postoperative period, damage to the vascular intima and activation of blood coagulation factors are also found in the small-caliber vessels of the pulmonary arteries. This, in turn, reveals the main point of the process of migration of the transferred thrombi and thrombogenesis.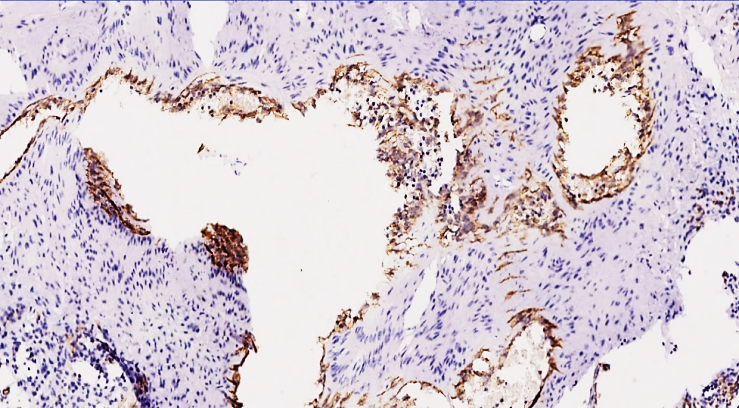 | Figure 5. 41-year-old 4 days after surgery. High positive expression of the SD 63 marker is determined mainly on the surface of the inner surface of the vessel endothelium and thromboblastin substrate. This also means that the vessel is damaged and the process of thrombogenesis is advanced. Paint Dab chromogenic. Size 4x10 |
 | Figure 6. 44-year-old 5 days after surgery. High positive expression of SD 63 marker. On the surface of the vascular endothelium, platelet adhesion aggregation and sediments in the granular form of different sizes are determined. Aggregation of platelets preserved in thrombi formed in the vessel cavity is determined in granular form. Dab chromogen. The size is 20x10 |
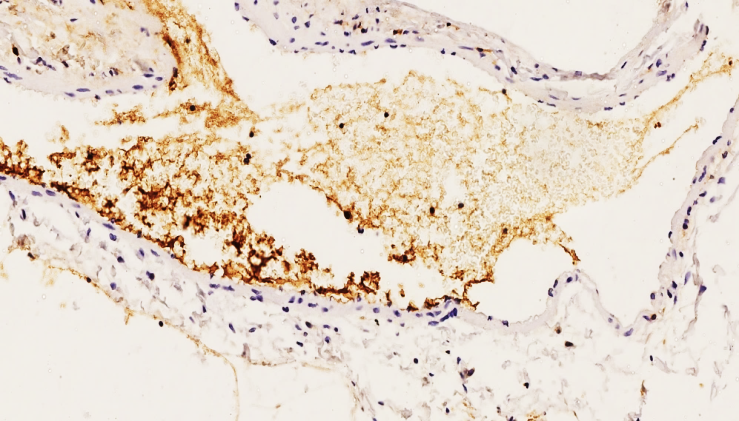
Specifically, activation of Weibel-Palade bodies is manifested by the sequestration of leukocytes around the perimeter of thrombogenic foci, as a result of simultaneous activation of the P-selectin adhesive protein.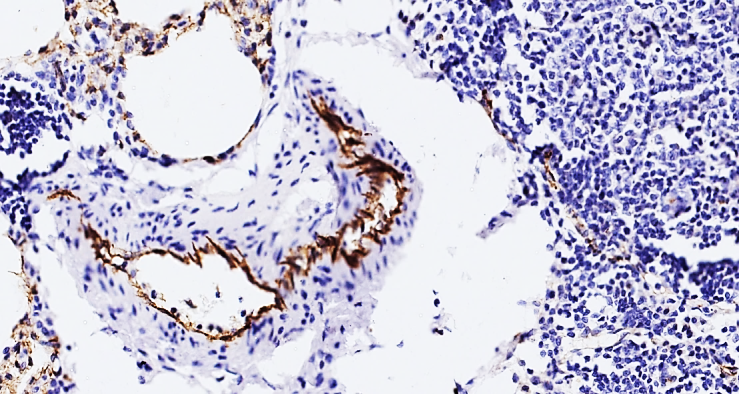 | Figure 7. 42-year-old woman. 5 days after surgery. High positive expression of the CD63 marker. Adhesive aggregation of platelets in the form of plaques on the surface of the vascular endothelium and Weibel-Palade bodies in the cytoplasm of endothelial cells are detected. Staining is Dab chromogen. Size 20x10 |
Thus, in group 1, aged 18-44, the lesion develops not only with thromboembolism, but also after surgery (see Fig. 7-10). In 80.76% of cases, high positive expression was detected, and Weibel-Palade bodies were clearly masked in the cytoplasm of endothelial cells of the vascular intima. As a result of the predominance of the intravascular response in the 18-44-year-old contingent, the process of thrombogenesis on the surface of the 3rd, 4th and 5th lamellas of the lower branches of the pulmonary tissue is characterized by the development of such characteristic signs as aging, which also confirm the decrease in the morphofunctional parameters of the vessels.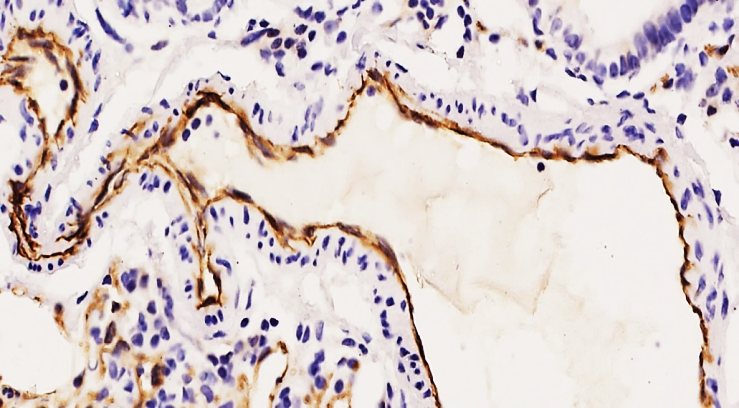 | Figure 8. 37-year-old woman. 4 days after surgery. Moderately high positive expression of the CD63 marker. Adhesive aggregation of platelets in the form of flat sheets on the surface of the vascular endothelium and Weibel-Palade bodies in the cytoplasm of endothelial cells are detected. Staining is Dab chromogenic. Size 40x10 |
Thus, in our group 2, the expression of the CD 63 marker in 45-59-year-olds was detected in granular deposits formed by platelet aggregation in thrombi in 13 out of 26 cases (61.9%), confirming the quiescence of the vascular endothelium and the sharp decrease in the response of the vascular inner surface to any damage. In 3 out of 11.54% of cases, the CD 63 marker was found to be moderately and lowly positive on the vascular endothelium surface in 45-49-year-olds, which also confirms the existence of critical age-related transition points in the age boundaries of the transitional group 2.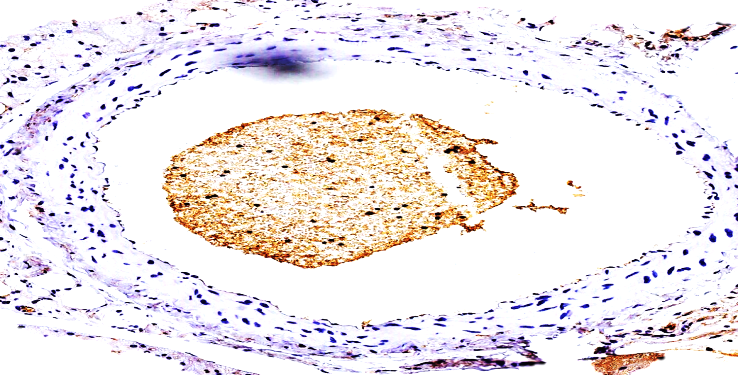 | Figure 9. Group 2. 51-year-old woman. Positive expression of thromboplastins in the form of adhesive granular thromboplastins in the vascular lumen. No changes in the vascular endothelium are detected. Staining is Dab chromogenic. Size 20x10 |
That is, among the 18-44 and 45-59-year-olds, the degree of damage in the 41-44 and 45-49-year-olds is close to each other, which confirms the fact that the vascular reactivity is close to each other, and after the age of 50, these indicators decrease sharply.In 10 of our group, low and negative expression of the SD 63 marker was detected, totaling 38.46%. This also confirms our above-mentioned ideas, and the presence of granular adhesive thromboplastins in the migrated thrombus is mainly characterized by the presence of endothelial cells on the surface of the vascular intima.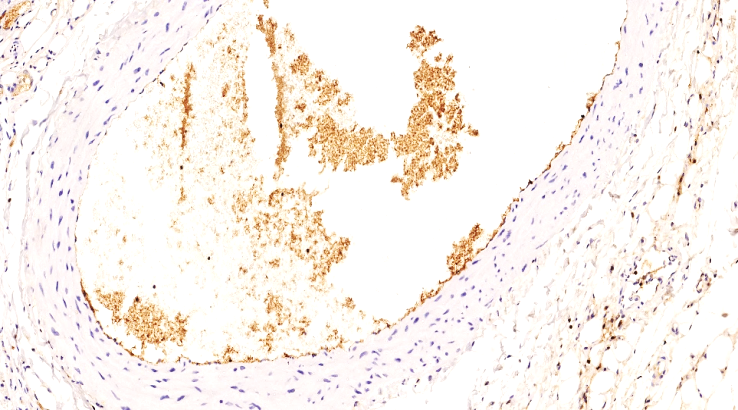 | Figure 10. Group 2. 49-year-old woman. Moderate positive expression of the CD 63 marker is detected in the thrombus in the vascular lumen and in granular platelets adhered to the vascular wall. Focal changes are detected in the vascular endothelium. Staining Dab chromogenic. Size 20x10 |
Thus, in group 2, the positive reaction of the CD 63 marker is 1.5 times lower than in group 1, and the complication of thromboembolism is that vascular damage occurs mainly due to migrated thrombi, and in acute thromboembolism, the presence of thrombi in the vascular lumen and various secretory active secretions of the endothelium in response to them were detected in 11.54% of 26 cases. This means that in various types of vascular embolism, there is a decrease in active secretion from the endothelial cells and an age-related decrease in vascular response, and an increase in vascular resistance. This confirms the involutive manifestation of biological aging in the age-related endothelium of the vessels.Clinically, morphologically, it is also proven that with age, in various thromboembolic syndromes, vessels become more fragile and prone to damage, mainly due to foreign thrombi in the vascular lumen and a very small amount of biologically active substances are produced on the inner surface of the vessel.In our next group, it is found that the mortality rate from thromboembolic complications is lower than in group 2, manifested by the retention of thrombi in areas of migration and an age-related decrease in the local response of the vessels.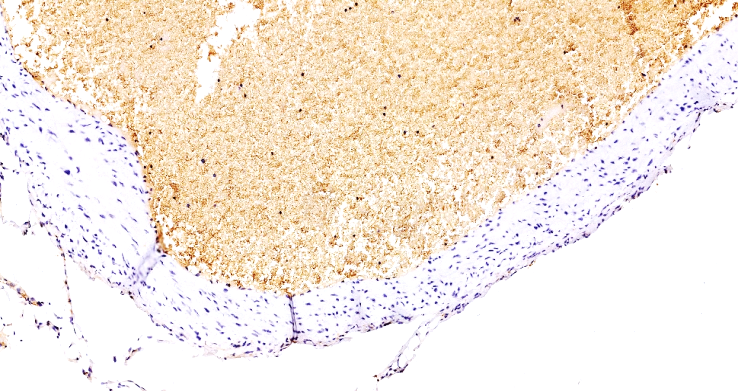 | Figure 11. Group 3. 74-year-old woman. Massive obturating pulmonary artery anterior wall thrombus, massively adhered granular platelets in the thrombus, high positive expression of the CD 63 marker. Staining is Dab chromogenic. Size 20x10 |
In particular, in our group 3, out of 26 cases from 60-74 years old, moderate positive expression of the CD63 marker was detected in 8 cases (31.13%), mainly in platelets that underwent massive granular adhesion in the thrombus in the vascular lumen, indicating that the thrombus adhered directly to the vascular wall, and Weibel-Palade bodies were almost not detected in the endothelial cells on the inner surface of the vascular wall.This indicates a decrease in the response of the endothelium to acute injuries, as well as a sharp decrease in the von Willebrand factor. This is also clinically morphologically due to age-related biological aging in the body, as a result of a sharp decrease in water, a decrease in the ability of endothelial cells to produce biologically active substances, including a critical decrease in the content of granular vesicles that bind the CD 63 marker, von Willebrand factor, and P-selectin (see Fig.10-11).In this 3rd group, a total of 18 CD 63 markers showed negative expression in relation to the inner surface of the vessel, while moderate positive expression was detected in relation to migrated thrombi. This is explained by the fact that platelets in the composition of the migrated thrombi were mainly detected in granular adhesive protein molecules.So, in group 3, among 60-74-year-olds, 8 out of 26 cases in 31.13% had medium positive expression of the SD 63 marker, 18 had low positive reaction in 69.23%, and medium positive expression in thrombi. The expression of this SD 63 marker was obtained mainly in relation to the vessel, excluding granular adherent platelets in the migrated thrombi, and the positive reactions were mainly attributed to the content of the thrombus. This confirms that the level of positive expression of the SD 63 marker in 60-74-year-olds decreased by 2.45 times compared to 18-44-year-olds, and by 1.54 times compared to 45-59-year-olds.
4. Conclusions
In pulmonary artery thromboembolism, it was found that the immunohistochemical CD 31 PECAM marker of the pulmonary arteries showed low positive expression in 55-59-year-olds and medium positive expression in 18-44-year-olds, indicating a high degree of damage to the vascular endothelium, which was explained by the production of biologically active substances by endothelial cells, and manifested as a vascular response. Based on the immunohistochemical analysis of CD63 marker expression in different age groups, distinct age-related differences in vascular and thromboembolic processes were identified. In group 2 (45–59 years), moderate positive expression of CD63 was detected both in the thrombus and in platelets adhered to the vascular wall, with focal endothelial changes. The frequency of positive endothelial reactions was 1.5 times lower than in younger individuals (group 1), indicating the beginning of endothelial involutional changes. The formation of thromboembolic complications in this group was primarily associated with vascular damage caused by migrated thrombi, while local endothelial secretory activity and reparative capacity were partially preserved. In group 3 (60–74 years), CD63 expression decreased sharply. Moderate positivity was observed only in 31.13% of cases, mainly within thrombi and granular platelets, whereas endothelial cells on the vascular surface exhibited negative or minimal reactions. The content of Weibel–Palade bodies and von Willebrand factor was significantly reduced, confirming endothelial functional exhaustion. The diminished endothelial secretory response and increased vascular rigidity reflect age-dependent biological involution of the vascular wall. Overall, with aging, there is a progressive reduction in endothelial reactivity, suppression of CD63 expression, and depletion of secretory granules containing adhesion molecules such as von Willebrand factor and P-selectin. These changes lead to reduced vascular resilience, increased susceptibility to thromboembolic complications, and impaired thrombus resolution. Thus, the obtained data morphologically and immunohistochemically substantiate that vascular aging is accompanied by a decline in CD63-mediated platelet-endothelium interactions, resulting in impaired regulation of thrombus formation and contributing to the increased incidence and severity of thromboembolic events in elderly individuals.
References
| [1] | Bhoopalan K., Rajendran R., Alagarsamy S., Kesavamoorthy N. Successful extraction of refractory thrombus from an ectatic coronary artery using stent retriever during primary angioplasty for acute myocardial infarction: a case report // Eur Heart J Case Rep. – 2019. – Vol. 3, № 1. – Article yty161. – DOI: 10.1093/ehjcr/yty161. |
| [2] | Björck M., Earnshaw J. J., Acosta S. et al.Editor’s Choice – European Society for Vascular Surgery (ESVS) 2020 Clinical Practice Guidelines on the Management of Acute Limb Ischaemia // European Journal of Vascular and Endovascular Surgery. – 2020. – Vol. 59, № 2. – P. 173–218. |
| [3] | Blom J. W., Doggen C. J., Osanto S., Rosendaal F. R.Old and new risk factors for upper extremity deep venous thrombosis // J. Thromb. Haemost. – 2005. – Vol. 3, № 11. – Р. 2471–2478. |
| [4] | Burov V. P., Kapranov S. A.Temporary placement of the cava filter to the suprarenal segment of the inferior vena cava // Angiol. Sosud. Khir. – 2005. – Vol. 11, № 2. – P. 45–47. |
| [5] | Bush R. L., Lin P. H., Bates J. T. et al. Pharmacomechanical thrombectomy for treatment of symptomatic lower extremity deep venous thrombosis: safety and feasibility study // J. Vasc. Surg. – 2004. – Vol. 40, № 5. – P. 965–970. |
| [6] | Chiam P., Kwolc V., Johan B. A., Chan C. Major pulmonary embolism treated with a rheolytic thrombectomy catheter // Singapore Med. J. – 2005. – Vol. 46, № 9. – P. 479–482. |
| [7] | De Gregorio M. A., Gamboa P., Gimeno M. J. et al. The Gunther Tulip retrievable filter: prolonged temporary filtration by repositioning within the inferior vena cava // J. Vasc. Interv. Radiol. 2003. Vol. 14, № 10. – P. 1259–1265. |
| [8] | DeYoung E., Minocha J. Inferior vena cava filters: guidelines, best practice, and expanding indications // Seminars in Interventional Radiology. – 2016. – Vol. 33, № 2. – P. 65–70. |
| [9] | Donnellan E., Khorana A.A. Cancer and venous thromboembolic disease: a review // Oncologist. – 2017. – Vol. 22, № 2. – P. 199–207. |
| [10] | Eur Respir J. 2021.Modified Geneva score for estimating the clinical probability of pulmonary embolism / European Respiratory Journal. 2021. 17 p. |
| [11] | European Society of Cardiology (ESC). 2019 ESC guidelines for the diagnosis and management of acute pulmonary embolism. European Heart Journal, 2019. – 36 p. |
| [12] | Fedullo P.F., Tapson V.F. Clinical practice. The evaluation of suspected pulmonary embolism // N. Engl. J. Med. – 2003. – Vol. 349. – P. 1247–1256. |
| [13] | Garcia M.J., Luksteijn R., Malhotra R., Amin A., Blitz L.R., Leung D.A. и др.Endovascular treatment of deep vein thrombosis using rheolytic thrombectomy: final report of the prospective PEARL multicenter register (peripheral application) AngioJet Rheolytic Thrombectomy…) // J Vasc Interv Radiol. – 2015. – Т. 26. – С. 777–785. DOI: 10.1016/j.jvir.2015.01.036. |
| [14] | Ginsberg M.S., Bach A., Kickuth R., et al. Suprarenal inferior vena cava filter placement prior to transcatheter tumor thrombus ablation // Abdominal Imaging. – 2000. – Vol. 25, № 3. – P. 233–238. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML









