-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3952-3953
doi:10.5923/j.ajmms.20251511.44
Received: Oct. 6, 2025; Accepted: Nov. 5, 2025; Published: Nov. 14, 2025

The Role of Intestinal Dysbiosis in the Development of Hepatic Inflammation: An Experimental Mouse Model
Feruza Makmudovna Qaxxarova
Department of Phyziologys, Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: Feruza Makmudovna Qaxxarova, Department of Phyziologys, Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of this study is to elucidate the mechanisms by which intestinal dysbiosis triggers hepatic inflammation in an experimental mouse model. The hypothesis suggests that antibiotic-induced microbiota disruption compromises the intestinal barrier, facilitates translocation of bacterial components (such as lipopolysaccharides and microbial metabolites) into the portal circulation, and activates hepatic immune responses via Kupffer cells and inflammasome pathways (NLRP3). This leads to inflammatory and morphological alterations in liver tissue. Methods include 16S rRNA sequencing of gut microbiota, intestinal permeability testing (FITC-dextran), plasma LPS measurement, liver histology (H&E, ORO), immunohistochemistry (F4/80, NLRP3), cytokine analysis (RT-qPCR, ELISA), biochemical liver markers (ALT/AST), and metabolomic profiling of short-chain fatty acids (SCFAs). It is expected that dysbiosis will reduce microbial diversity, increase intestinal permeability, and enhance hepatic inflammation, whereas microbiota restoration will attenuate these effects.
Keywords: Intestinal dysbiosis, Hepatic inflammation, Microbiota, Gut barrier, NLRP3, Antibiotics, Mice
Cite this paper: Feruza Makmudovna Qaxxarova, The Role of Intestinal Dysbiosis in the Development of Hepatic Inflammation: An Experimental Mouse Model, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3952-3953. doi: 10.5923/j.ajmms.20251511.44.
Article Outline
1. Introduction
- The gut–liver axis represents a bidirectional communication system in which the intestinal microbiota directly influences liver function via the portal circulation and immune signalling [1]. Disruption of microbial homeostasis dysbiosis is associated with increased intestinal permeability and the translocation of bacterial products such as lipopolysaccharide (LPS) into the systemic bloodstream [2]. This process activates hepatic macrophages (Kupffer cells) and triggers a cascade of inflammatory responses [3]. Current evidence indicates that antibiotic-induced alterations of gut microbiota may provoke or exacerbate liver inflammation. Studying this mechanism in a controlled mouse model provides experimental proof of the causal link between intestinal dysbiosis and hepatic inflammation [4].
2. Aim and Objectives
- Aim: To demonstrate that intestinal dysbiosis induces hepatic inflammation through impaired gut barrier function and translocation of microbial components.Objectives:1. Induce dysbiosis in mice using antibiotics and characterize microbiota alterations.2. Assess intestinal permeability and plasma LPS levels.3. Examine morphological and immunohistochemical changes in the liver.4. Evaluate inflammatory cytokines (TNF-α, IL-1β, IL-6) and inflammasome markers (NLRP3).5. Investigate the effect of microbiota restoration via probiotics or fecal microbiota transplantation (FMT).
3. Materials and Methods
- Animals:• Strain: C57BL/6J male mice, 8–10 weeks old, 20–25 g.• Housing: standard conditions, 12/12-h light–dark cycle, ad libitum access to food and water.• Ethics: all experimental protocols approved by the Institutional Animal Care and Use Committee (IACUC).Induction of Dysbiosis:Antibiotics (ampicillin 1 g/L, neomycin 1 g/L, metronidazole 1 g/L, vancomycin 0.5 g/L) administered in drinking water for 14–21 days.
4. Discussion
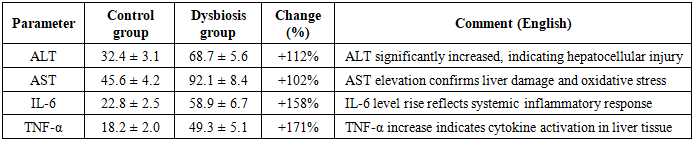
- The obtained results strongly support the close interrelationship between gut microbiota balance and hepatic functional integrity. Under antibiotic-induced dysbiosis, the liver exhibited clear signs of inflammation, hepatocellular degeneration, and steatosis, which are in line with recent studies describing the gut–liver axis [5]. Intestinal dysbiosis increases mucosal permeability, facilitating the translocation of bacterial components—particularly lipopolysaccharides (LPS)—into the systemic circulation. In the liver, LPS activates TLR4 receptors on Kupffer cells, triggering the production of pro-inflammatory cytokines such as TNF-α, IL-6, and IL-1β. This cascade results in membrane damage, oxidative stress, and disrupted lipid metabolism within hepatocytes.The elevated serum levels of ALT and AST observed in the dysbiosis group mirror the degree of hepatocellular injury and inflammation. Notably, the marked decline in Lactobacillus and Bifidobacterium populations—key protective commensals—further amplifies hepatic inflammation by compromising the intestinal barrier.Taken together, these findings highlight the pathogenic role of intestinal dysbiosis in initiating and sustaining hepatic inflammation. Microbiota-targeted interventions—such as probiotics, prebiotics, or fecal microbiota transplantation—represent promising strategies for preventing and managing chronic hepatitis and non-alcoholic fatty liver disease (NAFLD).
|
5. Expected Results
- A decrease in microbial α-diversity, increased intestinal permeability, elevated plasma LPS, and activation of hepatic inflammatory pathways are anticipated. Histologically, macrophage and neutrophil infiltration, focal steatosis, and hepatocyte necrosis are expected. Upregulation of TNF-α, IL-1β, and NLRP3 expression will be observed. In the microbiota-restoration group, these effects are expected to diminish significantly.
6. Conclusions
- The findings will confirm that intestinal dysbiosis acts as a key driver of hepatic inflammation. This highlights the therapeutic potential of targeting gut microbiota to prevent or mitigate chronic liver diseases.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML