-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3895-3902
doi:10.5923/j.ajmms.20251511.29
Received: Oct. 22, 2025; Accepted: Nov. 12, 2025; Published: Nov. 14, 2025

Frontotemporal Dementia: Clinical and Neuroimaging Features and Correlational Relationships with Hormonal Status
Abramyan Arevik Armikovna
Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Abramyan Arevik Armikovna, Andijan State Medical Institute, Andijan, Uzbekistan.
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

According to WHO data, there are currently over 55 million people worldwide suffering from dementia, more than 60% of whom live in low- or middle-income countries. Approximately 10 million new cases of dementia are recorded annually. Dementia currently ranks seventh among the leading causes of death globally and is among the main causes of disability in older people and their dependence on external assistance. In 2019, the global economic costs of dementia amounted to 1.3 trillion US dollars, of which approximately 50% of the costs are attributable to informal care and supervision of patients (e.g., by their relatives and close friends), provided on average for 5 hours a day. Frontotemporal dementia accounts for 5-7 cases of dementia.
Keywords: Dementia, Frontotemporal dementia, Primary progressive aphasia, Pick's disease, Cortisol, Dehydroepiandrosterone sulfate
Cite this paper: Abramyan Arevik Armikovna, Frontotemporal Dementia: Clinical and Neuroimaging Features and Correlational Relationships with Hormonal Status, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3895-3902. doi: 10.5923/j.ajmms.20251511.29.
Article Outline
1. Introduction
- Frontotemporal dementia (FTD) or lobar-temporal dementia (LTD) refers to a group of degenerative pathologies of the central nervous system, with predominant involvement of the frontal and anterior temporal segments of the brain, leading to the development of behavioral, speech, and personality disorders in patients [1,2,10,19,40]. This pathology is often combined with degeneration of subcortical areas [41]. In Russian literature, the term "Lobar-temporal dementia" is more commonly used, while in foreign literature, the term "Frontotemporal dementia" is mainly used.The study of frontotemporal dementia began in the 19th century with Arnold Pick, who described a patient with speech and behavioral disorders. Arnold Pick linked this condition to atrophy of specific brain regions [2,17,34]. Inclusions found during histological examination were named Pick bodies, and the disease itself was called Pick's disease. Subsequently, the term "Pick's disease" began to be applied only to a limited number of conditions with such a symptom complex, as not all diseases with speech and behavioral disorders had a histological picture characteristic of Pick's disease [2,18,33,34].The disease most often begins at 50–60 years of age, but can debut at 20–40 years or in old age, including rare cases at 25 and 89 years [4]. The prevalence among men and women is approximately equal, although some studies note a slightly higher frequency in men [2,6]. The average life expectancy in FTD varies from 6 to 10 years [6]. Progression is faster with greater atrophy in the anterior cingulate cortex, as well as with older age, more pronounced behavioral disorders, and the presence of a known pathogenic mutation for frontotemporal dementia [11]. Carriers of genetic mutations allow for monitoring from the presymptomatic to the symptomatic stage, making them ideal for accurate monitoring of disease progression from the preclinical stage. In contrast, sporadic cases are usually diagnosed years after symptom onset, leading to later stages at the time of identification [16].FTD results from frontotemporal lobar degeneration, leading to atrophy of these brain regions [6]. In 30–50% of cases, the disease is associated with genetic mutations in the MAPT, progranulin, and C9orf72 genes [2].As studies have shown, the genetic factor does not always contribute to the development of frontotemporal dementia, and the relationship between neurohormone levels and the development of this disease and cognitive impairments has not yet been studied.The behavioral variant accounts for more than half of cases of frontotemporal degenerations [20,23,36]. This form of frontotemporal dementia is characterized by a combination of behavioral, cognitive, and affective disorders. The main criteria for diagnosis are gradual onset and steady progression, personality and social behavior disorders, emotional indifference, reduced critical thinking. In most cases, at the onset of the disease, patients do not exhibit any pathological symptoms in the neurological sphere, but with the worsening of the pathological process, patients show grasping reflexes, hypokinesia, pelvic disorders, etc. [4,9,25,37]. The clinical picture of the behavioral variant of frontotemporal dementia is dominated by 6 main signs: disinhibition, apathy, loss of empathy, altered eating behavior, stereotypical behavior, and impaired executive functions. At least three of these must be present for clinical diagnosis [12,38]. The dominant manifestation of the behavioral variant of FTD is social disinhibition, loss of manners, inappropriate behavior, volitional disturbances, apathy, or inertia [6]. According to Fieldhouse, Jay L.P et al., younger patients exhibit more pronounced euphoric, apathetic, and depressive symptoms than older patients with the behavioral form of FTD [16]. Although libido is usually reduced in such patients, they are sexually disinhibited and may engage in sexual acts in public. Such patients also stop taking care of their appearance, do not bathe, do not observe hygiene rules, and wear dirty clothes. Social interests narrow, and patients lose the ability to sympathize, empathize, and feel compassion [6,35]. There may also be simple stereotypical manifestations: rocking, humming, throat clearing, clapping, rubbing, touching something or someone, stomping feet, scratching, smacking, and so on [2,13,36]. Many patients experience changes in eating behavior, possibly overeating, lack of satiety, consumption of inedible items, and a preference for sweet foods [6]. As for cognitive functions in the behavioral form of frontotemporal dementia, they are not fully understood and are still under investigation, but it is known that executive functions are most affected, initiative decreases, planning ability is reduced, and it is difficult for the patient to switch from one cognitive activity to another.Another form of frontotemporal dementia is primary progressive aphasia syndrome. Speech disorders are associated with damage to the temporal lobes of the brain. According to literature sources, primary progressive aphasia is classified into primary progressive aphasia (PPA) without reduced speech fluency (semantic form), PPA with reduced speech fluency (agrammatic form), as well as the logopenic form of PPA. In addition, a combined form of PPA is distinguished, which is characterized by both the semantic form (SF) and the agrammatic form (AF PPA). In the agrammatic form of primary progressive aphasia, the patient's speech becomes laconic, erroneous, and intermittent; grammatical structure is impaired: the patient speaks in disconnected words or phrases. Patients find it difficult to express their thoughts, cannot construct coherent sentences, and counting is impaired. In the semantic form of frontotemporal dementia, the patient does not understand speech addressed to them, does not understand the meaning and significance of words. They often replace some words with others that are similar in meaning. Reading aloud is usually not impaired in such patients. The logopenic form of frontotemporal dementia is similar to Alzheimer's disease, with the patient experiencing difficulty in word finding and struggling to repeat a phrase or sentence.Recently, increasing attention has been paid to studying the influence of various hormones on brain activity, particularly on cognitive functions. The production of some hormones remains at the same level throughout life, with minor deviations, such as cortisol. The production of other hormones changes throughout a person's life, in particular, the production of dehydroepiandrosterone significantly decreases with age. In this regard, scientists are increasingly focusing on studying the influence of hormones on the human body and on cognitive functions, in particular. Various studies have been conducted on the effect of corticosteroids, particularly cortisol, on cognitive activity. Cortisol is a hormone produced in the adrenal cortex from cholesterol under the influence of pituitary adrenocorticotropic hormone, the production of which, in turn, is regulated by hypothalamic corticotropin-releasing hormone [22,24]. Cortisol affects metabolism, blood pressure levels, and is also the main stress hormone. After release from the adrenal cortex, cortisol easily penetrates the blood-brain barrier, binding to specific intracellular receptors in the brain, particularly in areas involved in cognitive functions [27,30]. Short-term exposure to moderate doses of cortisol, for example, during short-term stress, leads to improved memory, while prolonged exposure to high doses of cortisol, for example, during prolonged, chronic stress, leads to impaired memory and executive functions. Girling's studies proved the link between elevated cortisol levels and a decrease in gray matter volume in the brain [31]. Other studies have shown a link between high doses of cortisol and hippocampal atrophy [31,32,42]. Increased cortisol levels lead to impaired episodic memory, spatial memory, information processing speed, and social maladaptation. Patients with Alzheimer's disease also showed elevated levels of the hormone cortisol, and the level of this hormone directly correlated with the level of cognitive impairments and emotional disturbances [15,30,31].Another hormone that affects cognitive functions is dehydroepiandrosterone sulfate, which is produced in the adrenal glands. It is noteworthy that only primates, particularly humans and monkeys, are capable of synthesizing DHEA [3]. A certain part of dehydroepiandrosterone is also formed in the central nervous system. The highest increase in the production of this hormone is observed by the age of 30, after which its production sharply declines. In this regard, the normal range for determining this hormone in the blood of patients also changes depending on age and gender. According to various studies, dehydroepiandrosterone sulfate is known to improve cognitive functions, emotional background, and mood [7]. There is evidence that DHEA can stimulate neurogenesis and protect against neuronal damage, counteracting the neurotoxic effects of glucocorticoids in hippocampal and cortical structures [28,29]. DHEA(S) has neuroprotective, antioxidant, antihypertensive, and anti-inflammatory properties, and also reduces conditioned fear responses in rodents and humans [26,39]. According to a number of authors, the fact that DHEA production decreases with age is more significant in the development of cognitive deficit with age than the decrease in sex hormone production [3,5]. A number of brain diseases accompanied by cognitive impairments, including Alzheimer's disease, are accompanied by a decrease in DHEA levels [3,5]. Impaired neuronal differentiation and a decrease in neuronal contacts between them are also associated with low levels of the hormone DHEA [5].Thus, it becomes clear that studying the effect of neurohormone levels on cognitive functions in frontotemporal dementia opens up wide opportunities for uncovering the mechanisms of this pathology's development and searching for ways to treat and prevent frontotemporal dementia.
2. Materials and Methods of Research
- We examined 114 patients in the age range from 60 to 75 years (elderly age, according to the WHO classification), divided into 3 groups. The first group included 33 (28.9%) patients with frontotemporal dementia: of these, 21 (63.6%) patients had the behavioral variant of frontotemporal dementia (bvFTD), 12 (37.5%) patients had primary progressive aphasia (PPA); the second group consisted of 41 (35.9%) patients with chronic cerebral ischemia of stages II–III with moderate and severe cognitive disorders; the third control group consisted of 40 (35%) patients without cognitive disorders (Figure 1).
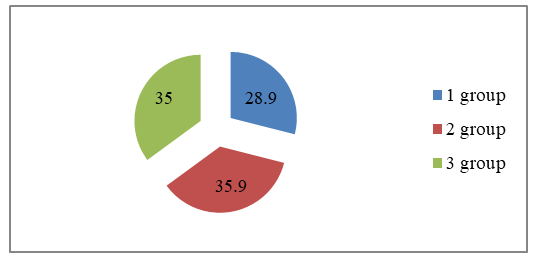 | Figure 1. Distribution of patients by groups (%) |
 | Figure 2 |
3. Research Results and Their Discussion
- Among the clinical manifestations of frontotemporal dementia, the following were most frequently observed, in descending order: memory impairments – in 33 patients (100%), behavioral disorders – in 33 patients (100%), speech disorders – in 25 patients (75.8±7.5%), emotional disorders – in 24 patients (72.7±7.8%), sleep disturbances – in 12 patients (36.4±8.4%), headache – in 11 patients (33.3±8.2%), dizziness – in 8 patients (24.2±7.5%), increased blood pressure was observed in 4 patients (12.1±5.7%). Patients in the second group, in turn, predominantly reported general complaints: memory impairments – in 39 patients (95.1±3.4%), headache – in 39 patients (95.1±3.4%), emotional disorders – in 24 patients (58.5±7.7%), sleep disturbances – in 17 patients (41.5±7.73%), dizziness – in 38 patients (92.7±4.1%), increased blood pressure – in 13 patients (1.7±7.3%). Patients in the second group did not have behavioral or speech disorders. As for patients in the control group, only 1 (2.5±2.5%) patient had a headache, 9 (22.5±6.6%) patients had emotional disorders, 10 (25.0±6.8%) patients complained of sleep disturbances, 6 (15.0±5.6%) patients complained of headache and the same number of increased blood pressure, 4 (10.0±4.7) patients complained of dizziness (Figure 3).
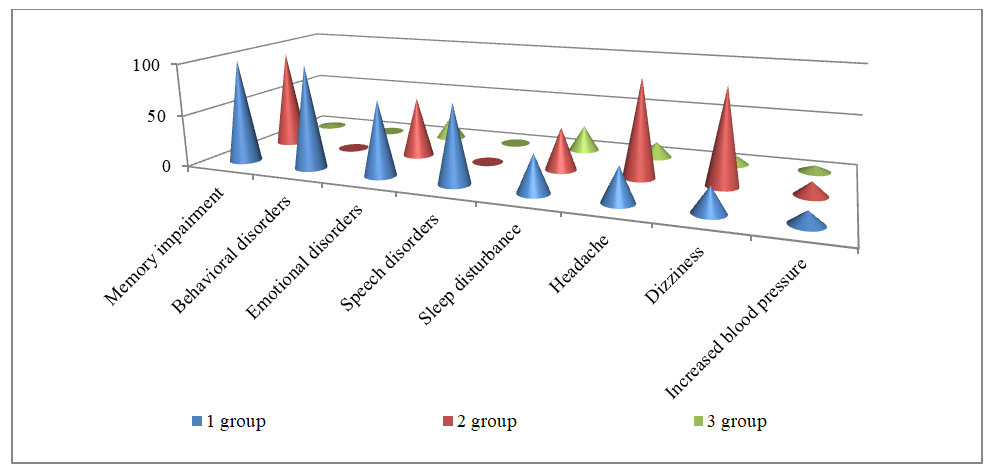 | Figure 3. Main clinical manifestations of patients in all three groups |
 | Figure 4 |
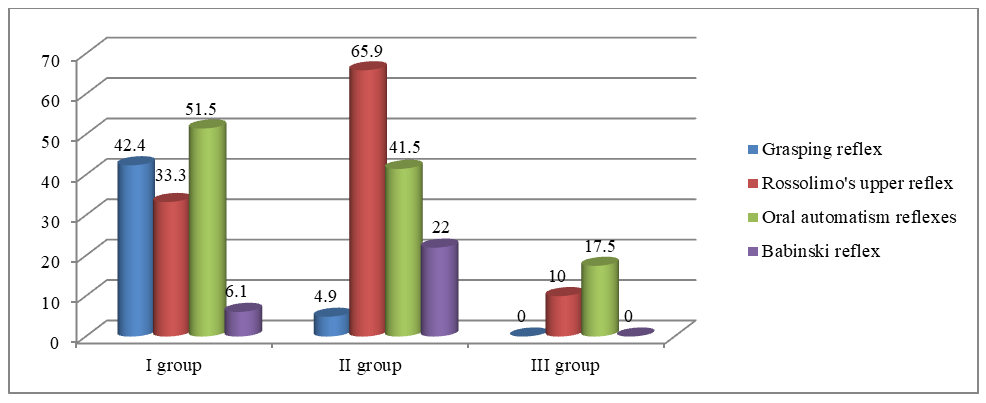 | Figure 5. Frequency of pathological reflexes, % |
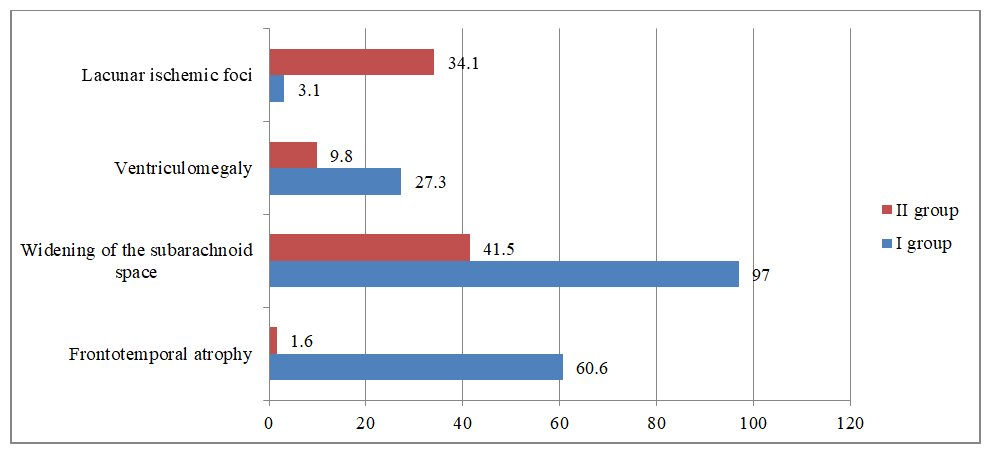 | Figure 6. Frequency of MRI signs in patients, % |
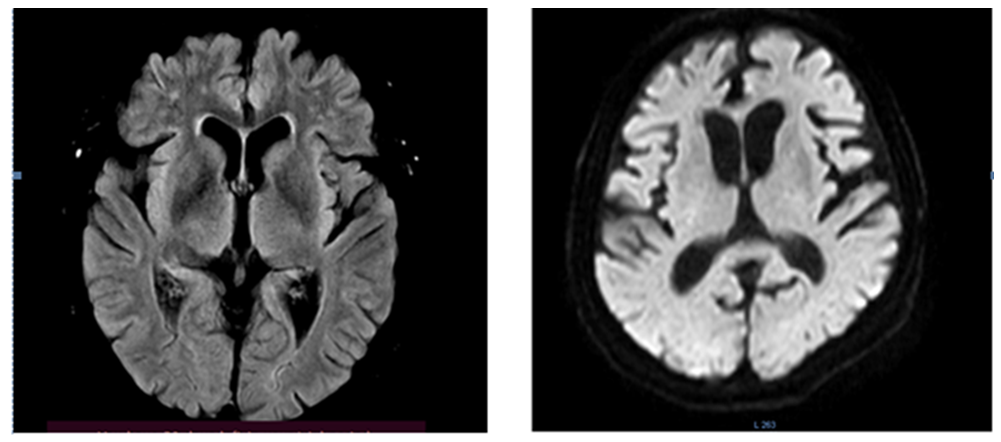 | Figure 7. Patient 67 years old. Atrophy of the frontotemporal regions of the brain |
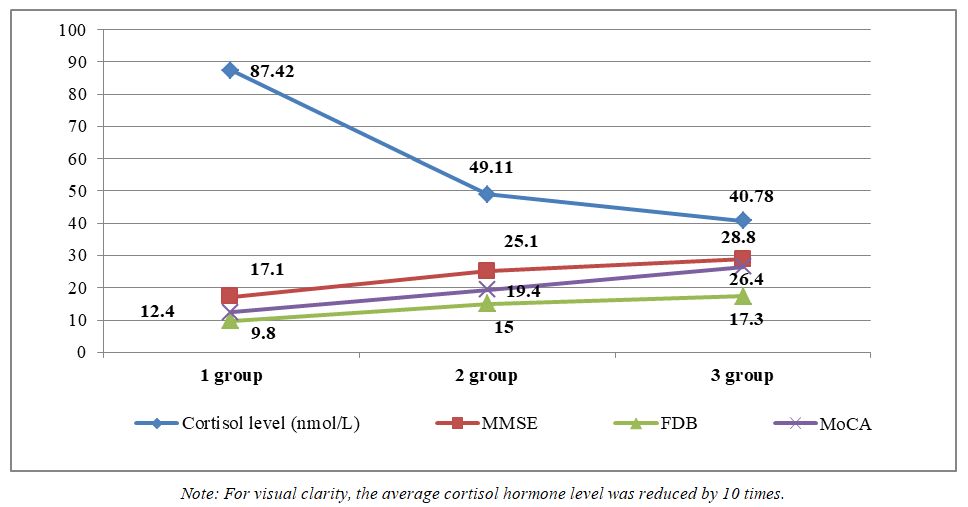 | Figure 8. Correlation between neurocognitive scale levels and cortisol hormone levels |
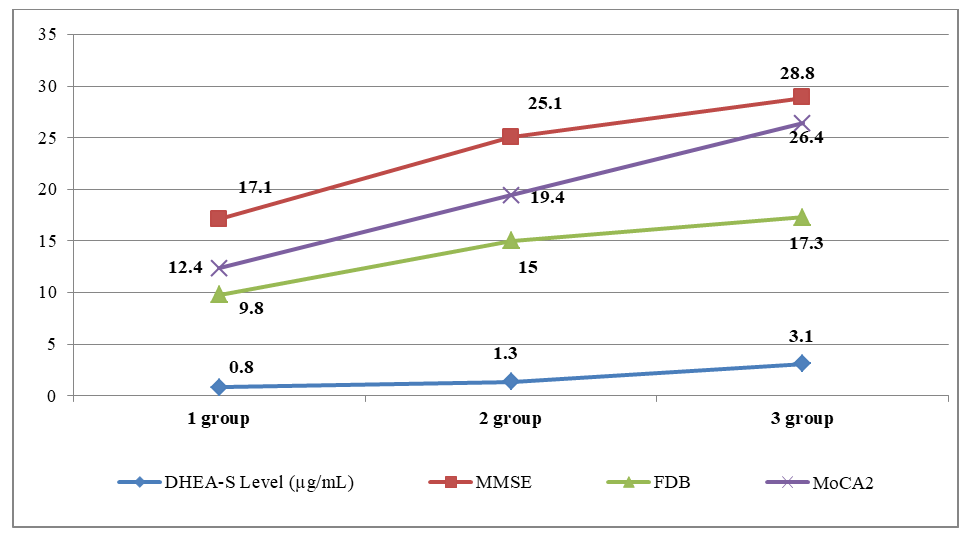 | Figure 9. Correlational relationships between neurocognitive scale levels and cortisol hormone levels |
4. Conclusions
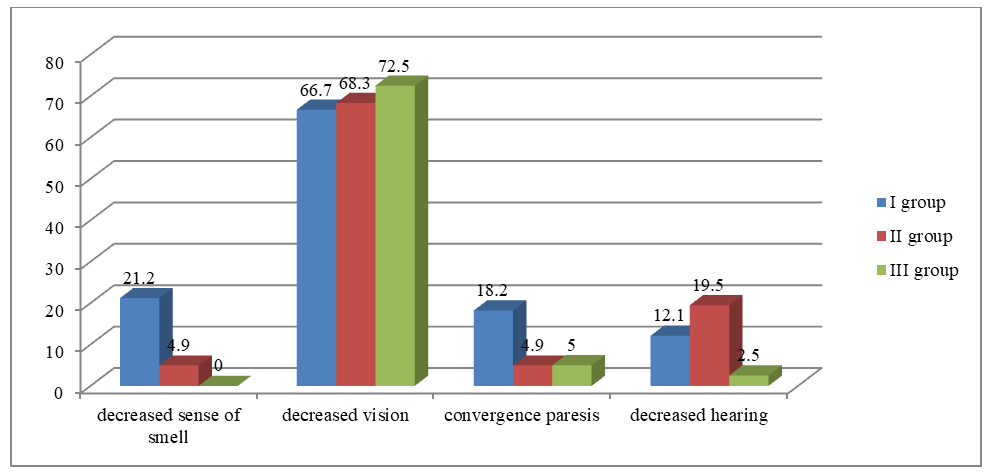
- 1. The results of the study showed that among the clinical manifestations of frontotemporal dementia, memory impairments, behavioral disorders, emotional disorders, and speech disorders come to the forefront.2. In patients with frontotemporal dementia, the olfactory and auditory nerves are most frequently affected. This fact confirms the bilateral involvement of the temporal lobes of the cerebral cortex in the pathological process.3. In patients with frontotemporal dementia, pathological symptoms of oral automatism and the grasping reflex prevailed, indicating the involvement of the frontal lobes of the brain in the process.4. Neuroimaging studies showed that in patients with FTD, atrophy of the frontotemporal regions of the brain prevailed, while in patients of the second group with CIM, lacunar ischemic foci statistically significantly prevailed, which indicated a predominantly vascular etiology of the lesion.5. Correlation analysis between the cognitive level of patients with frontotemporal dementia, patients with CIM, and healthy patients and the level of the hormone cortisol showed an inverse correlation of moderate strength.6. Correlation analysis between the cognitive level of patients with frontotemporal dementia, patients with CIM, and healthy patients and the level of the hormone dehydroepiandrosterone sulfate showed a direct correlation of moderate strength.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML