-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3873-3877
doi:10.5923/j.ajmms.20251511.25
Received: Oct. 9, 2025; Accepted: Oct. 28, 2025; Published: Nov. 8, 2025

The Efficiency of Hypolipidemic Therapy in Combination with Inclisiran in Patients with Atherosclerotic Cardiovascular Diseases Underwent Coronary Artery Angioplasty
N. M. Latipov, B. A. Alyavi
Republican Scientific and Practical Medical Center of Therapy and Medical Rehabilitation, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the study was to evaluate the efficiency and safety of adding inclisiran to standard lipid-lowering therapy in patients with atherosclerotic cardiovascular diseases after coronary artery angioplasty who have not achieved target low-density lipoprotein levels on statin and ezetimibe therapy. Background. Atherosclerotic cardiovascular diseases continue to be the leading cause of death and disability worldwide, including Uzbekistan. One of the keystone elements in the treatment of patients with atherosclerotic cardiovascular diseases is achieving target levels of low-density lipoproteins which is directly associated with a reduction in the risk of recurrent cardiovascular events, including myocardial infarction and cardiovascular death. Material and methods. Clinical trials were conducted at the NeoMedCardio private hospital in 2024-2025. Patients undergoing inpatient treatment were selected for the study. The patients' ages ranged from 42 to 69 years (mean age was 61.4±7.2 years). Results. All three treatment regimens demonstrated a positive effect on the hemodynamic parameters of the brachiocephalic arteries, which was reflected in improved blood flow and reduced vascular resistance. The most significant improvements in all parameters studied were achieved in the group of patients receiving triple therapy with inclisiran, confirming the additional efficiency of this drug in the complex treatment of atherosclerosis. Conclusion. Adding inclisiran to standard therapy with rosuvastatin and ezetimibe provides a more pronounced reduction in low-density lipoproteins, total cholesterol, and triglycerides, as well as an increase in high-density lipoproteins.
Keywords: Atherosclerotic cardiovascular diseases, Low-density lipoproteins, Ischemic heart disease, Inclisiran
Cite this paper: N. M. Latipov, B. A. Alyavi, The Efficiency of Hypolipidemic Therapy in Combination with Inclisiran in Patients with Atherosclerotic Cardiovascular Diseases Underwent Coronary Artery Angioplasty, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3873-3877. doi: 10.5923/j.ajmms.20251511.25.
Article Outline
1. Introduction
- Atherosclerotic cardiovascular diseases (ASCVD) remain the leading cause of death and disability worldwide, including in Uzbekistan. According to WHO data, cardiovascular diseases make up more than 40% of all deaths in low- and middle-income countries. In Uzbekistan, the prevalence of coronary heart disease (CHD), including cases requiring percutaneous coronary intervention (PCI), is steadily increasing, requiring a review of approaches to secondary prevention [1].One of the cornerstones of treating patients with ASCVD is achieving target low-density lipoprotein (LDL) levels, which is directly associated with a reduction in the risk of recurrent cardiovascular events, including myocardial infarction and cardiovascular death. However, despite the widespread use of statins—both in monotherapy and in combination with ezetimibe—a significant proportion of patients do not achieve recommended LDL target values, especially in the postangioplasty period, when the risk of complications remains extremely high. Data from registries and clinical observations confirm that many patients retain a so-called “residual lipid load” that requires additional therapeutic interventions [2].Inclisiran is a representative of a new class of lipid-lowering drugs based on small interfering RNA (siRNA) that suppress PCSK9 expression in the liver. Its long-term action (twice administered per year) and high efficiency in reducing LDL allow it to be considered as a promising agent for the complex therapy of patients with high and very high cardiovascular risk [3]. The efficiency and safety of inclisiran have been confirmed in a number of international clinical trials, including ORION-10 and ORION-11, which showed a significant reduction of LDL in patients with CAD and other forms of ASCVD on standard therapy. In addition, inclisiran is well tolerated and does not require frequent medical monitoring, which is particularly important in settings with limited healthcare resources [4].According to the updated 2025 guidelines from the European Society of Cardiology (ESC) and the European Atherosclerosis Society (EAS), inclisiran is recommended for patients with very high cardiovascular risk, especially when standard therapy is not adequate. Thus, its use becomes a reasonable step in clinical practice aimed at achieving strict lipid targets [5].In spite of the accumulated data on inclisiran in international literature, there is still a lack of local studies in Uzbekistan and Central Asian countries evaluating the efficiency and safety of this drug in real clinical practice, especially in the group of patients who have undergone coronary artery angioplasty. Taking into account the high incidence of cardiovascular disease and the difficulties involved in ensuring long-term monitoring of patients, research into the use of inclisiran as part of comprehensive lipid-lowering therapy is particularly relevant [2].The aim of the study was to evaluate the efficiency and safety of adding inclisiran to standard lipid-lowering therapy in patients with atherosclerotic cardiovascular diseases after coronary artery angioplasty who have not achieved target low-density lipoprotein levels on statin and ezetimibe therapy.
2. Material and Methods
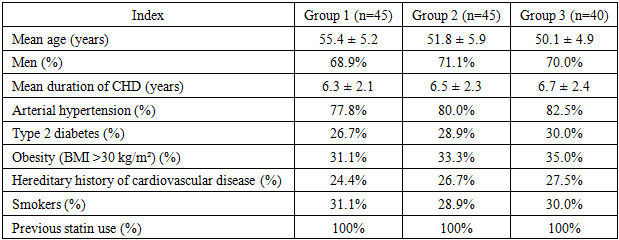
- A total of 130 patients with CHD undergoing treatment in the cardiology department of the “NeoMedCardio” private clinic from 2024 to 2025 were selected for the study. The age of patients ranged from 42 to 69 years (mean age was 61.4±7.2 years). According to the World Health Organization (WHO, 2021) classification, CHD with hypercholesterolemia is most common in middle-aged patients (84.1%). All patients were divided into 3 groups: Group 1 (n=45) consisted of patients received monotherapy with rosuvastatin at a standard dosage (10-20 mg/day); Group 2 (n=45) patients received combination therapy with rosuvastatin and ezetimibe; Group 3 (n=40) consisted of patients received triple therapy: rosuvastatin, ezetimibe, inclisiran. These groups were randomized based on the main clinical and anamnestic data and the therapy received.Inclusion criteria were as follows: • age between 34 and 62 years, • inpatient treatment, • presence of CHD, • hypercholesterolemia. Exclusion criteria: • acute myocardial infarction,• acute cerebrovascular accident,• heart defects,• acute infectious diseases,• hemostatic disorders,• severe concomitant diseases (e.g., cancer or chronic liver/kidney disease),• severe allergic reactions to drugs,• pregnancy and breastfeeding,• severe left ventricular dysfunction (EF below 40%) (Tab. 1).
|
3. Results
- After 12 months of therapy, all groups showed a significant improvement in lipid profile indices compared to baseline (in all cases p < 0.05) (Tab. 2).
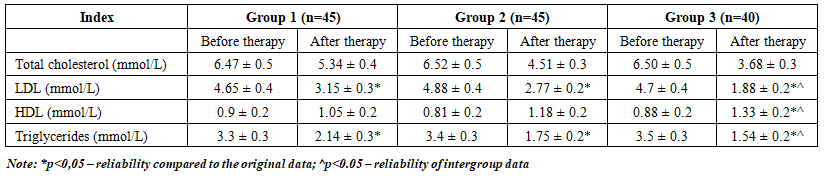 | Table 2. Dynamics of lipid spectrum indices against the background of hypolipidemic therapy |
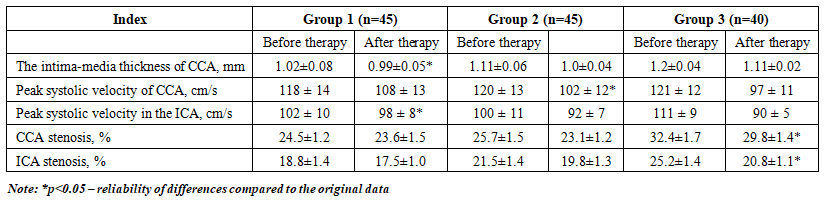 | Table 3. Doppler parameters of brachiocephalic arteries in patients of three groups |
4. Discussion
- The obtained results of our study provide a strong basis for believing that the inclusion of inclisiran in triple lipid-lowering therapy in patients with ASCVD after PCI significantly enhances the lipid-lowering effect, improves lipid spectrum parameters and has a beneficial effect on the state of blood vessels according to Doppler ultrasonography of the brachiocephalic arteries. These findings are consistent with and complement previously published data on inclisiran and mechanistic understanding of the role of PCSK9 inhibition.In the randomized, double-blind ORION-10 and ORION-11 studies, the addition of inclisiran to background therapy resulted in a reduction in LDL-C of approximately 50–52% (time-adjusted effect) compared with placebo [2]. Our results (–60% reduction in LDL in the triple therapy group) exceed the average figures from these trials. It may be due to the features of the population, the background therapy (rosuvastatin + ezetimibe), as well as a longer follow-up period (12 months in our study).In these studies, the safety profile of inclisiran was favorable, and injection reactions were reported more frequently, but were mostly mild. It was noted in ORION-1 that single or double administration of inclisiran resulted in LDL reductions ranging from approximately -28% to -52% by day 180 (depending on the dose) with a favorable safety profile [6].Our data fall within the upper range of the effect, which can be explained by the intensification of statin therapy, optimal dose selection and strict adherence monitoring.In the ORION-5 study (patients with homozygous familial hypercholesterolemia), in patients with extremely high LDL levels, inclisiran, although significantly reduced PCSK9, did not demonstrate a significant reduction of LDL compared to placebo by day 150 [3]. It indicates that in cases of severe receptor metabolism defects of lipoproteins, the efficiency of siRNA therapy may be limited, especially in the absence of functioning LDL receptors.According to Novartis press releases, the open-label extension of ORION-8 confirms the stability of inclisiran's effect with twice-yearly injections for up to 6 years: average reduction of LDL by approximately 49–50%, and approximately 78.4% of patients achieved target levels. These long-term data indicate not only the durability of the effect, but also an acceptable safety profile during prolonged use [7].In a post-clinical analysis of pooled data from ORION-9, ORION-10 and ORION-11, the LDL-lowering effect of inclisiran was shown to be largely independent of diabetes status and body weight (BMI), with reductions ranging from -47.6% to -51.9% across subgroups. It is consistent with the universality of the action mechanism of inclisiran and suggests that our patients with concomitant diabetes did not experience a significant reduction in efficiency.The fact that our Group 3 achieved an average reduction in LD of 60% while ensuring that all patients reached the target level (<1.8 mmol/L) indicates an optimal combination of therapy and strict control of adherence and dosing regimen. It indicates that in real clinical practice, with good organization, it is possible to achieve results that even exceed the average figures of large trials.The increase in HDL (+51.1%) and decrease in triglycerides (-56%) in the triple therapy group is an additional beneficial effect that is often observed in studies of inclisiran (for example, in ORION-10/11 - the decrease in LDL was accompanied by a moderate increase in HDL and a decrease in TG). Improving of these parameters may further reduce the atherogenic potential of the lipid profile.We found that, in addition to positive lipid dynamics, there were significant changes in vascular function parameters: a decrease in intima-media thickness, a reduction in peak blood flow velocities, and a decrease in the degree of stenosis in the brachiocephalic arteries. It suggests a possible restorative or stabilizing effect on the vascular wall. Although there is little data like this in the literature, it adds weight to the idea that aggressive lipid-lowering therapy may, in addition to biochemical changes, lead to structural improvements.A 12-month observation period is sufficient for evaluating the lipid response, but not for estimating the impact on clinical outcomes (heart attacks, strokes, etc.). [8].Our results confirm that inclisiran can be an effective tool for “adapting” therapy in patients with residual hypercholesterolemic risk, especially after PCI, when it is particularly important to rapidly and sustainably reduce LDL. Achieving target LDL levels in all patients in the triple therapy group is an important finding: it demonstrates that with adequate therapy and adherence monitoring, it is theoretically possible to bridge the “therapeutic gap” which is often observed in practice. Improved vascular function suggests that the effect of inclisiran may go beyond simple lipid reduction, possibly contributing to plaque stabilization and vascular wall repair.Thus, discussing our data in the context of global research, we can conclude that the use of inclisiran as part of triple therapy represents a practical and potentially transformative strategy for patients with high residual lipid risk, especially in populations similar to the Uzbek one. This study is one of the first in Uzbekistan, conducted at the “NeoMedCardio” private hospital, which evaluates the efficiency of triple therapy with inclisiran in real-world conditions in patients with ASCVD after angioplasty. Evidence has been presented demonstrating improved adherence to therapy thanks to the convenient administration regimen of inclisiran (twice a year), which is an important factor in the long-term prevention of complications in high-risk patients. Thus, the article contributes to the development of lipid-lowering therapy, confirming the efficiency and safety of inclisiran in combination with traditional drugs and expanding the possibilities of a personalized approach to the treatment of patients with ASCVD in modern clinical practice. The results of this study confirm the advisability of introducing inclisiran into clinical practice in Uzbekistan.
5. Conclusions
- Adding inclisiran to standard therapy with rosuvastatin and ezetimibe provides a more pronounced reduction in LDL, total cholesterol, and triglycerides, as well as an increase in HDL. All patients in Group 3 achieved target LDL levels <1.8 mmol/L.Combined therapy with inclisiran has a positive effect on the structural and functional parameters of blood vessels according to Doppler ultrasound data.The results obtained may contribute to the development of national guidelines for the management of patients with high cardiovascular risk and promote the improvement of secondary prevention in the region.
Conflict of Interests’ Statement
- The authors declare no conflict of interest. This study does not include the involvement of any budgetary, grant or other funds. The article is published for the first time and is part of a scientific work.
ACKNOWLEDGEMENTS
- The authors express their gratitude to the management of the multidisciplinary clinic of “NeoMedCardio” private hospital for the material provided for our study.
Ethical Approval and Consent to Participate
- The Research Ethics Board of our institution does not require review or approval of case reports. Our research was carried out in accordance with the World Medical Association Code of Ethics (Declaration of Helsinki).
Source of Funding
- Each of the authors has reviewed and approved this manuscript. None of the authors has a conflict of interest, financial or otherwise. This manuscript is original, no part of it has been previously published and is not being considered for publication elsewhere. The corresponding author agrees to accept full responsibility for authorship at the submission and review stages of the manuscript.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML