-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3841-3846
doi:10.5923/j.ajmms.20251511.18
Received: Oct. 14, 2025; Accepted: Nov. 2, 2025; Published: Nov. 6, 2025

Correlation Analysis of Hemodynamic Parameters and Renal Artery Morphology: Results of a Comprehensive Assessment Using 320-Row CT
Nigora Djuraeva1, Asalhon Davidhodjaeva2
1Doctor of Medical Sciences, Head of the Department of MSCT and MRI Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov, Tashkent, Uzbekistan
2Physician, Department of Ultrasound Diagnostics Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

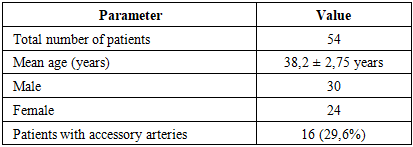
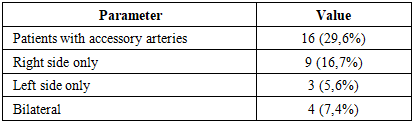
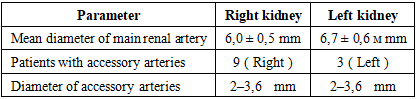
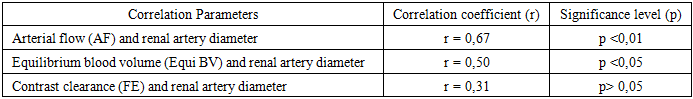
Background: Multidetector computed tomography (MSCT), particularly when performed with high-resolution 320-row systems, has become a valuable imaging modality for the assessment of renal anatomy and function. These techniques allow for precise visualization of renal vasculature and perfusion characteristics, providing critical information for the diagnosis of vascular abnormalities and for preoperative planning in procedures such as nephrectomy and kidney transplantation. Aim: The present study aims to investigate the relationship between morphological characteristics of the renal arteries (diameter and number, including the presence of accessory vessels) and key hemodynamic parameters—arterial flow, equilibrium blood volume, and contrast clearance—derived from 320-row CT perfusion analysis. Methods: A prospective observational study was conducted between June 2021 and March 2024, including 54 potential kidney donors (30 men, 24 women; age range 19–58 years; mean 38.2±27.5 years). All participants underwent 320-row CT, incorporating CT angiography and volumetric CT perfusion (VCTP). Perfusion parameters were analyzed using the single-slope method and the Patlak model. Results: The mean diameter of the right renal artery at the ostium was 6.0±0.5 mm, and that of the left renal artery was 6.7±0.6 mm. Accessory renal arteries were found in 16 patients (29.6%): 9 (16.7%) on the right, 3 (5.6%) on the left, and 4 (7.4%) bilaterally. No significant interrenal differences were observed in perfusion indices. Correlation analysis showed a strong positive relationship between renal artery diameter and arterial flow (r = 0.671, p < 0.01), whereas the number of arteries had no significant effect on blood flow. Conclusion: Renal artery diameter is a key determinant of arterial flow and renal hemodynamics, whereas the number of arteries does not significantly influence perfusion parameters. Nonetheless, the presence of accessory arteries—particularly bilateral—underscores the necessity of detailed preoperative imaging for surgical planning.
Keywords: Multidetector Computed Tomography, Kidneys, Arterial Flow, Blood Volume, Contrast Clearance, Renal Arteries
Cite this paper: Nigora Djuraeva, Asalhon Davidhodjaeva, Correlation Analysis of Hemodynamic Parameters and Renal Artery Morphology: Results of a Comprehensive Assessment Using 320-Row CT, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3841-3846. doi: 10.5923/j.ajmms.20251511.18.
Article Outline
1. Introduction
- The study of renal angioarchitecture and its related hemodynamic parameters is of considerable interest in the context of modern approaches to the diagnosis and treatment of kidney diseases, as well as in the planning of surgical interventions such as nephrectomy and kidney transplantation. Renal arteries play a key role in ensuring adequate perfusion of renal tissue, and disturbances in their structure or function may lead to serious clinical consequences, including ischemic nephropathy, chronic kidney disease, and hypertension [1-3].With the advent of multislice computed tomography (MSCT), particularly high-resolution systems such as 320-row scanners, it has become possible to perform both morphological and functional assessments of the kidneys. This is especially important for early diagnosis of vascular anomalies, evaluation of donor kidneys prior to transplantation, and planning of reconstructive procedures [4,5]. A crucial issue remains the relationship between morphological features of renal arteries—such as vessel diameter and number—and functional indicators, including arterial flow and equilibrium blood volume [6,7].Despite existing data, the literature still contains contradictory aspects concerning the influence of accessory renal arteries on overall renal hemodynamics. Some studies suggest that accessory arteries may play a compensatory role, whereas others indicate their minimal impact on total renal blood flow [8,9]. Furthermore, unresolved questions remain regarding the interaction between morphological and functional kidney characteristics, particularly under conditions of atypical anatomy. Thus, a more in-depth investigation of these relationships using modern imaging methods is required [10].The aim of this study is to determine the relationships between morphological characteristics of renal arteries (diameter, number, presence of accessory vessels) and key hemodynamic parameters such as arterial flow, equilibrium blood volume, and contrast clearance. Addressing this objective will deepen the understanding of the role of renal angioarchitecture in maintaining functional activity and improve approaches to diagnosis and surgical planning, including kidney transplantation.
2. Materials and Methods
- Study DesignThis study was observational, single-center, prospective, selective, and uncontrolled. It did not involve any intervention in patient treatment or care.The study workflow consisted of several stages:1. Selection of patients based on inclusion and exclusion criteria.2. Performing multislice computed tomography (MSCT) using a 320-row scanner.3. Post-processing and analysis of obtained parameters.4. Registration of primary and secondary study outcomes.Eligibility CriteriaInclusion criteria: participants aged 18 to 60 years, absence of comorbidities that could affect renal hemodynamics (such as diabetes mellitus or arterial hypertension), and status as candidates for living-related kidney donation.Non-inclusion criteria: allergy to contrast agents, pregnancy or lactation, and advanced chronic kidney disease (stage 4 or higher).Exclusion criteria: voluntary withdrawal from the study or technical errors during MSCT or data post-processing.Study SettingThe study was conducted at the Republican Specialized Scientific and Practical Medical Center of Surgery named after academician V. Vakhidov, located in Tashkent, Republic of Uzbekistan. It was performed in a specialized department using standard equipment and software.Study DurationThe study took place between June 2021 and March 2024, with a two-year enrollment period. The protocol included control checkpoints, such as baseline MSCT and post-processing within one week. No significant deviations from planned timelines were recorded.Description of Medical ProceduresAll patients underwent comprehensive renal hemodynamic assessment, including CT angiography and volumetric perfusion CT (PCT). MSCT with PCT was performed, and perfusion maps were generated at the workstation to evaluate cortical and medullary blood flow.The examinations were carried out on a 320-row spiral CT scanner (Aquilion One, Canon Medical Systems, Japan) with a 0.5 mm slice thickness in soft-tissue reconstruction mode. The scanning protocol was optimized to minimize radiation dose: tube voltage was 100 kV, exposure 60 mAs, sufficient for dynamic studies with a maximum z-axis coverage of 160 mm. Additional scan parameters included collimator size 0.5 × 320 mm, matrix 512 × 512, field of view 320–350 mm, and tube rotation time 0.275 s.The contrast medium (iodixanol, 350 mg/ml) was administered at 0.5 ml/kg body weight, injection rate 5 ml/s. At an iodine concentration of 350 mg/ml and flow rate of 5 ml/s, the iodine delivery rate (IDR) equaled:IDR = 350 × 5 = 1750 mg iodine/s,meaning patients received 1750 mg of iodine per second.Following contrast administration, 50 ml of 0.9% saline was injected at the same rate. Scanning was performed in volumetric mode, starting 7 seconds after contrast injection. Imaging was acquired every 2 seconds from 12 to 30 seconds, then every 3 seconds from 33 to 48 seconds, and every 10 seconds from 55 to 110 seconds.Data post-processing and perfusion curve analysis were carried out on the Vitrea workstation using the 4D Single Input Perfusion protocol. Tissue density was measured in two regions of interest (ROI): the afferent artery (abdominal aorta) and the target tissue (cortical and medullary layers). Based on time-density curves (TDC), perfusion maps were constructed. Perfusion was evaluated using two mathematical models: the single-slope method and the Patlak model.Blood flow (arterial flow, AF, ml/min/100 ml) was assessed using the single-slope method. Equilibrium blood volume (Equi BV, ml/100 ml) and contrast clearance (FE, ml/min/100 ml) were calculated using the Patlak model (Fig. 1).
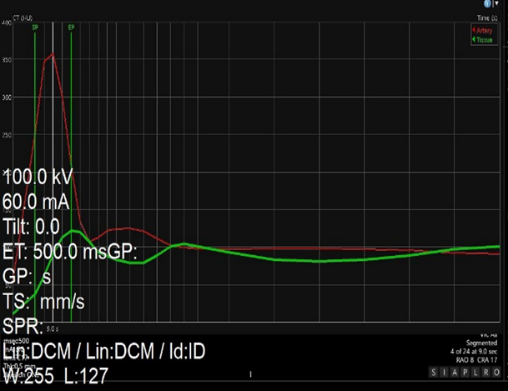 | Figure 1. Time Density Curve (TDC) Plot |
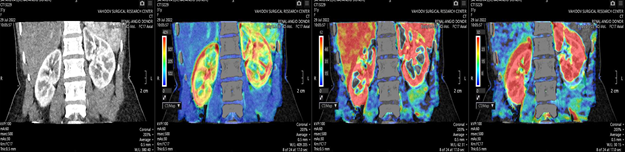 | Figure 2. Renal perfusion maps |
3. Results
- Study PopulationA total of 54 patients who met the inclusion criteria were enrolled in the study. The mean age was 38.2 ± 2.75 years, with 30 males (55.6%) and 24 females (44.4%). Baseline demographic and clinical characteristics are summarized in Table 1.
|
|
|
|
4. Discussion
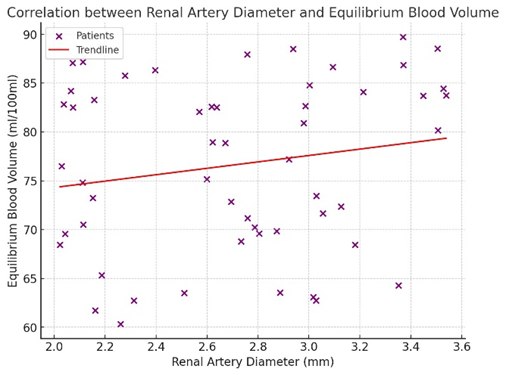
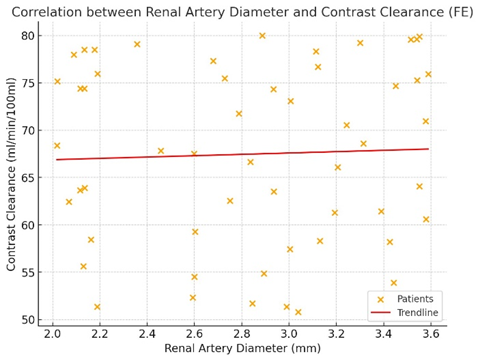
- Summary of Key FindingsThis study demonstrated that renal artery diameter exerts a significant effect on arterial flow (AF), showing a strong positive correlation (r = 0.67, p < 0.01), whereas the number of renal arteries did not significantly influence renal hemodynamics. These findings emphasize the role of renal angioarchitecture in maintaining adequate renal perfusion. Figure 3 illustrates the scatterplot of the correlation between renal artery diameter and AF.
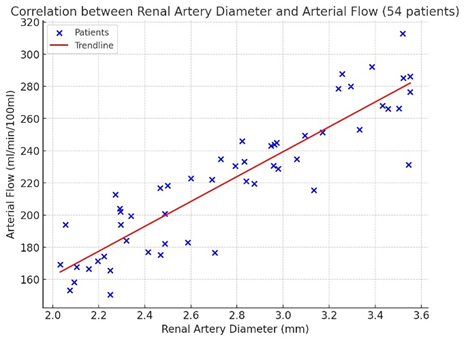 | Figure 3. Scatter plot of correlation analysis data between renal artery diameter (X-axis) and arterial flow -AF (Y-axis) in 54 patients |
|
5. Conclusions
- This study confirms the high diagnostic value of multidetector computed tomography in the evaluation of renal angioarchitecture. While previous research has highlighted the importance of vascular morphology in maintaining renal perfusion, several aspects—such as the role of accessory arteries and their interaction with hemodynamic parameters—remain insufficiently understood.Our findings demonstrate a significant positive correlation between renal artery diameter and arterial flow, underscoring the critical role of vessel caliber in sustaining adequate renal perfusion. In contrast, the number of renal arteries showed only limited influence on hemodynamics, particularly when their diameter did not exceed a threshold value.These results contribute to a deeper understanding of renal perfusion mechanisms and may inform optimization of preoperative planning, especially in the selection of living-related kidney donors. The findings further highlight the need for continued investigation into additional factors—including vascular resistance, parenchymal condition, and systemic influences—that may play a decisive role in regulating renal blood flow.Future research should aim to translate these insights into refined diagnostic criteria and personalized surgical planning strategies, ultimately enhancing outcomes in nephrectomy and kidney transplantation.
Conflict of Interest Statement
- The authors declare no conflicts of interest related to this study.
ACKNOWLEDGEMENTS
- The authors received no specific acknowledgments for this work.
Funding
- No funding was received for the completion of this study.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML