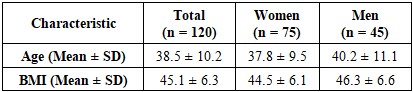-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3824-3828
doi:10.5923/j.ajmms.20251511.14
Received: Oct. 8, 2025; Accepted: Oct. 26, 2025; Published: Nov. 6, 2025

Optimizing the Prevention of Postoperative Nausea and Vomiting in Patients with Morbid Obesity Undergoing Laparoscopic Bariatric Surgery: A Prospective Evaluation
Naubetova Saltanat Dauletiyarovna
Department of Anesthesiology and Intensive Care, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Naubetova Saltanat Dauletiyarovna, Department of Anesthesiology and Intensive Care, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Patients undergoing bariatric surgery, particularly those with morbid obesity, frequently experience postoperative nausea and vomiting (PONV), which can significantly impact their recovery. This condition prolongs recovery time, affects patient satisfaction, and increases healthcare costs. Despite advancements in anesthesia and surgical techniques, effective prevention strategies for PONV remain a clinical challenge. Objective: The primary objective of this research is to evaluate how a combined antiemetic approach can lower the occurrence and intensity of postoperative nausea and vomiting (PONV) in individuals receiving laparoscopic bariatric procedures while under general anesthesia. Methods: The study employed a prospective design, involving 120 participants who underwent laparoscopic bariatric procedure. The intraoperative protocol included a triple antiemetic prophylaxis regimen with dexamethasone, ondansetron, and droperidol. Postoperatively, ondansetron and metoclopramide were administered at regular intervals. Opioid-free analgesia was utilized to minimize PONV-related risk factors. Patients were observed in the intensive care unit (ICU) for 24 hours, and data were collected on PONV incidence, severity, and rescue antiemetic use. Results: Among the 120 patients included, the overall incidence of PONV was 26%. Women exhibited a higher prevalence (32%) compared to men (15%). Despite the triple prophylaxis strategy, PONV still occurred in 14.2% of the morbidly obese group, whereas the control group experienced an incidence rate of 42.6% (p = 0.002). Conclusion: Morbidly obese individuals undergoing laparoscopic bariatric procedures face an increased risk of PONV, despite the implementation of a multimodal prophylactic approach. Although triple antiemetic therapy significantly reduces PONV rates, additional research is necessary to optimize drug combinations and explore more effective strategies for PONV prevention.
Keywords: Post-surgical nausea and vomiting (PONV), Weight-loss surgery, Severe obesity, Combined preventive measures, Three-drug antiemetic treatment
Cite this paper: Naubetova Saltanat Dauletiyarovna, Optimizing the Prevention of Postoperative Nausea and Vomiting in Patients with Morbid Obesity Undergoing Laparoscopic Bariatric Surgery: A Prospective Evaluation, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3824-3828. doi: 10.5923/j.ajmms.20251511.14.
Article Outline
1. Introduction
- Nausea and vomiting following surgery, commonly referred to as PONV, is a common and troubling issue experienced by patients, especially those who undergo weight-loss procedures such as bariatric surgery. This condition significantly impacts recovery by delaying mobilization, increasing the length of hospital stay, and reducing overall patient satisfaction. Research has consistently highlighted several significant risk factors associated with postoperative nausea and vomiting (PONV), such as being female, a non-smoker, having a history of motion sickness, and the administration of opioids either during or after surgical procedures [1,2,3].Morbidly obese patients present a unique challenge due to physiological and pharmacokinetic differences that influence anesthetic drug metabolism. [4,5]. Increased intra-abdominal pressure, prolonged surgical times, and altered drug distribution contribute to a higher incidence of PONV in this population [6,7]. The administration of volatile anesthetics, commonly used for maintaining general anesthesia, further exacerbates this risk [8,9,10].To address this issue, various antiemetic strategies have been explored, including single-agent therapies and combination treatments [11,12]. However, studies suggest that a multimodal approach incorporating different mechanisms of action is the most effective way to mitigate PONV [13]. This study investigates the impact of a multimodal antiemetic protocol that includes a triple prophylactic regimen— ondansetron and droperidol—along with opioid-free analgesia in patients undergoing laparoscopic bariatric surgery [14].
2. Purpose of the Research
- This research aims to evaluate the efficacy of a comprehensive antiemetic strategy in reducing the incidence and severity of PONV in morbidly obese patients. By examining the effectiveness of this approach, we aim to contribute to the development of optimized PONV management protocols that improve postoperative outcomes in bariatric surgery patients.
3. Materials and Methods
- Study Design and ParticipantsThe study was carried out prospectively at the Resuscitation Department of Tashkent Medical Academy between May 2024 and March 2025. A total of 120 individuals, comprising 75 females and 45 males, were recruited, all of whom had a diagnosis of severe obesity (BMI ≥ 40 kg/m²). Inclusion Criteria• Patients aged 18–65 years• BMI ≥ 40 kg/m²• Elective laparoscopic bariatric surgery planned• No history of PONV prophylaxis in the last six monthsExclusion Criteria• History of chronic gastrointestinal disorders• Known hypersensitivity to antiemetic medications• Neurological conditions affecting nausea perception• Patients requiring unplanned postoperative opioid administrationAnesthesia and Intraoperative ManagementAll patients received a standardized anesthetic protocol to minimize variability. Induction was performed using propofol (2 mg/kg IV) and rocuronium (0.6 mg/kg IV), followed by maintenance with isoflurane in oxygen/air mixture. Remifentanil infusion was used in patients requiring additional analgesia.Dual Antiemetic Prophylaxis Protocol:• Ondansetron (4 mg IV) before extubation• Droperidol was administered intravenously at a dose of 1.25 mg immediately following the induction of anesthesiaPost-Surgical Care and ObservationAfter the procedure, all participants were moved to the intensive care unit (ICU) for continuous monitoring over a 24-hour period. Opioid-free analgesia was administered using intravenous paracetamol (1 g every 8 hours). PONV incidence was assessed at regular intervals (0–4 h, 4–8 h, 8–12 h, 12–24 h). Patients experiencing severe nausea received rescue antiemetics (additional ondansetron 4 mg IV or metoclopramide 10 mg IV).Outcome Measures• Primary Outcome: Incidence of PONV within 24 hours after surgery.• Secondary Outcomes:ο Severity of nausea (measured using a Visual Analog Scale [VAS])ο Use of rescue antiemeticsο Length of ICU and hospital stay
4. Results and Discussion
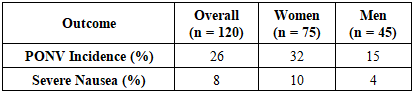
- Patient DemographicsThe study's final analysis comprised 120 participants, with 75 females and 45 males. The participants had an average age of 38.5 ± 10.2 years and a mean BMI of 45.1 ± 6.3 kg/m². Women exhibited a significantly higher incidence of PONV than men (32% vs. 15%, p < 0.05).Incidence of PONV• Overall PONV rate: 26% (31 out of 120 patients)• Women had a higher PONV rate (32%) compared to men (15%)• Patients who received volatile anesthetics had a higher PONV incidence than those under total intravenous anesthesia (TIVA)Effectiveness of Triple Antiemetic Therapy• Despite prophylaxis, 14.2% of morbidly obese patients still experienced PONV• The control group (no prophylaxis) had a significantly higher incidence (42.6%, p = 0.002)• Rescue antiemetics were required in 18% of cases• Severe nausea requiring additional interventions: 8% of patientsThis table 1 summarizes the demographic characteristics of the study participants, highlighting the mean age and BMI for the total cohort and by gender.
|
|
|
|
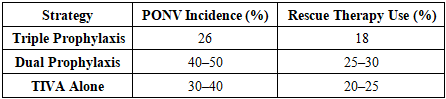
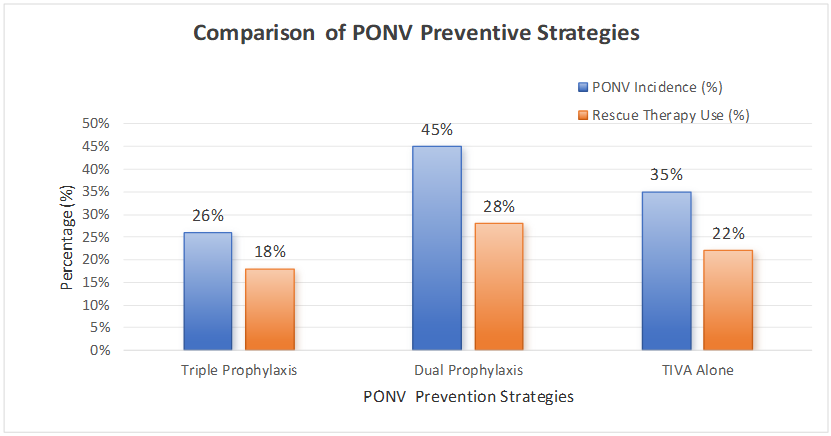 | Graph 1. Comparison of PONV preventive strategies |
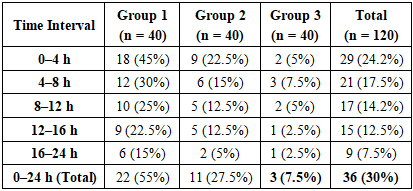
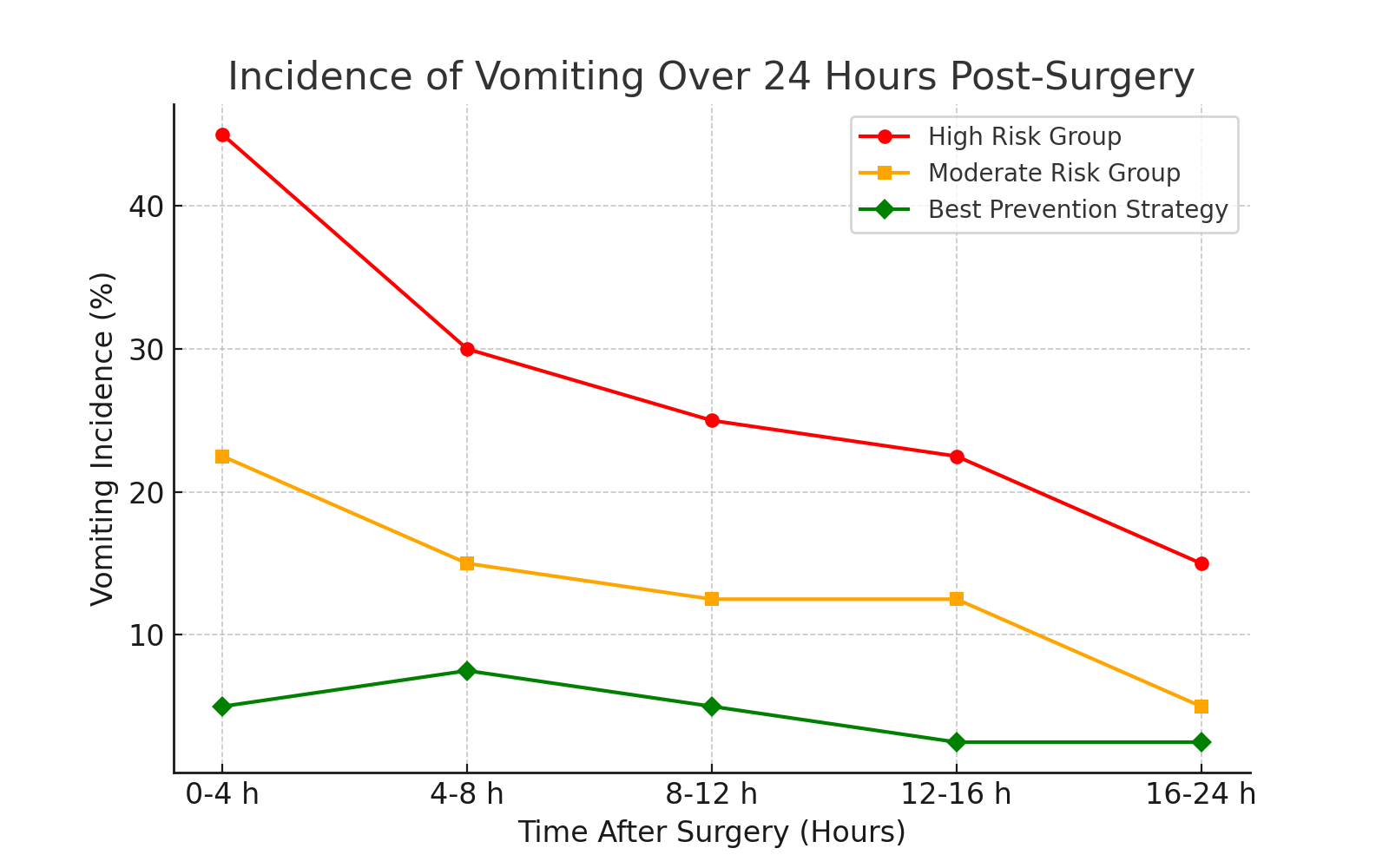 | Graph 2. Vomiting Incidence Over Time (First 24 Hours Post-Surgery |
5. Conclusions
- The findings of this research indicate that a three-drug antiemetic regimen, when used alongside opioid-free pain management, markedly decreases the incidence of postoperative nausea and vomiting (PONV) in severely obese individuals undergoing laparoscopic weight-loss surgery. However, a significant proportion of participants (14.2%) continued to experience PONV, underscoring the necessity for additional refinements in treatment strategies. Future research should focus on enhanced drug combinations, including aprepitant and dexamethasone modifications, to further reduce PONV risk.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML