-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3807-3811
doi:10.5923/j.ajmms.20251511.11
Received: Oct. 1, 2025; Accepted: Oct. 25, 2025; Published: Nov. 6, 2025

Subacute Sclerosing Panencephalitis in Children: Uzbekistan Current Situation After COVID-19
Shaanvar Shamansurov1, Saidazizova Shakhlo1, Firdavs Inomov1, 2, Nodirahon Malikovna1
1Department of Child Neurology, Center for the Development of Professional Qualification of Medical Workers, Tashkent, Uzbekistan
2Department of Pediatric Neurology, National Children’s Medical Center, Tashkent, Uzbekistan
Correspondence to: Firdavs Inomov, Department of Child Neurology, Center for the Development of Professional Qualification of Medical Workers, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Subacute sclerosing panencephalitis (SSPE) is a deadly form of encephalitis that occurs after measles in children. Ways to: We conducted a retrospective analysis of 188 pediatric cases utilizing clinical, EEG, MRI, and serological data. Results: In the early stages of the disease, seizures and problems with gait and coordination were the most common symptoms. Now, speech problems (74.5%), cognitive decline (64.9%), coordination problems (63.3%), and seizures (61.7%) are the most common neurological problems. Mental and autonomic disorders are less common. EEG usually showed periodic complexes, and early MRI was often normal. Over time, it showed changes in the white matter and atrophy in the cortex and subcortex. The main treatments were isoprinosine, interferon alfa-2b, ribavirin, and IVIG. Many people had side effects, and 8.5% (16/188) of those who died. Conclusions: A thorough multimodal assessment facilitates prompt diagnosis, organized seizure management, and uniform immuno-antiviral protocols, with measles vaccination remaining the paramount preventive strategy.
Keywords: SSPE, EEG, MRI, Children, Seizures, Myoclonus, Immunotherapy
Cite this paper: Shaanvar Shamansurov, Saidazizova Shakhlo, Firdavs Inomov, Nodirahon Malikovna, Subacute Sclerosing Panencephalitis in Children: Uzbekistan Current Situation After COVID-19, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3807-3811. doi: 10.5923/j.ajmms.20251511.11.
Article Outline
1. Introduction
- Subacute sclerosing panencephalitis (SSPE) is a rare, progressive neurological disorder primarily affecting children and young adults, resulting from a persistent measles virus infection. Neurophysiologically, SSPE is characterized by distinct electroencephalogram (EEG) patterns, including periodic high voltage discharges and slow wave complexes, which are crucial for diagnosis. These EEG features, such as long interval periodic complexes and burst suppression patterns, are observed in a significant number of patients, although they may not be present at all stages of the disease [1], [2], [3]. Radiologically, magnetic resonance imaging (MRI) often reveals abnormalities such as hyperintensities in the frontoparietal region, diffuse cerebral atrophy, and periventricular white matter changes, although some cases may initially present with normal imaging [1], [4], [5]. The clinical presentation of SSPE includes cognitive decline, myoclonic seizures, and motor impairments, with atypical presentations such as akinetic rigid syndrome and acute disseminated encephalomyelitis-like symptoms also reported [1]. The disease progression is marked by rapid neurocognitive decline, leading to severe disability or death, often within a few years of diagnosis [4]. Despite vaccination efforts, SSPE remains prevalent in regions with low immunization rates, underscoring the importance of vaccination in preventing this fatal complication [2], [6]. The prognosis of SSPE is generally poor, with many patients becoming bedridden or entering a vegetative state, highlighting the need for increased awareness and early diagnosis to manage the disease effectively [4].
2. Materials and Methods
- Over the past 3 years, we have conducted a retrospective study of 188 patients with PE at the Center for the development of professional qualification of medical workers and the Children's National Medical Center. The following clinical neurological, EEG, MRI, serological (measles virus IgM and IgG levels in the blood) and cerebrospinal fluid studies were conducted and evaluated in patients. The obtained results were analyzed and refined in Excel 2016 software. All inspections were carried out with the permission of parents and the decision of the National Center (PK-451g) and without challenging the consortium. Thorough physical and systemic examinations were conducted, including a detailed nervous system examination.Specific investigations were performed, including routine examination of cerebrospinal fluid (CSF) and serum, as well as testing for measles antibodies in both CSF and serum. The diagnosis of SSPE was supported by a combination of clinical features and laboratory findings, such as compatible EEG results and neuroimaging findings, although brain biopsy was not performed due to practical constraints.
3. Results
- Since 2024, 188 cases have been identified through observation. The number of sick children in relation to their birth year: in 2016 - 1; in 2017 - 1; in 2018 - 13; in 2019 - 153; in 2020 - 1; in 2023 - 8 new cases were identified. By gender: 137 boys (77.4%), 40 girls (22.6%); regarding measles vaccination: 165 patients were infected with the measles virus before reaching vaccination age, 12 patients were infected with the measles virus after vaccination. During the period of measles infection, the disease was mainly severe, and these patients were primarily 4-8 months old. Sick children were generally vaccinated against measles at the age of 1 year. 9 (5%) patients were infected with the COVID-19 virus. A measles antibody test was conducted on the mothers of sick children, which detected active immunity to the measles virus in all mothers. Mothers were not infected with the measles virus during pregnancy. Among sick children, 15 (8.5%) were not breastfed, while the remaining 91.5% (162) were breastfed until the age of 2 years. 104 (58.7%) of the sick children attended kindergarten. Mothers were not infected with the measles virus during pregnancy. Of the 188 patients, 16 died from SSPE, which means that 8.5% of the cases died during the study period.Among the parents, 7 families had marriages between close relatives. In sick children, the first signs of the disease began to appear 3-3.5 years after measles infection. In patients, the main clinical manifestations initially included changes in speech, mental acuity, and visibility of foreign objects or shapes. Later, seizures appeared, mainly in the form of atonic and myoclonic seizures. Motor retardation began to appear 4-6 months after the onset of the disease.In the diagnosis of patients, based primarily on the Dyken criteria, cerebrospinal fluid and blood plasma samples were taken from 106 patients and tested for measles virus IgG and IgM (through ELISA). In all of them, the ratio of IgG and IgM in the cerebrospinal fluid was higher than 1:4 and in blood plasma was higher than 1:256. The remaining 73 patients were diagnosed based on anamnesis data, the presence of signs characteristic of SSPE (initially Radermecker complex) on EEG (Figure 1.), and the clinical course.
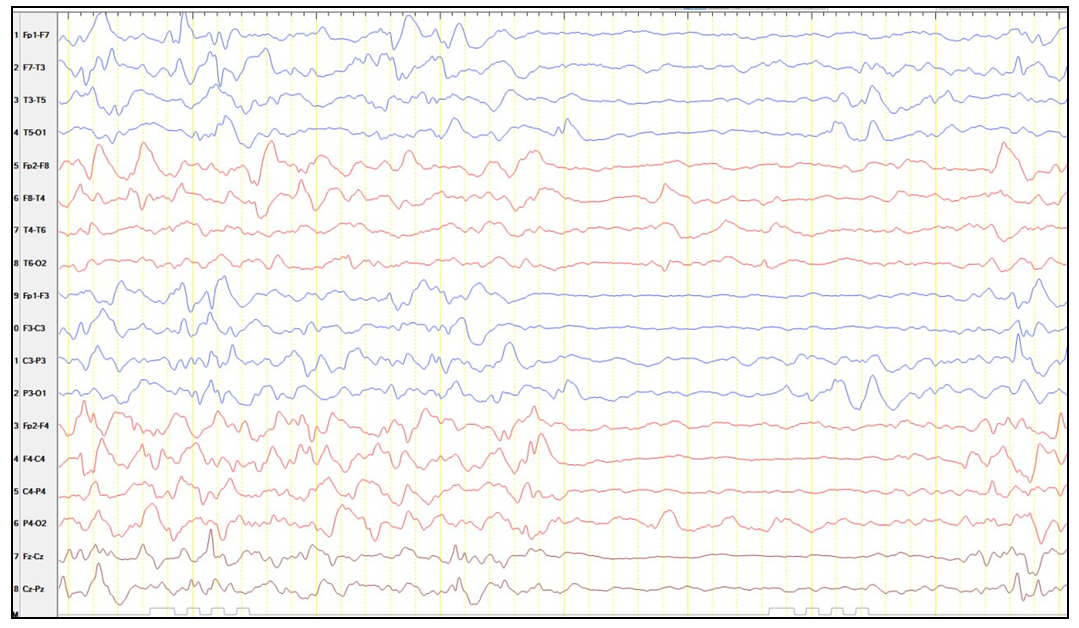 | Figure 1. EEG characteristics of one the patients |
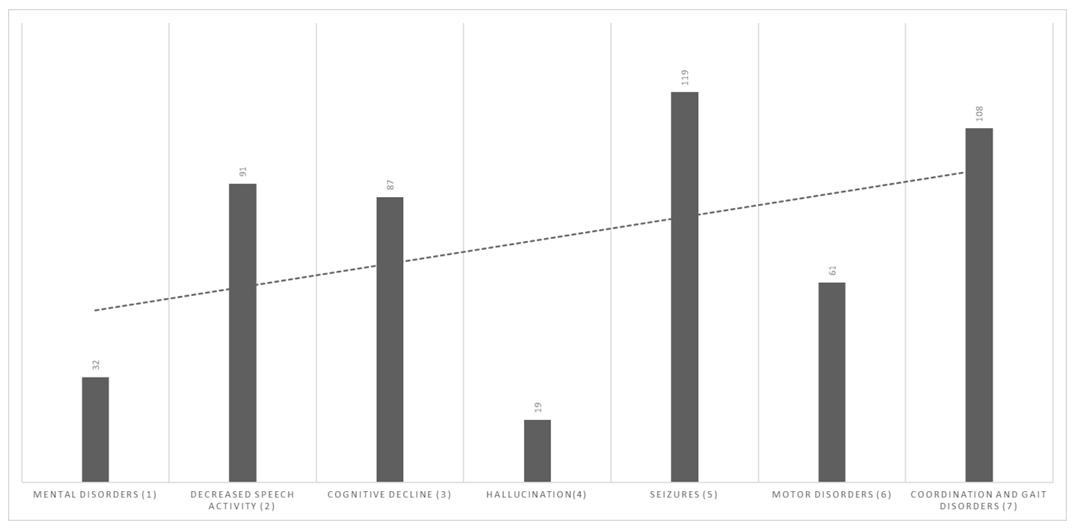 | Figure 2. Early signs of the disease |
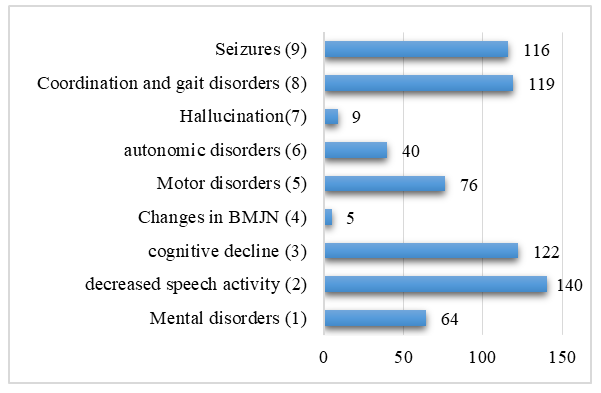 | Figure 3. Current neurological changes |
4. Discussion
4.1. EEG Pattern Classification
- 44 studies(out of 350 relevant articles) reported typical periodic complexes as the predominant EEG pattern in pediatric SSPE, with atypical or normal EEG patterns less frequently observed, highlighting the diagnostic importance of periodic complexes [3], [7], [8].Several case reports and series documented atypical EEG features including burst suppression, focal epileptiform discharges, and sharp wave activity, often associated with atypical clinical presentations or disease progression [7], [9], [10].A few studies emphasized the diagnostic value of EEG parameter optimization and benzodiazepine administration to elicit typical periodic complexes in initially atypical or normal EEGs [11], [12].
4.2. Neural Connectivity Measures
- Only a limited number of studies employed advanced neurophysiological techniques to assess neural connectivity, notably one study integrating MEG and EEG source localization to identify thalamic and cortical generators of periodic complexes [13].The majority of studies relied on conventional EEG and MRI without quantitative connectivity analyses, indicating a significant gap in understanding functional and effective connectivity alterations in SSPE.Neuroimaging findings were commonly reported but rarely integrated with EEG connectivity data, limiting comprehensive network-level insights (Panda et al., 2024) (Praveen-Kumar et al., 2015).
4.3. Clinical Correlation Index
- 30 studies found moderate to strong correlations between neuroimaging abnormalities and clinical disease stage or severity, whereas EEG abnormalities showed variable or weaker correlations with clinical parameters (Panda et al., 2024) (Kundu & Hossain, 2021) (Wasim et al., n.d.).Several reports highlighted the association of EEG periodic complexes with myoclonus and clinical progression, supporting EEG as a useful biomarker for disease monitoring [2], [13], [14].Some studies noted that atypical EEG patterns or normal EEGs could delay diagnosis, underscoring the need for clinical vigilance and complementary diagnostic tools [7], [15].
4.4. Longitudinal EEG Dynamics
- 10 studies provided longitudinal EEG data demonstrating evolution from atypical or normal patterns to typical periodic complexes as SSPE progressed, reflecting dynamic neurophysiological changes [2], [16], [17].Disease duration was positively correlated with the emergence of typical periodic delta wave complexes, indicating EEG changes as markers of disease stage [8].Few studies tracked EEG changes over extended periods or in response to interventions, revealing a need for more longitudinal research [16].
4.5. Methodological Rigor
- Study designs varied widely, including single case reports, small case series, retrospective cohorts, and a few prospective studies, with sample sizes ranging from single patients to over 100 [18], [19].Advanced analytical techniques such as MEG-EEG source localization were rare, with most studies relying on descriptive EEG and MRI assessments [13].Several studies lacked longitudinal follow-up or standardized protocols, limiting generalizability and comprehensive understanding of EEG and connectivity changes in SSPE [3], [20].The literature on EEG patterns and neural connectivity in children with subacute sclerosing panencephalitis (SSPE) reveals a comprehensive yet heterogeneous body of research. Studies consistently identify characteristic EEG features such as periodic complexes, but there is notable variability in the presentation and progression of these patterns. Neural connectivity investigations, particularly those employing combined MEG-EEG techniques, provide novel insights into the pathophysiology of SSPE, although such studies remain limited in scale. Correlations between EEG abnormalities, clinical severity, and neuroimaging findings are inconsistently reported, reflecting methodological differences and sample size constraints. Furthermore, the diagnostic utility of EEG is emphasized, yet atypical presentations and evolving EEG patterns pose challenges for early and accurate diagnosis. Overall, while the research advances understanding of SSPE neurophysiology, methodological limitations and variability in findings highlight the need for standardized approaches and larger, longitudinal studies.
5. Conclusions
- In this cohort of 188 patients, SSPE in children initially manifested with seizures and coordination/gait difficulties, subsequently advancing to significant speech reduction and cognitive decline. EEG periodic complexes were very helpful, but MRI was often normal at first and then showed cortical-subcortical atrophy and white-matter involvement. Management typically involved a combination of isoprinosine, interferon alfa-2b, ribavirin, and IVIG; however, adverse effects were common, and the overall mortality rate persisted at 8.5%. These results underscore the necessity for prompt identification through integrated clinical, EEG, and serological criteria, anticipatory seizure management, meticulous surveillance of drug toxicity, and uniform treatment protocols. Strengthening measles immunization is still the best way to stop this disease from spreading.
ACKNOWLEDGEMENTS
- Center for the Development of Professional Qualification of Medical Workers and the National Children’s Medical Center for providing archival information and technical support.
DECLORATION
- Conflict of interest: The authors declare no conflict of interest.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML