-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3803-3806
doi:10.5923/j.ajmms.20251511.10
Received: Sep. 22, 2025; Accepted: Oct. 10, 2025; Published: Nov. 6, 2025

Pathogenetic and Epigenetic Aspects of the Co-Occurrence of Rheumatoid Arthritis and Liver Diseases
Fazilat Shukurova1, Marif Karimov2, Aida Eshmurzayeva3, Marina Sibirkina4, Noila Khudayberganova4, Dilorom Abzalova5
1DSc, Associate Professor, Tаshkent State Medicаl University, Tashkent, Uzbekistan
2DSc, Professor, Head of Depаrtment Propaedeutics of Internal Diseases, Tаshkent State Medicаl University, Tashkent, Uzbekistan
3DSc, Professor, Tаshkent State Medicаl University, Tashkent, Uzbekistan
4PhD, Associate Professor, Tаshkent State Medicаl University, Tashkent, Uzbekistan
5PhD, Assistant, Tаshkent State Medicаl University, Tashkent, Uzbekistan
Correspondence to: Fazilat Shukurova, DSc, Associate Professor, Tаshkent State Medicаl University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Purpose: to determine the pathogenetic aspects expression-rate of miRNAs 122 and 221 in the serum of sufferers with rheumatoid arthritis (RA) as serum biomarkers associated with the causes of liver damage in this type of patients. Methods: MicroRNAs 122 and 221 (miRNA122/221) were isolated from peripheral blood samples of a total of 148 patients diagnosed with RA (17 patients with RA and chronic viral hepatitis B and C (СHB and C) in the 1st subgroup, 45 patients with RA and non-alcoholic fatty disease liver (NAFLD) in the 2nd subgroup and in the 3rd subgroup, 86 patients were diagnosed with RA without primary concomitant liver diseases), and their expression levels were determined using real-time reverse transcription polymerase chain reaction (PCR). Aspects of the results related to the pathogenesis of pathological conditions were then analyzed and their correlation with other signs of liver function was established. Results: The expression level of miRNA122 in sufferers with diagnosis RA and chronic hepatitis was significantly lower compared to patients in groups 2 and 3, and the expression level of miRNA221, on the contrary, was higher than in patients of other groups (p = 0,005). In addition, the expression degree of miRNA221 in patients with both RA and NAFLD was slightly higher than in patients of the three groups, and the difference was statistically significant (p = 0,005). The degrees of miR 221 were found correlated with fibrotic changes in liver tissue, and the expression-rate of miR 122 were found to significantly correlate with ALT (p < 0,005) and AST (p < 0,001). Conclusions: From the research results, we can conclude that complex correlations of microRNA 122/221 expression with RA and diffuse liver diseases have been identified, which may be a promising marker of RA and liver pathologies.
Keywords: Rheumatoid arthritis, Chronic diffuse liver diseases, microRNA 122, microRNA 221
Cite this paper: Fazilat Shukurova, Marif Karimov, Aida Eshmurzayeva, Marina Sibirkina, Noila Khudayberganova, Dilorom Abzalova, Pathogenetic and Epigenetic Aspects of the Co-Occurrence of Rheumatoid Arthritis and Liver Diseases, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3803-3806. doi: 10.5923/j.ajmms.20251511.10.
1. Introduction
- Increasing research in the field of molecular biology is leading to the emergence of new approaches, such as the study of microRNAs. When comparing microRNAs with proteins, it was found that they are directly and more closely related to cellular genomic activity as biomarkers. This forces us to consider biomarkers as an additional effective method to the generally accepted methods (histological analysis) for clarifying and verifying the clinical diagnosis. The use of epigenetic methods in rheumatoid arthritis (RA), in particular the analysis of microRNA expression levels as biomarkers for early non-invasive diagnosis and prediction of RA, allows early diagnosis of the severe clinical manifestations and complications that occur in these patients. This, in turn, reduces the rate of disability and death from complications in this type of patients.Therefore, the purpose of this study was focused on conducting a comparative analysis of serum expression-rate of disengaged microRNAs in patients with RA diagnosis and chronic diffuse diseases of the liver (CDLD), and to evaluate their diagnostic and prognostic significance.Epigenetic regulation in rheumatoid arthritis and chronic liver diseaseImproving the efficiency of early diagnosis of complex concomitant pathologies such as RA and CDLD requires the implementation of highly sensitive and specific biomarkers [1]. Evaluation of the development, clinical course and complications of these diseases also depends to a large extent on the study of CDLD and RA biomarkers [2]. Biomarkers are also important in developing individualized treatment plans. In the last two decades, which we can call the new era of molecular diagnostics, great progress has been made in the study of interactions between genes, their products and environmental factors. A special feature of epigenetic regulation is that with its help, changes in gene expression occur without damaging the DNA structure, and this change can be stably transmitted over several generations. An important area of epigenetics is the study of epigenome properties in different tissues of the body under normal and pathological conditions.Recent studies indicate an important role of microRNAs in RA, and dysregulated microRNA expression has been hypothesized to contribute to the molecular mechanisms of the disease. In this direction, based on the evidence accumulated in recent years, microRNAs can be abnormally expressed both in the inflamed synovial membranes and in the circulating blood of RA patients [3], and the significance of microRNAs in RA patients may meet the criteria for their use as new molecular diagnostic markers [4].Aberrant expression of several microRNAs in different cell types has already been identified in RA, and these microRNAs may regulate specific pathways leading to the inflammatory environment that occurs in RA [5].According to research results, it can be said that the decrease of miR-221 expression is conversely proportional to the level of circulating anti-inflammatory cytokines [6]. Increased expression of miR-221 in RA leads to an increase in VEGF (vascular endothelial growth factor), MMP-1 and MMP-3 (Matrix metalloproteinases), which are mediators of angiogenesis and inflammation [6,7]. In addition, overexpression of miR-221 in RA can enhance the activation of synovial fibroblasts and increase their resistance to apoptosis [6].miR-222 shares the same origin regions as miR-221, targets the same genes as miR-221 [8], and also affects angiogenesis and inflammation [9]. Its expression increases with increasing RA disease activity [10]. High expression levels of miR-221/222 indicate that RA is undergoing high activity.The results of recent studies show that, microRNAs are expressed in the liver and are known to change and influence its various functions [11]. Micro-RNA molecules are involved in the regulation and differentiation of all types of liver cells, including Kupffer cells. Overexpression of various specific microRNAs can attenuate or promote the development of liver fibrosis [12]. It can be said that specific microRNAs are of causative importance in the formation and development of liver diseases. For example, in viral hepatitis B and C, there are studies that micro-RNA expression is involved in stopping viral replication and suppressing inflammation [13]. It is known that liver fibrosis is caused by the activation of lipocytes in excess of the normal amount outside the cell, and this activation has been found to be regulated by microRNAs [14]. This regulation occurs in 2 different directions: promoting fibrosis by inducing cell proliferation, migration and collagen secretion through different signaling pathways (microRNA-9a-5p, microRNA-17-5p, microRNA-21, microRNA-27, microRNA-31, microRNA -33a, microRNA-34a/s, microRNA-125, microRNA-126, microRNA-130a/b, microRNA-181b, microRNA-214-5p, microR-NK-195, microRNA-199a/b, microRNA-221, mi -croRNA-222) and/or antifibrotic (microRNA-16, microRNA-19b, microRNA-29, microRNA-30, microRNA-101, mi-croRNA-122, microRNA133a, microRNA-144, microRNA-146a, microRNA-150, microRNA-155, microRNA-192, microRNA-195, microRNA-335, microRNA-454, microRNA-483) in the direction.A state of increased lipocyte activity showed a decrease in the expression-rate of mi RNA-122, which in turn led to fibrosis. However, the importance of mi RNA-122 and the underlying mechanisms of its degradation have not been fully explored until now [15]. However, according to the latest scientific data, microRNAs are potential markers for detecting and monitoring the state of liver tissue, including fibrosis processes [11,16,17].MicroRNA-122 is involved in regulating the expression of genes involved in carbohydrate, lipid metabolism and iron metabolism in the body. The data of scientific experimental studies devoted to the study of microRNA-122 showed that these molecules are also associated with pathogenesis of hepatitis C, thereby ensuring the life cycle of the virus in the cell.
2. Materials and Methods
- 86 patients diagnosed with RA, but primary pathology of the liver was excluded, and 45 patients who applied to the departments of cardiorheumatology, rheumatology and arthrology, hepatology of the Tashkent Medical Academy (TTA) for treatment (the diagnosis of RA was confirmed in each patient (n=148)) RA with non-alcoholic fatty liver disease (NAFLD) and 17 patients with chronic viral hepatitis (CH) and RA treated at the Department of Chronic Hepatitis of the Epidemiological, Microbiological and Infective Diseases Institute (EMIDIU) were included in the study. According to the category of liver pathology mentioned above, it is divided into 3 subgroups. A written information letter was given by the patients included in the study about their voluntary participation in the study and their consent. Only cases that met the exclusion criteria were selected.The diagnosis of RA was established for results of clinical, instrumental and laboratory studies according to the classification criteria of ACR/EULAR (2010) [18].In the diagnosis of chronic hepatitis, we used the classification of chronic hepatitis recommended by the International Congress of Gastroenterologists held in Los Angeles in 1994 based on the order of the Ministry of Health № 560 dated October 30, 2000.Diagnosis of patients with NAFLD based on the European Association for the Study of Liver Disease (EASL), the European Association for the Study of Diabetes (EASD) and the European Association for the Study of Obesity (EASO) clinical guidelines for the diagnosis, treatment and follow-up of patients with non-alcoholic fatty liver disease (2009) was performed [19]. 5 ml blood samples from each patient were collected in EDTA tubes and used for detection of hepatitis virus serological markers as well as RNA quantification by real-time PCR. Serum extracted from whole blood was stored at a temperature not lower than 70°C and 0,75 ml of Trizol were used for microRNA extraction and detection of microRNA-122 and 221 expression changes by real-time PCR. RNA concentrations were determined using a Nano Drop ND 2000 ( Nano Drop Tech. of USA). Reverse transcription was performed on the biomaterial using the "miScript Reverse Transcription Kit" product of the German company "QIAGEN". As maintained by the manufacturer's requirements was studied the expression-degree of mi RNA-122and 221 (mi Script Green PCR Kit - QIAGEN, Germany). The miRNA level was calculated based on the formula, normalized to U6 sn RNA, and presented in conventional values. Amplification was done on Rotor Gene 6000 and Rotor Gene Q (Qiagen, Germany). The obtained data is analyzed using software program as a graphics. The results were processed using Microsoft Office 2013 and Statistica 6 computer software package. 5% was taken as the critical quantity of the veracity level. The examination of whether the distribution conforms to the law of the normal distribution of features was performed in the instance mi RNA (r=0,0161), r<0,05 miRNA feature distribution is different from normal.
3. Results and Discussion
- The expression levels of serum microRNAs in the examined 3 groups of patients showed a remarkable microRNA profile.The amount of microRNA 221 in the first group was significantly different from the values of the patients in the other 2 groups. It is highly probable that the increase in the expression-rate of microRNA 221 was caused by the replication of HBV and HCV viruses. In addition, RA activity in this group was the highest compared to other groups. The expression of miRNA 122 was low compared to other groups. MicroRNA 221 is known to be a profibrotic microRNA, and its high expression level has been found to promote fibrosis processes and correlate with decreased expression of microRNA 122 in blood serum. In this group of patients, the F1-F2 fibrosis stage took a significant share (r < 0,05).When comparing the expression of microRNAs in the group of patients diagnosed with RA and NAFLD to the patients of group 1 and 3, it was found that the expression of microRNA 122 was significantly higher than the level in the serum of group 1 patients. In parallel, the signs of lipid metabolism were clearly expressed in the blood. When we compared the results with those of the 3rd group of patients, it was found that the expression of microRNA 122 in these patients was high. At the same time, the expression level of microRNA 221 was also noted to be high and it was inversely proportional to the inflammatory activity in RA (r < 0,05 in all cases).In the results of group 3, which did not have primary pathologies in the liver and received long-term basic treatment (metatrexate), it was found that the expression level of microRNA 221 decreased and the indicators of the degree of microRNA increased significantly. These data showed that while treatment of RA with basic, genetically engineered biological drugs (GIBP) suppresses the disease activity, their side effects activate inflammatory processes in the liver and lead to disruption of protein, carbohydrate and fat metabolism. Especially if viral pathologies in the liver are added to this process, the markedly high expression of microRNA 122 can support the entire life cycle of viruses as a result of increasing viral replication and stimulating infectious production of hepatitis viruses.The range of microRNA-122 expression levels was shown in the following 4 graded values: 0,001 – 0,14 minimal values to 0,15 -1,05 medium; 1,05 – 12,88 is highly expressed and >12,89 is the highest value.Analysis of microRNA-122 expression level showed that patients with subordinate volue serum microRNA-122 levels were mainly in group 1.The diversity in prevalence of patients in subgroups 1 and 2-3 of patients with microRNA-122 range in the range of 0,001-0,14 was 56,9% versus 6,7% and 5,8%, respectively. In this range, the estimated odds ratio and risk of complications were 7,9 (95% CI 1,13– 55,58) and 15,7 (95% CI 1,67– 148,1), respectively. OR=15,7 and RR=7,9 compared to high rates, such a difference was statistically significant (χ2=5,9, r>0,01) (Table 1).
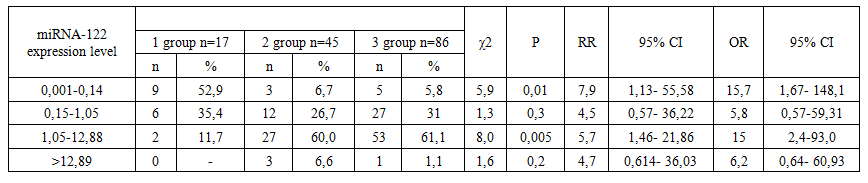 | Table 1. Significant difference in the prevalence of microRNA-122 expression levels in different patient groups with RA and chronic diffuse liver diseases (case-control model) |
4. Conclusions
- Expression levels of miRNA122 and 221 in the serum of RA patients can be used to diagnose and/or predict clinical aspects of the underlying disease and concomitant chronic liver diseases in these patients with high sensitivity and specificity. From the results of the study, we can conclude that the complex correlations of miRNA122 and 221 expression with RA and diffuse liver diseases have been revealed, which may be a promising marker of RA and liver pathologies.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML