-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3789-3795
doi:10.5923/j.ajmms.20251511.08
Received: Sep. 17, 2025; Accepted: Oct. 12, 2025; Published: Nov. 6, 2025

Prognostic Significance of Immunohistochemical Markers in Wilms Tumor in Children
Nishanov D. A., Mustafayev T. K.
Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology of the Republic of Uzbekistan, Scientific and Practical Medical Center for Pediatric Oncology, Hematology and Immunology, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Relevance. Wilms tumor remains one of the leading causes of cancer-related mortality in early childhood. Immunohistochemical markers may potentially improve risk stratification and prognostication, but their clinical significance remains uncertain. Objective. To assess the prognostic value of CD34, Ki-67, and p53 expression in pediatric Wilms tumor in relation to recurrence, metastasis, and overall survival. Materials and Methods. A retrospective study was conducted involving 91 children with histologically confirmed Wilms tumor treated at the Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology (RSSPMCOR) from 2015 to 2022. Epidemiological, morphological, therapeutic, and immunohistochemical data (CD34, Ki-67, p53) were analyzed. Pearson’s χ² test, Kaplan–Meier method, and log-rank test (p < 0.05) were used for statistical evaluation. Results. A significant association was found between tumor stage and histological differentiation (χ² = 101.7; p < 0.00001). Poorly differentiated tumors (G3) were more frequently detected at advanced stages. The prognostic significance of CD34, Ki-67, and p53 for recurrence and metastasis was not statistically confirmed. Overall 5-year survival was 85.6 ± 6.3%. A trend toward poorer outcomes was observed in patients without IHC assessment. Conclusion. Although CD34, Ki-67, and p53 did not demonstrate independent prognostic significance, the absence of their evaluation was more often associated with unfavorable outcomes, highlighting the need to include IHC in the diagnostic standard.
Keywords: Wilms tumor, Children, Immunohistochemistry, CD34, Ki-67, p53, Survival, Recurrence, Metastasis, Prognosis
Cite this paper: Nishanov D. A., Mustafayev T. K., Prognostic Significance of Immunohistochemical Markers in Wilms Tumor in Children, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3789-3795. doi: 10.5923/j.ajmms.20251511.08.
1. Introduction
- Nephroblastoma (Wilms Tumor) is the most common malignant kidney tumor in children, accounting for approximately 6–8% of all malignant neoplasms in pediatric oncology [3,6]. The incidence of nephroblastoma ranges from 8 to 10 cases per million children per year, with a peak occurrence between the ages of 2 and 4. In developed countries, survival rates can reach up to 90% with early diagnosis and appropriate comprehensive treatment [1], while in resource-limited settings, survival remains significantly lower due to delayed detection, lack of diagnostic infrastructure, and non-compliance with international treatment protocols [7].The clinical course of nephroblastoma is often asymptomatic in its early stages, which contributes to a high proportion of advanced cases. In most instances, the tumor is discovered incidentally during abdominal palpation by parents or pediatricians. Standard diagnostic methods are based on ultrasound imaging, CT or MRI scans, followed by histological confirmation after nephrectomy or biopsy. Morphologically, nephroblastoma is a triphasic tumor consisting of blastemal, epithelial, and stromal components, though the histological presentation can vary greatly, necessitating additional stratification [5,10].One of the promising directions in the diagnosis and prognosis of nephroblastoma is immunohistochemical (IHC) analysis, which enables the assessment of the tumor's molecular and biological characteristics. Among the most studied markers are:• WT1, involved in kidney development and nephron differentiation;• p53, which plays a key role in apoptosis regulation;• Ki-67, indicating proliferative activity; and• CD34, a marker of angiogenesis.Studies suggest that high Ki-67 expression may correlate with aggressive tumor behavior and reduced sensitivity to chemotherapy [8] (Yalçın et al., 2014), while TP53 mutations associated with disrupted p53 protein expression are linked to poor prognosis and an increased risk of relapse [9]. CD34, as an indicator of microvascular density, is being investigated for its prognostic value and potential role in therapeutic response, although its significance remains debatable (Yalçın et al., 2014).Despite therapeutic advances — including preoperative chemotherapy (within SIOP protocols) and risk-adapted treatment strategies (as per COG guidelines) — challenges persist in risk stratification and treatment personalization. Not all IHC markers have yet been adopted in clinical practice, despite accumulated evidence of their potential prognostic importance. In settings with limited access to molecular diagnostics, it is particularly critical to identify a minimal panel of diagnostic markers that can enhance the accuracy of stratification and prognostication.The five-year survival rate for nephroblastoma in countries with high-quality healthcare systems ranges from 85–92% [4,6]. However, in cases of late detection, anaplastic forms, or metastatic disease, survival drops to 50–60% or lower. Early diagnosis and comprehensive morpho-immunohistochemical verification remain the key factors determining favorable outcomes, enabling the selection of individualized therapeutic pathways [5].Thus, in the current context, there is an increasing need for comprehensive diagnostics of nephroblastoma that necessarily includes immunohistochemical (IHC) profiling. This approach not only refines the morphological characterization of the tumor but also improves prognostic accuracy and therapeutic efficacy. The present study aims to analyze the clinical, morphological, and immunohistochemical features of nephroblastoma in children at the Republican Oncology Center, with a specific focus on identifying prognostic markers and survival-related factors.Study ObjectiveTo evaluate the clinical, morphological, and immunohistochemical characteristics of nephroblastoma in children and to determine the prognostic significance of CD34, Ki-67, and p53 expression in relation to tumor recurrence, metastatic spread, and overall survival.
2. Materials and Methods
- This study was conducted in a retrospective design and is based on the analysis of clinical, morphological, demographic, and immunohistochemical data from 91 patients with histologically confirmed nephroblastoma who received treatment at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR) and its regional branches between 2015 and 2022.Inclusion criteria for the study were: patient age under 15 years at the time of diagnosis, presence of histologically verified nephroblastoma, completion of a full course of comprehensive treatment (surgical intervention in combination with chemotherapy and/or radiotherapy), and availability of complete clinical and diagnostic documentation sufficient for further analysis.Exclusion criteria included: absence of morphological verification of the diagnosis, presence of other malignant neoplasms, insufficient medical documentation, refusal of treatment or deviation from approved protocols, as well as age over 15 years.Within the framework of the analysis, the following parameters were studied: patient age and sex, duration of anamnesis before medical consultation, nature of clinical symptoms, tumor localization, disease stage according to the TNM system and clinical classification, degree of histological differentiation of the tumor, volume and type of surgical intervention, sequencing and structure of the treatment provided, and presence of recurrence or metastatic spread of the disease.Morphological verification of the diagnosis was carried out based on standard histological sections stained with hematoxylin and eosin. If necessary, immunohistochemical (IHC) analysis was performed using the markers CD34 (angiogenesis), Ki-67 (proliferation), and p53 (tumor suppressor), in accordance with international standards, with visual and semi-quantitative assessment of expression.Tumor classification and staging were performed in accordance with the current recommendations of the World Health Organization (WHO) and the TNM classification of malignant neoplasms.Statistical analysis of the data was carried out using methods of descriptive and analytical statistics. The Pearson chi-square (χ²) test was used to compare categorical variables. For survival analysis, the Kaplan–Meier method was applied, and differences between subgroups were assessed using the log-rank test. The critical level of statistical significance was set at p < 0.05.
3. Study Results
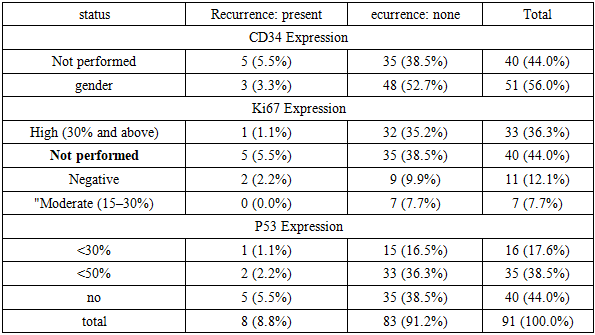
- An analysis of the clinical, morphological, and therapeutic characteristics of patients with nephroblastoma revealed several statistically significant patterns related to age and sex distribution, tumor staging, degree of differentiation, and correlations between treatment regimens and the risk of recurrence or disease progression. The key findings below reflect the structure of the sample and reliable relationships between the parameters studied.Among the 91 patients with nephroblastoma treated at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR), the predominant majority were children under the age of 3 — accounting for 66.0% (n = 60). This finding reflects the typical epidemiological pattern of this malignancy, which most frequently manifests in early childhood. Patients older than 3 years made up the remaining 34.0% (n = 31) of the cohort, which is also consistent with well-known clinical and statistical data on the age distribution of nephroblastoma.The gender composition of the study cohort showed a slight predominance of females: 56.0% (n = 51) were girls, while 44.0% (n = 40) were boys. However, the χ² test revealed no statistically significant differences in gender distribution, either in the overall sample or when stratified by age groups (χ² = 0.00; p = 1.00). These results indicate no gender-specific predisposition to nephroblastoma in this cohort, supporting the widely accepted notion that children of both sexes are equally susceptible to this type of tumor.
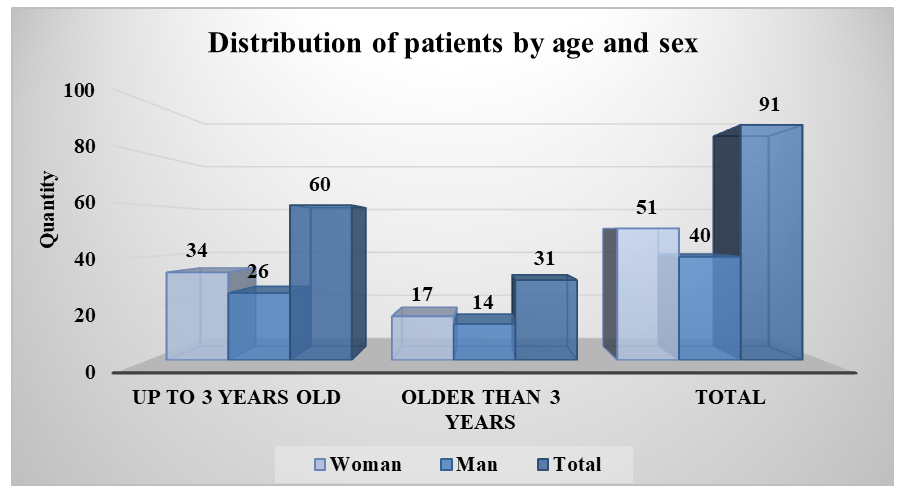 | Figure 1. Distribution of patients by age and gender categories |
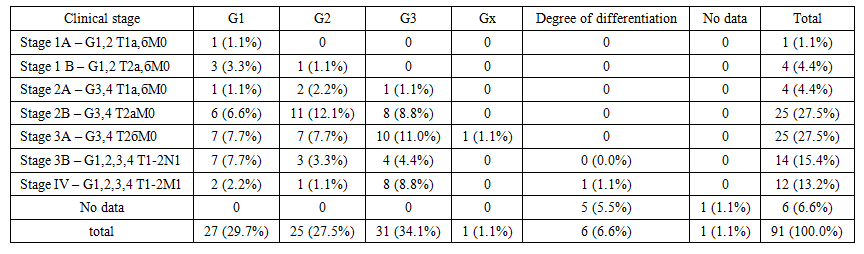 | Table 1. Distribution of patients by clinical stage of the tumor process and tumor differentiation grade |
|
|
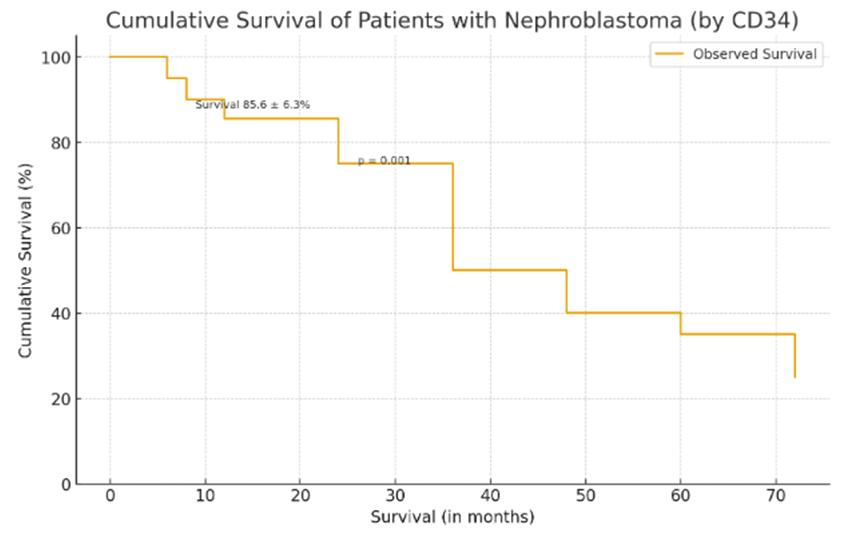 | Figure 2. Cumulative overall survival of patients with nephroblastoma depending on CD34 marker expression |
4. Discussion
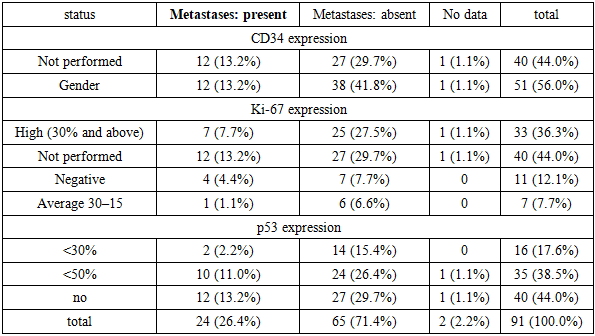
- The results of this retrospective study, which included 91 pediatric nephroblastoma cases treated at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR) and its regional branches, reflect both global epidemiological patterns and specific aspects tied to the realities of diagnostics and treatment in the national oncological setting.The age and sex distribution of the cohort confirmed the typical epidemiology of nephroblastoma, with the highest incidence in children under 3 years of age (66.0%), consistent with international studies. For example, according to Dome et al. (2014), the median age at diagnosis is approximately 3 years, attributed to the embryonal origin and early manifestation of the tumor [3]. Gender distribution showed no statistically significant difference between males and females (χ² = 0.00; p = 1.00), aligning with data from GPOH and SIOP protocols indicating an approximately equal prevalence among sexes [2].The analysis of clinical stage and histological differentiation revealed a significant correlation between tumor progression and morphological signs of malignancy (χ² = 101.7; p < 0.00001). High-grade tumors (G3) were more commonly diagnosed at advanced stages (3B–IV), reflecting the general pathogenetic trend of decreased cellular differentiation as the disease advances. These findings support the concept of morphological evolution in nephroblastoma, as suggested by Breslow et al. (2003), whereby progression is accompanied by genetic instability and loss of differentiation [1].However, it is important to note that the use of the G1–G3 grading scale is not universally standardized for nephroblastoma in international practice. Most protocols, including SIOP and COG, rely on histological subtyping (blastemal, epithelial, mixed, etc.), underscoring the need to adapt morphological terminology to align with global standards [10].The prognostic assessment of immunohistochemical markers CD34, Ki-67, and p53 did not reveal statistically significant associations with recurrence or metastatic spread. CD34 expression, indicating angiogenic activity, was present in 56.0% of patients but showed no correlation with recurrence (p = 0.4632) or metastasis (p = 0.7635). This confirms the ambiguous role of angiogenesis in nephroblastoma, as previously reported by Yalçın et al. (2014), who found that microvascular density may be an auxiliary but not an independent prognostic factor [11].Similarly, a high Ki-67 proliferation index (≥30%) was observed in 36.3% of patients but was not linked to poorer survival or increased recurrence risk. According to Irtan et al. (2015), Ki-67 may be more useful in evaluating tumor responsiveness to neoadjuvant chemotherapy rather than as an independent prognostic marker [8]. The lack of clear associations in this study could be attributed to the limited scope of IHC data and incomplete documentation.Regarding p53, its expression also failed to show prognostic significance (p = 0.5411), despite literature suggesting its association with poor prognosis and chemoresistance in nephroblastoma [9]. A likely explanation is the high proportion of patients (44%) without IHC analysis, among whom the worst outcomes were observed. This highlights both diagnostic variability and the possible prognostic importance of lacking molecular data itself, as noted by Howard et al. (2008) in their study of pediatric cancer epidemiology in resource-limited settings [7].The overall 5-year survival rate, calculated using the Kaplan–Meier method (85.6 ± 6.3%), is consistent with outcomes reported in international multicenter studies under NWTS, SIOP, and COG protocols [6]. The "plateau" observed in the survival curve after 36 months aligns with the natural history of nephroblastoma, where the greatest risk of progression and mortality occurs in the first 2–3 years after diagnosis [4]. However, the apparent differences in survival between marker expression subgroups (p = 0.001), despite visual distinctions, require further stratification and log-rank testing, without which statistical interpretation remains limited.
5. Conclusions
- This study confirmed the epidemiological, morphological, and clinical-prognostic features of nephroblastoma in children treated at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR). A statistically significant correlation was established between the clinical stage and the degree of tumor differentiation, underscoring the prognostic value of morphological analysis.Although immunohistochemical markers (CD34, Ki-67, p53) were widely used, their expression in this cohort did not show a statistically significant association with recurrence, metastasis, or overall survival. However, the predominance of adverse outcomes among patients who did not undergo immunohistochemical (IHC) evaluation highlights the critical importance of molecular diagnostics in risk stratification.The findings emphasize the need to standardize approaches to diagnosis, treatment, and monitoring of nephroblastoma through the integration of modern biomarkers and adherence to internationally recognized protocols.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML