-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3775-3778
doi:10.5923/j.ajmms.20251511.05
Received: Sep. 10, 2025; Accepted: Oct. 10, 2025; Published: Nov. 3, 2025

The Role of Biomarkers by Age and Sex in Acute Ischemic Stroke
Vakhabova N. M.1, Bokiyeva F. A.2, Abduvahobov A. A.2, Nuraliyeva A. Sh.3, Kodirova S. A.3
1Doctor of Medical Sciences, Tashkent State Medical University, Tashkent, Uzbekistan
2Neurologist of Department of Neurology and Medical Psychology, Tashkent State Medical University, Tashkent, Uzbekistan
3Master’s Student, Department of Neurology and Medical Psychology, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Vakhabova N. M., Doctor of Medical Sciences, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In recent years, increasing attention has been paid to “novel” risk factors that promote the development of chronic dysmetabolic processes in the vascular walls and brain tissues. Among these, hyperhomocysteinemia (HHcy) holds a special place. According to several authors, HHcy is closely associated not only with damage to intracranial and visceral vessels but also plays a role in the pathogenesis of cerebral vascular accidents. HHcy adversely affects vascular autoregulation, lipid metabolism, and the blood's coagulation potential.
Keywords: Acute ischemic stroke, Hyperhomocysteinemia, Endothelial dysfunction, Homocysteine, Thrombomodulin, Endothelin-1
Cite this paper: Vakhabova N. M., Bokiyeva F. A., Abduvahobov A. A., Nuraliyeva A. Sh., Kodirova S. A., The Role of Biomarkers by Age and Sex in Acute Ischemic Stroke, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3775-3778. doi: 10.5923/j.ajmms.20251511.05.
1. Introduction
- One of the key objectives faced by angioneurologists is to provide effective and appropriate care not only in the acute phase of ischemic stroke (IS) but also during the early recovery phase. This necessitates an in-depth study of the pathogenesis of brain structural damage occurring in the early post-stroke period [1]. According to some researchers, there is a rapid increase in endothelial dysfunction markers following acute IS. Endothelial dysfunction is often associated with increased total plasma homocysteine levels and may be partially related to oxidative stress. Other authors suggest that thrombomodulin is associated with cerebrovascular ischemic conditions and may serve as an important biomarker for the early onset of ischemic stroke. Our findings confirmed that thrombomodulin levels in AIS patients were significantly higher than in both the control group and CCI patients (p<0.05), indicating severe endothelial dysfunction in the acute phase [2].
2. Materials and Methods
- A study of biochemical markers of endothelial dysfunction (homocysteine, thrombomodulin, and endothelin-1) was conducted in 165 patients with acute ischemic stroke (AIS), of whom 29 had chronic cerebral ischemia (CCI). The control group consisted of 30 practically healthy individuals without clinical signs of chronic cerebrovascular disorders. Among the 136 patients with AIS in the acute phase, 26.5% (36 patients) were of young age, 40.4% (55 patients) of middle age, and 33.1% (45 patients) of elderly and senile age.
3. Results and Discussion
- The analysis showed that thrombomodulin (TM) levels in AIS patients during the acute phase were significantly higher compared to those in the control group (p<0.05) and in patients with CCI (p<0.05), indicating a high degree of endothelial dysfunction. Moreover, an age-related increase in TM levels was observed. In young patients, TM levels averaged 58.9±5.6 µg/L, while in elderly and senile patients, the levels were 2.2 times higher (p<0.05).The level of endothelin-1 (ET-1) in AIS patients during the acute phase was on average 3.4 times higher than in the control group (p<0.01). In CCI patients, ET-1 levels were 1.7 times higher than in the control group (p<0.05). With advancing age, ET-1 levels increased from 8.6±0.12 pg/mL in young patients to 10.8±0.55 pg/mL in elderly and senile patients (p<0.001).Correlation analysis showed a moderate to strong direct correlation between age and levels of homocysteine (r=0.568; p=0.002), thrombomodulin (r=0.732; p=0.003), and endothelin-1 (r=0.668; p=0.003).With aging, these indicators increased significantly: TM levels in elderly and senile patients were 2.2 times higher than in young individuals (p<0.05). Similarly, ET-1 levels were 3.4 times higher in AIS patients than in controls (p<0.01), and 1.7 times higher in CCI patients (p<0.05). In older patients, ET-1 levels reached up to 10.8±0.55 pg/mL, compared to 8.6±0.12 pg/mL in younger patients (p<0.001). Homocysteine levels also rose with age: 2.6-fold increase in younger patients (p<0.05) and 4.8-fold in elderly patients compared to controls (p<0.001).Compared to the control group, patients with chronic cerebral ischemia (CCI) showed a statistically significant increase in homocysteine levels (p<0.05). In patients in the acute phase of ischemic stroke (IS), this marker was 1.9 times higher in younger individuals and 3.5 times higher in elderly and senile patients (p<0.001).The study also identified certain patterns in the relationship between biochemical markers of endothelial dysfunction (homocysteine, thrombomodulin, endothelin-1) and sex-related characteristics of the patients [3]. These patterns are presented in Table 1 and illustrate that the severity of endothelial dysfunction and its association with specific biochemical markers vary depending on the patient's sex [4]. This information is of practical importance for the further development of more precise approaches to the prevention and treatment of endothelial dysfunction.As shown in the data, male patients exhibited higher levels of both thrombomodulin and homocysteine across all age groups compared to female patients [5].
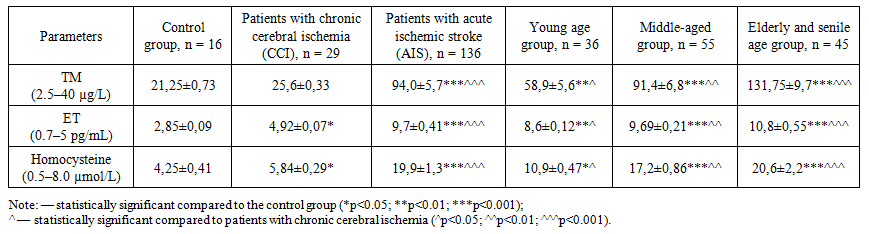 | Table 1. Presents age-related quantitative changes in biochemical markers of endothelial dysfunction (homocysteine, thrombomodulin, endothelin-1) in the examined patients |
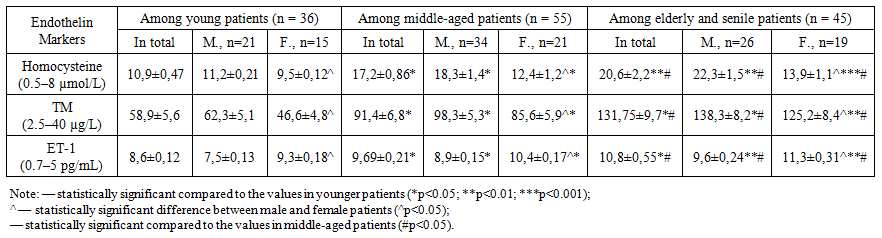 | Table 2. Sex- and age-related quantitative changes in biochemical markers of endothelial dysfunction (homocysteine, thrombomodulin, endothelin-1) in patients with acute ischemic stroke |
4. Conclusions
- An age-dependent increase in the levels of homocysteine, thrombomodulin, and endothelin-1 was observed in patients during the acute phase of ischemic stroke, confirming the presence of pronounced endothelial dysfunction. These elevations, which intensified with age, reflect significant impairments in endothelial function and highlight its critical role in stroke pathogenesis.Sex-specific differences in the levels of endothelial dysfunction biomarkers were also observed: men exhibited higher levels of homocysteine and thrombomodulin, whereas women showed significantly elevated endothelin-1 levels.The combined use of these biomarkers provides additional insight into the functional state of brain tissue during the hyperacute phase of ischemic stroke and may serve as an important tool for outcome prediction.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML