-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(11): 3753-3759
doi:10.5923/j.ajmms.20251511.01
Received: Oct. 5, 2025; Accepted: Oct. 28, 2025; Published: Nov. 3, 2025

Development of Acute Kidney Injury in Children with Oral Poisonings
Xamroyev Baxodir Olimkulovich1, Axmedov Yusufjon Mahmudovich2, Xursanov Yokubjon Erkinugli3
1Samarkand Branch of the Republican Center for Emergency Medical Care, Samarkand, Uzbekistan
2Professor, Head of the Department of Pediatric Surgery № 2, Samarkand State Medical University, Samarkand, Uzbekistan
3PhD, Department of Surgical Diseases № 2, Samarkand State Medical University, Samarkand, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study aimed to characterize renal dysfunction and assess the diagnostic value of early biomarkers in children with oral chemical poisonings. A retrospective review of 500 medical records (1990–2014) was followed by a prospective cohort of 120 patients aged 1–16 years (2015–2016); 40 healthy children served as controls. Standard clinical and imaging assessments were combined with urinary ELISA measurements of KIM-1, NGAL, and cystatin C. No serum creatinine elevation was observed, underscoring the limited sensitivity of conventional markers at early stages. In contrast, urinary NGAL was above control maxima in 11.6% and KIM-1 in 12.5% of patients; urinary cystatin C was frequently decreased and never exceeded control values, suggesting the need for complementary serum testing. The greatest biomarker elevations occurred with NSAID, neuroleptic, and caustic (including vinegar essence) poisonings. At one-month follow-up, KIM-1 declined >2-fold and NGAL >1.5-fold, indicating recovery. Conclusion: urinary NGAL and KIM-1 are sensitive early indicators of AKI and useful for treatment monitoring in pediatric oral poisonings, whereas urinary cystatin C is less informative and should preferably be assessed in serum. Early biomarker-based detection may reduce progression to chronic kidney disease.
Keywords: Acute kidney injury, Children, Oral poisonings, Toxic nephropathy, KIM-1, NGAL, Cystatin C, pRIFLE, KDIGO, Biomarkers
Cite this paper: Xamroyev Baxodir Olimkulovich, Axmedov Yusufjon Mahmudovich, Xursanov Yokubjon Erkinugli, Development of Acute Kidney Injury in Children with Oral Poisonings, American Journal of Medicine and Medical Sciences, Vol. 15 No. 11, 2025, pp. 3753-3759. doi: 10.5923/j.ajmms.20251511.01.
1. Introduction
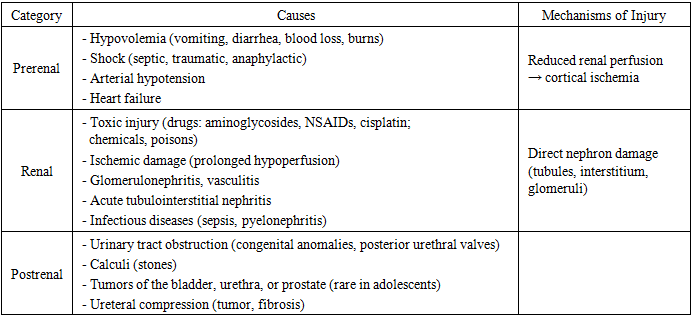
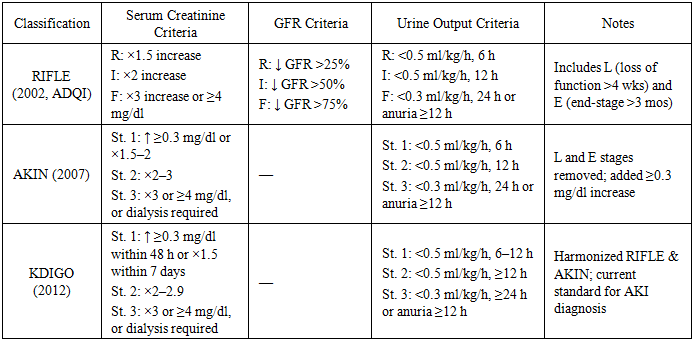
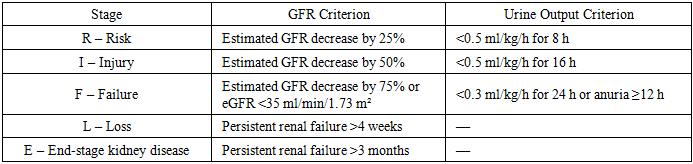
- At present, acute kidney injury (AKI), including forms induced by toxic exposures, remains one of the major causes of impaired renal function in children and often determines clinical outcomes. In critically ill pediatric patients, AKI-associated mortality reaches approximately 25% by day 28 [1].The first internationally accepted consensus definition of AKI was introduced in 2005 during the Acute Kidney Injury Network (AKIN, Amsterdam) conference. It recommended replacing the term “acute renal failure” with “acute kidney injury” (AKI), while reserving “acute renal failure” only for the most severe cases [2]. AKI is now recognized as a syndrome of progressive kidney dysfunction, ranging from minimal functional impairment to complete loss of renal ability to maintain water, electrolyte, and acid-base balance. Etiologies include severe trauma, major surgery, infectious diseases, and rapidly progressive hereditary disorders [3].Among the leading causes of AKI in children today are oral poisonings of chemical or drug-related origin. Toxic exposures frequently lead to toxic nephropathy, which is often complicated by AKI [4,5]. Nephrotoxic agents exert direct cellular toxicity, disrupt renal microcirculation, cause tissue hypoxia, and trigger immune-mediated damage. As a result, the ICD-10 introduced class N14 — “tubulointerstitial and tubular lesions induced by drugs” [6]. In hospitalized children, administration of nephrotoxic drugs is a major contributor to AKI [8,9]. Accidental or intentional ingestion of toxic doses (poisoning) further increases the risk. Critically ill children are particularly vulnerable due to the combined effects of sepsis, hypotension, polypharmacy, and difficulties in accurate renal function assessment [10,11]. Currently, no definitive curative treatment for AKI exists. The main modifiable risk factor remains avoiding unnecessary nephrotoxic agents when safer alternatives are available. Early detection is difficult since pediatricians and nephrologists still rely on serum creatinine levels, which rise only at later stages. Promising biomarkers such as NGAL, KIM-1, and cystatin C have the potential to identify AKI earlier, before serum creatinine increases [12]. From a pathophysiological perspective, three major categories of AKI are distinguished:• Prerenal (impaired renal perfusion due to hypotension, hypovolemia, or shock),• Renal (toxic or ischemic injury, inflammatory processes),• Postrenal (urinary tract obstruction) [13].In summary, oral poisonings in children represent one of the most important risk factors for AKI. This condition highlights the need for early recognition, cautious drug administration, and timely supportive treatment to prevent long-term renal complications Table 1.
|
|
|
|
2. Materials and Methods
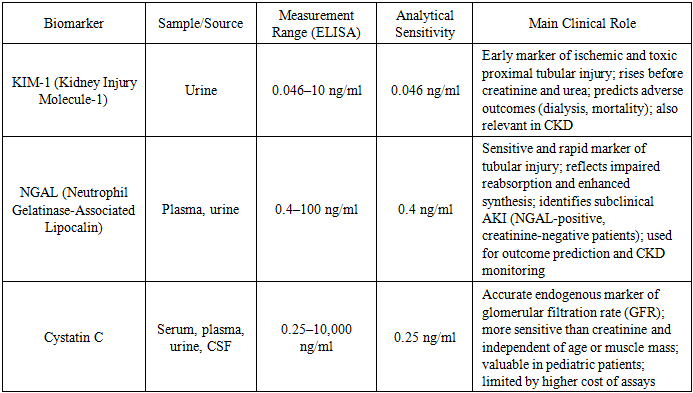
- Preliminary stage. At the first stage, a retrospective analysis of 500 medical records of children treated at the Toxicology Department of Filatov Children’s City Hospital No. 13 between 1990 and 2014 was performed. All patients had moderate to severe chemical poisonings.In younger children (ages 1–3 years), intoxications were most often accidental, involving:• caustic agents (vinegar essence, lighter fluids, detergents),• nasal decongestants,• less commonly — antihypertensive or psychotropic drugs and other substances.In older children and adolescents, poisonings were mainly intentional or mixed, including alcohol, psychotropic drugs, NSAIDs, antispasmodics, sedatives, and tranquilizers.Clinical patterns of specific intoxications:• Vinegar essence poisoning caused burns of the gastrointestinal tract and airways, severe pain, dysphagia, hypersalivation, and blood alterations. Kidney damage was due to acute hemoglobinuric nephrosis with discolored urine, proteinuria, hematuria, glucosuria, crystalluria, and oxaluria. Ultrasound revealed renal edema and poor corticomedullary differentiation, while Doppler studies showed increased resistance index.• Vasoconstrictors induced arterial hypotension, bradycardia, pallor, CNS depression, drowsiness, hypothermia, sweating; AKI developed due to renal ischemia from vasospasm.• NSAIDs caused abdominal pain, nausea, vomiting (sometimes with blood), tremor; ultrasound demonstrated increased renal echogenicity, and laboratory tests revealed enzymuria (elevated alkaline phosphatase and γ-glutamyltransferase).• Barbiturates and psychotropic drugs produced CNS depression, hypotension, bradycardia, impaired thermoregulation, oliguria/anuria, proteinuria, hematuria. Ultrasound revealed increased parenchymal thickness and echogenicity, with reduced GFR.Overall, 80% of patients showed metabolic abnormalities (crystalluria, phosphaturia, triple-phosphate crystals, uraturia); 60% had glomerular damage (proteinuria), and 10% had tubular dysfunction (glucosuria). These findings suggest that most chemically poisoned children developed AKI of varying severity, with the most profound changes observed in vinegar essence, NSAID, barbiturate, vasoconstrictor, and combustion product poisonings.Main stage. The prospective part included 120 children aged 1–16 years admitted to the same department from September 2015 to October 2016 with different chemical intoxications:• neuroleptics ,• NSAIDs (27),• vinegar essence (18),• antihypertensives (12),• phenazepam (9),• alcohol (6),• decongestants (6),• combustion products (3).Diagnostic methods included:• routine clinical and laboratory tests (urinalysis, complete blood count, biochemical blood and urine tests, GFR estimation, coagulation profile, acid-base balance);• imaging (renal ultrasound with Doppler);• selected tests for enzymuria (alkaline phosphatase, γ-glutamyltransferase).Novel biomarkers of renal injury. All patients were additionally screened for urinary biomarkers of kidney damage:• KIM-1 (Kidney Injury Molecule-1),• NGAL (Neutrophil Gelatinase-Associated Lipocalin),• Cystatin C.These markers were evaluated as promising diagnostic tools for early detection of subclinical AKI in children with oral chemical poisonings.Methods of Study and Rationale for Biomarker SelectionLaboratory techniques. Acute kidney injury (AKI) biomarkers were measured using enzyme-linked immunosorbent assay (ELISA) kits:• Cystatin C: measured in serum, plasma, urine, and cerebrospinal fluid (range: 0.25–10,000 ng/ml, sensitivity: 0.25 ng/ml).• NGAL (neutrophil gelatinase-associated lipocalin): measured in plasma, urine, and culture medium (range: 0.4–100 ng/ml, sensitivity: 0.4 ng/ml).• KIM-1 (Kidney Injury Molecule-1): quantified using Human Quantikine ELISA Kit in urine (range: 0.046–10 ng/ml, sensitivity: 0.046 ng/ml).All analyses were performed in the laboratory of the N. N. Blokhin Russian Cancer Research Center, Ministry of Health of Russia.Control group. To establish reference values, a group of 40 conditionally healthy children undergoing routine medical check-ups at the Filatov Children’s City Hospital was examined. None had any somatic pathology.Grouping criteria:• severity of poisoning (moderate, severe),• type of toxic agent,• reduction of urine output:○ <0.5 ml/kg/h for 16 h (RIFLE Injury),○ <0.3 ml/kg/h for 24 h or anuria ≥12 h (RIFLE Failure),• presence of urinary abnormalities (proteinuria, glucosuria, hematuria),• ultrasound/Doppler findings (parenchymal hyperechogenicity, edema, increased resistance index).Biomarker characteristicsKIM-1 is a 90 kDa transmembrane glycoprotein absent in healthy kidneys but highly expressed in proximal tubular cells after ischemic or toxic injury. Its urinary concentration rises much earlier than serum creatinine or urea, making it a reliable early marker of AKI. Elevated KIM-1 predicts adverse clinical outcomes, including dialysis requirement and mortality, and is also increased in chronic kidney diseases. Importantly, KIM-1 levels are stable in urine samples.NGAL is a 25 kDa protein produced under cellular stress. It binds iron, limits bacterial growth, suppresses apoptosis, and promotes tissue repair. Under normal conditions, NGAL is freely filtered and reabsorbed in proximal tubules. With tubular injury, urinary NGAL excretion rises sharply due to impaired reabsorption and increased local synthesis.NGAL is considered one of the most sensitive and early biomarkers of AKI. Importantly, its levels can rise even in the absence of elevated creatinine (“NGAL-positive, creatinine-negative” patients), indicating subclinical kidney injury and higher risk of poor outcomes. Urinary and plasma NGAL strongly correlate with creatinine, GFR, proteinuria, and histological kidney damage. NGAL is also used as a marker of CKD progression and severity.Cystatin C. Cystatin C is a nonglycosylated cysteine protease inhibitor and is now recognized as one of the most accurate endogenous markers of glomerular filtration rate. Unlike creatinine, cystatin C levels are largely independent of age, body mass, or muscle mass, making it especially useful in pediatric diagnostics.It is more sensitive than creatinine in detecting early kidney dysfunction but is limited by the relatively high cost of test kits. Cystatin C is measured using ELISA, PETIA, and PENIA, with ELISA offering the greatest accuracy at low concentrations.Currently, cystatin Cis regarded as a reliable and sensitive marker for evaluating renal filtration function. Ongoing discussions in the scientific literature address whether it can be considered the earliest biomarker of kidney dysfunction compared to other established methods.Cystatin C is a low-molecular-weight protein that is constantly synthesized at a stable rate by all nucleated cells. It freely passes through the glomerular filtration barrier, is completely metabolized in the proximal tubules, and—unlike creatinine—is not secreted by renal tubules. These characteristics ensure its stability and high accuracy as a marker of glomerular filtration rate (GFR).Beyond its diagnostic role, cystatin C is also a potent inhibitor of cysteine proteases. These enzymes are involved in protein and tissue degradation, and their excessive activity may lead to tissue injury. By inhibiting protease activity, cystatin C helps regulate normal physiological processes, maintain tissue homeostasis, and protect organs, including the kidneys.Thus, cystatin C serves not only as a promising biomarker for AKI and CKD but also as a regulatory protein with an important physiological role. Its diagnostic utility is particularly significant in children, since cystatin C levels are largely independent of age, sex, and muscle mass, making it more universal and reliable than creatinine.
3. Results of the Study
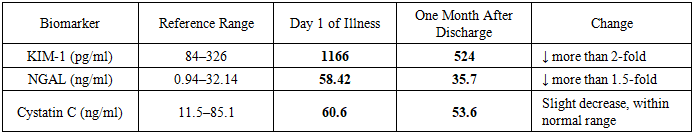
- The concentrations of acute kidney injury (AKI) biomarkers in urine varied depending on the severity of poisoning and the type of toxic agent. Notably, serum creatinine elevation was not observed in any of the examined children, highlighting its limited sensitivity for detecting early kidney damage.At the same time, 14 patients (11.6%) demonstrated urinary NGAL levels above the maximum values of the control group, while 15 patients (12.5%) had increased urinary KIM-1. In contrast, urinary cystatin C in most cases (79 children) was below the minimal control values, and in no case did it exceed the upper reference range. This frequent reduction in cystatin C excretion may reflect elevated serum levels, representing a kind of “mirror effect.” Thus, low urinary cystatin C could potentially serve as an additional diagnostic marker of AKI, although such a conclusion cannot be made without simultaneous serum measurements.Based on the study results, it can be concluded that the most informative urinary markers of AKI in children with chemical oral poisonings are NGAL and KIM-1. Their significant increase was most often observed in children with intoxications caused by NSAIDs, neuroleptic drugs, caustic agents, and vinegar essence—the same categories that, according to retrospective analysis, had the highest incidence of renal complications.Clinical case. Patient A., a 1-year-1-month-old girl, was admitted to the Toxicology Department of Filatov Children’s City Hospital No. 13 with the diagnosis: “Severe vinegar essence poisoning.” Upon admission, her condition was critical, with marked respiratory failure requiring mechanical ventilation. Clinical examination revealed extensive chemical burns of the oropharynx, upper respiratory tract, and upper gastrointestinal tract, as well as skin burns (anterior chest surface — approximately 10%).This case exemplifies the severity of systemic manifestations in caustic substance poisonings and the high risk of AKI, emphasizing the need for early biomarker-based diagnostics.Clinical Case Results. At admission, the following findings were obtained in a child with severe vinegar essence poisoning:• Complete blood count: hemoglobin — 80 g/L, neutrophilic left shift, ESR — 35 mm/h.• Acid–base balance: pH — 7.45; pCO₂ — 43.6 mmHg; Hb — 82 g/L; Ht — 25%.• Urinalysis: erythrocytes — 23 per HPF; protein — 0.9 g/L.• Renal ultrasound: right kidney — 62.8 × 28.6 mm, left kidney — 65.9 × 26.7 mm; parenchymal thickness right — 13 mm, left — 9 mm; preserved corticomedullary differentiation; diffuse cortical hyperechogenicity.• Chest X-ray: enhanced vascular pattern, subsegmental atelectasis in the left upper lobe.• ECG: sinus rhythm, heart rate 150–160 bpm.• Oliguria.• Biochemistry: no azotemia (creatinine — 38 µmol/L, urea — 5.8 mmol/L).• eGFR (Schwartz formula): 105.2 ml/min.By the end of the first day of illness, urinary AKI biomarkers were measured:• KIM-1: 1166 pg/ml (reference 84–326 pg/ml);• NGAL: 58.42 ng/ml (reference 0.94–32.14 ng/ml);• Cystatin C: 60.6 ng/ml (reference 11.5–85.1 ng/ml).These results confirmed the presence of acute kidney injury (AKI) already on the second day, well before the appearance of azotemia. Early diagnosis made it possible to promptly adjust therapy: in addition to infusion, antibacterial, and symptomatic treatment, energy-tropic and angioprotective agents were introduced for 30 days.Follow-up. At one month after discharge, repeat testing showed a clear decline in urinary AKI biomarkers:• KIM-1: 524 pg/ml (reference 84–326 pg/ml);• NGAL: 35.7 ng/ml (reference 0.94–32.14 ng/ml);• Cystatin C: 53.6 ng/ml (reference 11.5–85.1 ng/ml).Thus, early detection of AKI and timely therapy resulted in a favorable dynamic: KIM-1 decreased more than twofold, NGAL decreased by over 1.5 times. This case highlights the importance of novel biomarkers in identifying AKI before conventional signs such as azotemia appear, which is critical for prognosis in children with severe chemical poisonings. Table 5.
|
4. Conclusions
- In summary, the development of AKI in children exposed to chemical poisonings results from a combination of adverse factors, including systemic homeostatic imbalance, disturbances in systemic and renal hemodynamics, and water–electrolyte imbalance. Therefore, maintaining a high level of clinical vigilance in such cases is essential. Early diagnosis using novel biomarkers, together with timely initiation of pathogenetic therapy, reduces the risk of progression to CKD or end-stage renal disease (ESRD) and ensures more complete recovery of renal function.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML