-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3736-3741
doi:10.5923/j.ajmms.20251510.98
Received: Sep. 16, 2025; Accepted: Oct. 12, 2025; Published: Oct. 31, 2025

Hormonal Profile Results in Women with Menstrual Cycle Phase Dysfunction
Yuldasheva M. A., Shukurov F. I.
Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Shukurov F. I., Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background. Infertility is one of the most prevalent gynecological problems among women of reproductive age, with hormonal and ovulatory dysfunctions being key etiological factors. Hormonal imbalance during different phases of the menstrual cycle negatively affects folliculogenesis, ovulation, and endometrial receptivity. Objective. To evaluate hormonal profile indicators in women with menstrual cycle phase dysfunction and assess their association with reproductive outcomes. Materials and Methods. A total of 120 women aged 18–40 years were included and divided into four groups: follicular phase dysfunction (n=30), ovulatory phase dysfunction (n=30), luteal phase dysfunction (n=30), and a control group of healthy women (n=30). Serum levels of FSH, LH, estradiol, estrone, estriol, progesterone, total and free testosterone, and DHEA-S were measured using ECLIA. Results. Women with follicular phase dysfunction showed elevated FSH and reduced estradiol and progesterone levels; ovulatory phase dysfunction was characterized by increased LH, testosterone, and DHEA-S; luteal phase dysfunction demonstrated marked reduction of progesterone and estradiol, indicating luteal insufficiency. The control group had values within physiological ranges. Correlation analysis revealed a significant association between estradiol levels and dominant follicle diameter and endometrial thickness (r=0.61–0.68; p<0.01). ROC analysis identified estradiol (>85 pg/ml) and progesterone (>3.8 ng/ml) as highly diagnostic cut-off values for predicting ovulation and implantation. Conclusion. Hormonal profile alterations significantly affect reproductive function. Estradiol, progesterone, testosterone, and DHEA-S serve as reliable biomarkers for assessing ovulation and implantation potential.
Keywords: Infertility, Menstrual cycle, Hormonal dysfunction, Estradiol, Progesterone, Testosterone, Endometrial receptivity
Cite this paper: Yuldasheva M. A., Shukurov F. I., Hormonal Profile Results in Women with Menstrual Cycle Phase Dysfunction, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3736-3741. doi: 10.5923/j.ajmms.20251510.98.
Article Outline
1. Introduction
- Infertility is one of the most prevalent gynecological problems among women of reproductive age, with prevalence rates estimated at 10–20% across different countries [1]. According to the World Health Organization (WHO), female factors—particularly hormonal and ovulatory dysfunctions—account for at least half of all infertility cases and represent key pathogenetic causes [2,3].In the physiology of the menstrual cycle, the precise coordination of the follicular, ovulatory, and luteal phases plays a critical role. Hormonal imbalances at any phase can negatively influence ovulation, folliculogenesis, and endometrial receptivity [4]. For example, estrogen deficiency in the follicular phase may restrict follicular development and prevent dominant follicle formation [5]. Disruption of luteinizing hormone (LH) secretion during the ovulatory phase can lead to anovulation, while insufficient progesterone levels in the luteal phase result in endometrial hypoplasia, reduced vascularization, and diminished implantation potential [6–8].Hormonal dysfunctions often reflect only endocrine alterations and may not fully capture the morphofunctional changes in ovulation and endometrial receptivity. Therefore, in addition to hormonal profiling, echographic and Dopplerometric parameters, as well as metabolomic analyses, are considered essential for comprehensive evaluation [9,10].Recent studies have highlighted that estradiol, estrone, estriol, progesterone, testosterone fractions, and dehydroepiandrosterone sulfate (DHEA-S) serve as key biomarkers that determine ovulatory processes, endometrial proliferation, and implantation efficiency [11–13]. Identifying alterations in the hormonal profile is crucial not only for understanding the pathogenesis but also for enabling individualized prognosis and the selection of personalized therapeutic strategies [14–16].Moreover, hormonal profile analysis allows for the assessment of the diverse clinical manifestations of follicular, ovulatory, and luteal phase dysfunctions and their impact on reproductive function [17,18]. This contributes to a deeper understanding of infertility pathogenesis and provides opportunities to develop highly accurate diagnostic and prognostic models [19,20].Therefore, in this study, hormonal profile parameters were evaluated in women with menstrual cycle phase dysfunctions, with the aim of determining their impact on reproductive function and their clinical-prognostic significance.
2. Purpose of the Research
- To assess the hormonal profile parameters in women with menstrual cycle phase dysfunctions and evaluate their association with reproductive function.
3. Materials and Methods
- This study included a total of 120 women of reproductive age (18–40 years). All participants were stratified into four groups based on their hormonal status and menstrual cycle characteristics: Group I – women with follicular phase hormonal dysfunction (n = 30), Group II – women with ovulatory phase hormonal dysfunction (n = 30), Group III – women with luteal phase hormonal dysfunction (n = 30), and the comparison group – healthy women with physiological menstrual cycles (n = 30).Inclusion criteria were reproductive age (18–40 years), failure to conceive for at least 12 months, laboratory and clinical confirmation of hormonal imbalance, and provision of written informed consent for participation in the study. Exclusion criteria included organic pathologies of the reproductive organs (endometriosis, uterine fibroids, tubal-peritoneal factors), severe somatic and endocrine diseases (diabetes mellitus, thyroid disorders), infectious diseases, and a history of long-term hormonal therapy.For all women, blood samples were collected according to the respective phase of the menstrual cycle. The following parameters were determined: gonadotropins (FSH and LH), estrogen fractions (estradiol, estrone, estriol), progesterone, testosterone fractions (total and free), and dehydroepiandrosterone sulfate (DHEA-S). Additionally, the LH/FSH ratio was assessed. All hormonal measurements were performed using electrochemiluminescence immunoassay (ECLIA) on an Immulite 2000 XPI analyzer (Siemens, Germany).The obtained data were processed using SPSS version 25.0. Student’s t-test was applied for parametric variables, while the χ² test was used for nonparametric data. Correlations between hormonal parameters and reproductive indicators were evaluated using Pearson’s correlation analysis. In all cases, a p-value of <0.05 was considered statistically significant.
4. Results
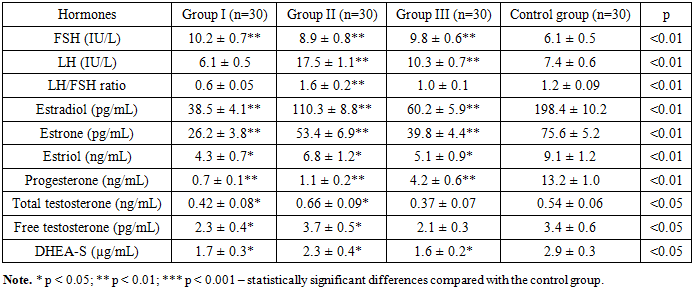
- Analysis of the results showed that in women of Group I with follicular phase hormonal dysfunction, mean FSH levels were 10.2 ± 0.7 IU/L, LH – 6.1 ± 0.5 IU/L, and the LH/FSH ratio – 0.6 ± 0.05. Estradiol concentration was 38.5 ± 4.1 pg/mL, estrone – 26.2 ± 3.8 pg/mL, estriol – 4.3 ± 0.7 ng/mL, progesterone – 0.7 ± 0.1 ng/mL, total testosterone – 0.42 ± 0.08 ng/mL, free testosterone – 2.3 ± 0.4 pg/mL, and DHEA-S – 1.7 ± 0.3 µg/mL. These findings reflect diminished ovarian reserve and impaired folliculogenesis, accompanied by reductions in estrogen and progesterone, which indicate decreased ovulatory activity.In Group II (ovulatory phase dysfunction), FSH was 8.9 ± 0.8 IU/L, LH – 17.5 ± 1.1 IU/L, and the LH/FSH ratio – 1.6 ± 0.2. Estradiol (E₂) levels were 110.3 ± 8.8 pg/mL, estrone (E₁) – 53.4 ± 6.9 pg/mL, estriol (E₃) – 6.8 ± 1.2 ng/mL, and progesterone – 1.1 ± 0.2 ng/mL. Total testosterone was 0.66 ± 0.09 ng/mL, free testosterone – 3.7 ± 0.5 pg/mL, and DHEA-S – 2.3 ± 0.4 µg/mL. Relatively elevated LH, testosterone, and DHEA-S levels suggested androgenization during ovulation, impaired follicular development, and a risk of hyperandrogenemia.In Group III (luteal phase dysfunction), mean FSH was 9.8 ± 0.6 IU/L, LH – 10.3 ± 0.7 IU/L, and the LH/FSH ratio – 1.0 ± 0.1. Estradiol was 60.2 ± 5.9 pg/mL, estrone – 39.8 ± 4.4 pg/mL, estriol – 5.1 ± 0.9 ng/mL, progesterone – 4.2 ± 0.6 ng/mL, total testosterone – 0.37 ± 0.07 ng/mL, free testosterone – 2.1 ± 0.3 pg/mL, and DHEA-S – 1.6 ± 0.2 µg/mL. The most prominent hormonal alterations in this group were significantly decreased progesterone and estradiol, reflecting luteal phase insufficiency and impaired implantation potential.In the control group, all parameters remained within physiological limits: FSH – 6.1 ± 0.5 IU/L, LH – 7.4 ± 0.6 IU/L, LH/FSH ratio – 1.2 ± 0.09, estradiol – 198.4 ± 10.2 pg/mL, estrone – 75.6 ± 5.2 pg/mL, estriol – 9.1 ± 1.2 ng/mL, progesterone – 13.2 ± 1.0 ng/mL, total testosterone – 0.54 ± 0.06 ng/mL, free testosterone – 3.4 ± 0.6 pg/mL, and DHEA-S – 2.9 ± 0.3 µg/mL (p<0.01).
|
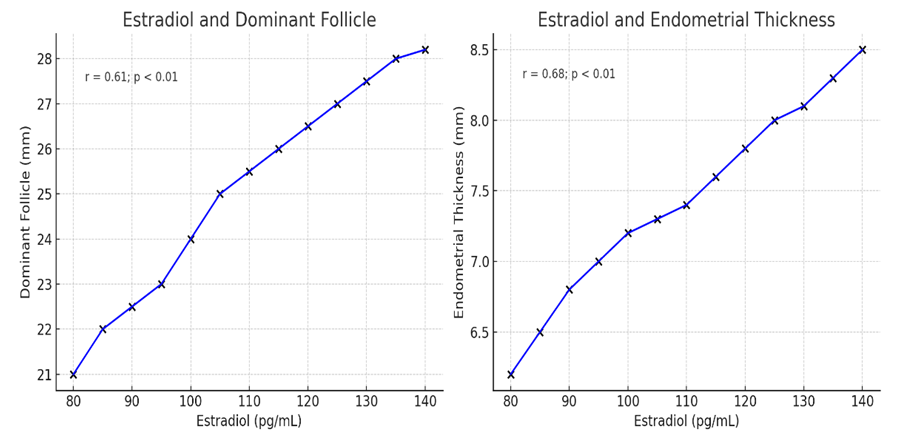 | Figure 1. Correlation between estradiol levels, dominant follicle size, and endometrial thickness in women included in the study |
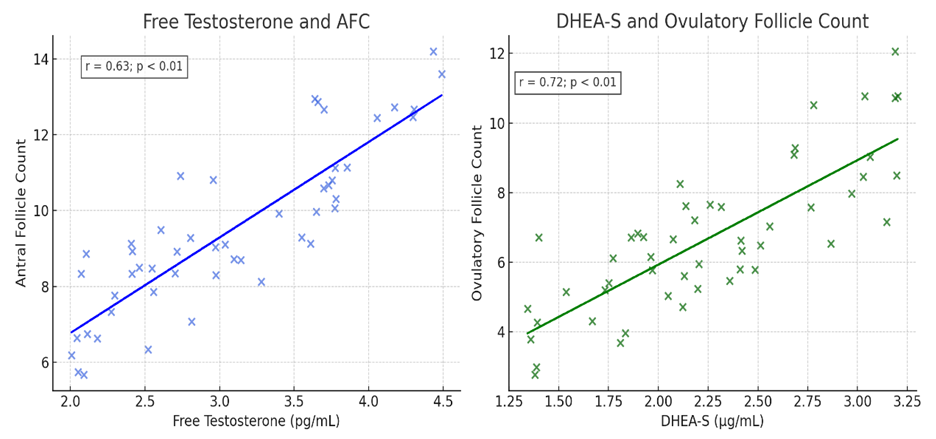 | Figure 2. Correlation between free testosterone, AFC, and ovulatory follicles in women included in the study |
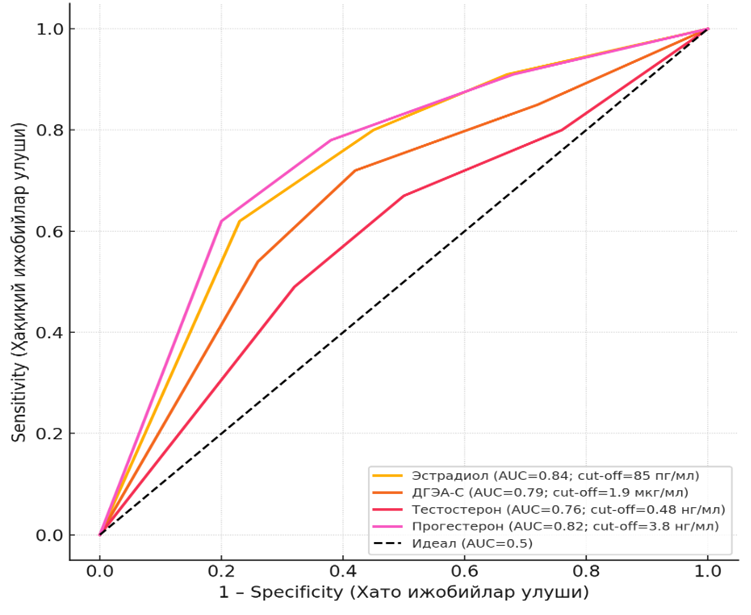 | Figure 3. ROC analysis of hormonal biomarkers in predicting ovulation and implantation |
5. Discussion
- The results of this study confirm that hormonal dysfunctions in different phases of the menstrual cycle play a decisive role in the pathogenesis of female infertility. Our data demonstrated that follicular phase disorders were associated with reduced estradiol and progesterone levels, impaired folliculogenesis, and diminished ovarian reserve, while ovulatory phase dysfunctions were characterized by excessive luteinizing hormone (LH) and hyperandrogenemia, and luteal phase insufficiency was marked by critically low progesterone and estradiol values. These findings are consistent with previous reports highlighting the central role of endocrine and ovulatory disorders in the etiology of infertility [1,3,4,7,11,12,13].In particular, the luteal phase findings of our study, showing significantly decreased progesterone levels, are in line with the concept of luteal phase deficiency as a major factor impairing endometrial receptivity and implantation potential [5,17]. This corresponds with earlier reports that insufficient progesterone production leads to defective secretory transformation of the endometrium, reduced vascularization, and increased implantation failure rates [6,10].The observed relationship between elevated LH, increased androgens (testosterone and DHEA-S), and impaired ovulatory dynamics reflects the pathogenic mechanisms of hyperandrogenemia in reproductive failure. Similar results have been reported in both clinical and experimental studies, confirming that excessive androgen activity suppresses follicular development, alters gonadotropin feedback, and negatively affects oocyte maturation [8,9,13].Another important outcome of our study is the identified correlation between estradiol levels and both follicular development and endometrial thickness (r=0.61–0.68, p<0.01). This supports the critical role of estrogens in regulating folliculogenesis and preparing the endometrium for implantation [14,18]. Moreover, reduced estrogen levels in infertile women with menstrual cycle dysfunctions explain the frequent findings of thin endometrium, hypoplasia, and impaired implantation rates.The integration of metabolomic perspectives into reproductive endocrinology also enhances the interpretation of our results. Recent metabolomics-based investigations of follicular fluid have confirmed that altered profiles of steroid hormones, androgens, and metabolic intermediates are directly associated with poor ovarian response and reduced pregnancy outcomes in assisted reproductive technology (ART) cycles [15,16,19,20]. This aligns with our findings, underscoring the importance of comprehensive multiparametric analysis—combining hormonal, echographic, and metabolomic data—for precise diagnosis and prognosis.The novelty of this study lies in its multiphase and multiparametric evaluation of hormonal dysfunction in infertile women. Unlike most previous studies that focused on single markers or single-cycle phases, our research provides a comprehensive analysis of estradiol, progesterone, androgens, and DHEA-S across different phases of the menstrual cycle, correlating them with ovarian reserve markers and endometrial receptivity. Such an integrated approach provides a more reliable prognostic framework for infertility diagnostics and the development of personalized therapeutic strategies.Clinically, these findings highlight the necessity of not only routine hormonal testing but also the inclusion of advanced diagnostic approaches such as ROC-analysis and metabolomic profiling to determine individualized cut-off values for predictive markers. For instance, in our study, the estradiol cut-off of 85 pg/mL (AUC=0.84) was a reliable predictor of ovulation, while the progesterone threshold of 3.8 ng/mL (AUC=0.82) allowed accurate estimation of luteal phase competence. These thresholds may serve as practical tools for optimizing preconception counseling, ART protocols, and personalized treatment strategies.Overall, our study supports the view that infertility in reproductive-aged women is not a result of isolated hormonal changes, but rather a multifactorial condition where endocrine dysfunction, ovarian reserve depletion, and endometrial receptivity impairment interact in a complex manner. Therefore, dynamic monitoring of the hormonal profile across menstrual phases, combined with metabolomic and echographic assessments, represents an evidence-based and clinically valuable approach to improving the outcomes of infertility treatment.
6. Conclusions
- This study demonstrated that hormonal dysfunction across different phases of the menstrual cycle has a significant negative impact on female reproductive function. Reduced levels of estrogen and progesterone, combined with elevated LH and androgens, were associated with impaired folliculogenesis, ovulatory dysfunction, and decreased endometrial receptivity. Correlation and ROC analyses confirmed that estradiol, progesterone, testosterone, and DHEA-S serve as highly valuable diagnostic markers for predicting ovulation and implantation outcomes. Therefore, comprehensive evaluation of the hormonal profile represents an essential tool for the early diagnosis of infertility and for guiding individualized therapeutic strategies.Conflict of Interest: The authors declare that there is no conflict of interest regarding the publication of this paper.Funding: This research was carried out as part of the institutional research activities of Tashkent State Medical University and received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML