-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3728-3735
doi:10.5923/j.ajmms.20251510.97
Received: Oct. 7, 2025; Accepted: Oct. 23, 2025; Published: Oct. 31, 2025

Surgical Treatment of Acute Pancreatitis Using Minimally Invasive Interventions
Zarpullayev Javokhir Salimjon ugli
Samarkand State Medical University, Samarkand, Uzbekistan
Correspondence to: Zarpullayev Javokhir Salimjon ugli, Samarkand State Medical University, Samarkand, Uzbekistan.
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

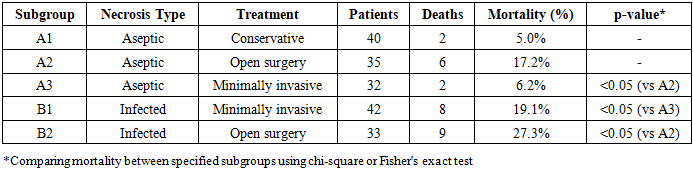
The study presents results of examination of 182 patients with moderate and severe acute pancreatitis aged 31-81 years, treated at the multidisciplinary clinic of Samarkand State Medical University and the Samarkand branch of the Republican Scientific Center for Emergency Medical Care during 2018-2023. Video laparoscopic lavage is the main method of surgical treatment for aseptic pancreatic necrosis, while percutaneous drainage under ultrasound guidance is effective for treating pancreatic abscess or infected fluid collections. Minimally invasive operations were performed in 51.4% of patients with severe acute pancreatitis. Minimally invasive interventions are preferable in treating patients with aseptic severe pancreatitis compared to open operations, justified by a significant reduction in mortality rates (6.2% and 17.2% respectively). In infected pancreatic necrosis, this indicator was 19.1% and 27.3%, representing statistically significant differences (p<0.05).
Keywords: Acute pancreatitis, Pancreatic necrosis, Minimally invasive surgery, Video laparoscopy, Percutaneous drainage, Mortality
Cite this paper: Zarpullayev Javokhir Salimjon ugli, Surgical Treatment of Acute Pancreatitis Using Minimally Invasive Interventions, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3728-3735. doi: 10.5923/j.ajmms.20251510.97.
Article Outline
1. Introduction
- Acute pancreatitis represents a significant healthcare challenge worldwide, characterized by increasing incidence, substantial economic burden, and considerable variation in clinical outcomes. The disease spectrum ranges from mild self-limiting inflammation to severe necrotizing pancreatitis with multi-organ failure, presenting complex diagnostic and therapeutic dilemmas for clinicians. Understanding the factors that influence disease progression and optimizing treatment strategies remain critical priorities in modern surgical practice [1,2].The epidemiological significance of acute pancreatitis cannot be overstated. Global studies report annual incidence rates between 13 and 45 cases per 100,000 population, with notable geographic variation reflecting differences in risk factors, particularly alcohol consumption and gallstone prevalence. Approximately 20-30% of patients develop severe disease, characterized by persistent organ failure or local complications including necrosis, which carries substantially higher morbidity and mortality. The economic impact extends beyond direct medical costs, encompassing lost productivity, long-term disability, and reduced quality of life [3,4].Mortality in severe acute pancreatitis remains unacceptably high despite advances in critical care and surgical techniques. While mortality in mild disease approximates 1-3%, severe forms demonstrate rates of 15-25%, escalating to 30-50% when infected necrosis develops. These statistics underscore the imperative for evidence-based treatment algorithms that optimize outcomes while minimizing intervention-related complications. The timing and nature of surgical intervention profoundly influence prognosis, making treatment selection decisions critically important [5].Traditional management of severe acute pancreatitis relied heavily on early surgical intervention through open necrosectomy. However, accumulated evidence demonstrated that early surgery during the acute inflammatory phase frequently precipitated or exacerbated systemic complications without addressing the underlying pathology. This recognition sparked a fundamental shift toward initial conservative management with delayed intervention only when clear indications emerged, particularly documented infection or symptomatic persistent sterile collections [6].Objective of the study. To improve the outcomes of acute pancreatitis treatment through implementation of a differentiated approach to selection of surgical correction methods with priority application of minimally invasive interventions.
2. Material and Methods of Research
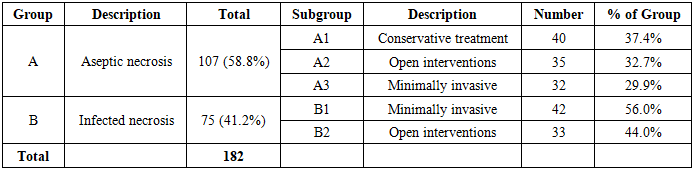
- This retrospective cohort study analyzed treatment outcomes in patients with moderate to severe acute pancreatitis managed at two tertiary referral centers: the multidisciplinary clinic of Samarkand State Medical University and the Samarkand branch of the Republican Scientific Center for Emergency Medical Care. The study period extended from January 2018 through December 2023, encompassing five complete years of consecutive patient enrollment. The institutional review boards of both participating centers approved the study protocol, and informed consent was obtained from all surviving patients or their legal representatives for inclusion of clinical data in research analyses.The study population comprised 182 patients diagnosed with acute pancreatitis of moderate severity or severe clinical course based on internationally accepted classification systems. Patient ages ranged from 31 to 81 years, with a mean age of 51.6±13.7 years. The cohort included 78 women (42.9%) and 104 men (57.1%), reflecting the well-established male predominance in acute pancreatitis epidemiology, particularly in populations with significant alcohol-related disease burden. All patients underwent comprehensive initial assessment including detailed clinical history, physical examination, and laboratory evaluation upon admission.Diagnostic criteria for severe acute pancreatitis incorporated multiple complementary modalities to ensure accurate classification. Clinical assessment focused on identifying systemic inflammatory response syndrome criteria, hemodynamic instability, and evidence of organ dysfunction. Laboratory investigations included serial measurements of pancreatic enzymes (amylase and lipase), complete blood counts with attention to leukocytosis and hematocrit changes suggesting hemoconcentration, comprehensive metabolic panels evaluating renal and hepatic function, inflammatory markers including C-reactive protein, and arterial blood gases when respiratory compromise was suspected.Imaging studies played a central role in diagnosis and severity assessment. All patients underwent abdominal ultrasonography at admission, which provided rapid bedside evaluation of pancreatic morphology, peripancreatic fluid collections, gallbladder pathology, and biliary ductal dilation. However, ultrasound has known limitations in visualizing the pancreas due to overlying bowel gas, particularly in the acute inflammatory setting. Therefore, computed tomography with intravenous contrast enhancement was performed in all patients to definitively establish diagnosis, assess extent of pancreatic and peripancreatic necrosis, and detect complications. CT scans were typically obtained 72-96 hours after admission to allow adequate time for necrosis development and optimal contrast between viable and non-viable tissue. The modified CT severity index was calculated for each patient to quantify disease severity.Diagnostic laparoscopy represented an additional diagnostic modality employed selectively in this patient population. This minimally invasive procedure allowed direct visualization of the peritoneal cavity, assessment of ascitic fluid characteristics, identification of pancreatic and peripancreatic inflammation, and collection of peritoneal fluid samples for biochemical and microbiological analysis. Laparoscopy was performed when diagnostic uncertainty existed regarding severity, when therapeutic intervention was contemplated, or when clinical deterioration suggested complications not adequately explained by imaging findings. The procedure was conducted under general anesthesia in the operating room using standard laparoscopic equipment with pneumoperitoneum pressures maintained at 12-14 mmHg.All 182 patients in the study cohort were categorized as having severe acute pancreatitis based on the comprehensive evaluation described above. Classification systems utilized included the revised Atlanta classification and the determinant-based classification, both of which emphasize persistent organ failure as the defining feature of severity. Organ failure was defined using the modified Marshall scoring system, with scores of 2 or greater in any organ system indicating failure. Common manifestations included respiratory failure requiring supplemental oxygen or mechanical ventilation, cardiovascular failure necessitating vasopressor support, and renal failure with rising creatinine or decreased urine output.A critical aspect of patient categorization involved distinguishing aseptic from infected pancreatic necrosis, as this distinction fundamentally influences treatment strategy. Infection was diagnosed based on combination of clinical deterioration despite appropriate management, positive blood cultures with organisms consistent with pancreatic infection, gas bubbles within necrotic collections on CT imaging suggesting gas-forming organisms, and microbiological analysis of samples obtained through percutaneous aspiration or during surgical intervention. Fine-needle aspiration was not routinely performed due to concerns about seeding infection, sampling error, and procedural complications. When specimens were obtained during therapeutic procedures, they were immediately transported to the microbiology laboratory for Gram staining, aerobic and anaerobic culture, and antibiotic sensitivity testing.Based on presence or absence of infection, patients were allocated to two main groups. Group A consisted of 107 patients (58.8% of the total cohort) with aseptic pancreatic necrosis. These patients demonstrated pancreatic or peripancreatic necrosis on imaging but without evidence of infection based on the criteria described above. Group B comprised 75 patients (41.2%) with documented infected pancreatic necrosis. The substantial proportion of patients with infected necrosis reflects the tertiary referral nature of the participating institutions, which receive transfers of severely ill patients from regional hospitals.Group A was further subdivided into three subgroups based on treatment approach. Subgroup A1 included 40 patients (37.4% of Group A) who were managed with conservative treatment only, without surgical intervention. This approach was selected for patients with limited necrosis, clinical stability or improvement with supportive care, and absence of complications requiring drainage or debridement. Conservative management encompassed aggressive fluid resuscitation guided by hemodynamic monitoring, pain control with analgesics, nutritional support preferentially via enteral route when tolerated, prophylaxis against stress ulceration and venous thromboembolism, and close monitoring for development of complications.Subgroup A2 consisted of 35 patients (32.7% of Group A) who underwent open surgical interventions. Indications for open surgery in aseptic necrosis included development of symptomatic fluid collections not amenable to percutaneous drainage, abdominal compartment syndrome requiring decompression, diagnostic uncertainty requiring exploration, or failure of less invasive approaches. Open procedures typically involved laparotomy through appropriate incisions based on necrosis location, debridement of non-viable tissue, copious irrigation, and placement of drains. Some patients underwent necrosectomy with closed continuous lavage systems, while others had packing and planned relaparotomy strategies.Subgroup A3 comprised 32 patients (29.9% of Group A) treated with minimally invasive interventions. These techniques included video laparoscopic exploration and lavage, percutaneous catheter drainage under ultrasound or CT guidance, and video-assisted retroperitoneal debridement when necrosis was accessible via this route. Selection of specific minimally invasive technique depended on location and extent of necrosis, patient anatomy, local expertise, and equipment availability. The general principle involved achieving adequate drainage or debridement while minimizing surgical trauma.Group B patients with infected pancreatic necrosis were divided into two subgroups. Subgroup B1 included 42 patients (56% of Group B) managed with minimally invasive interventions. Recognition that infected necrosis requires intervention, but that minimally invasive approaches might suffice in selected cases, guided this treatment selection. Techniques mirrored those used in subgroup A3, though typically requiring more extensive or prolonged drainage and often multiple procedures. The step-up approach philosophy, beginning with catheter drainage and escalating to minimally invasive necrosectomy only if drainage alone proved insufficient, was applied in many cases.Subgroup B2 consisted of 33 patients (44% of Group B) treated with open surgical operations. Indications for open surgery in infected necrosis included extensive necrosis involving multiple compartments, failure of minimally invasive approaches to control sepsis, anatomical factors precluding safe minimally invasive access, or clinical deterioration requiring urgent intervention before organizing walled-off necrosis developed. Open procedures in infected necrosis involved similar techniques to those described for subgroup A2, with emphasis on complete removal of infected necrotic tissue, thorough irrigation, and establishment of adequate drainage.Treatment protocols at both participating institutions followed standardized algorithms developed based on international guidelines and institutional experience. All patients received initial resuscitation and supportive care in intensive care or high-dependency units depending on severity. Fluid resuscitation targeted urine output of at least 0.5 mL/kg/hour, mean arterial pressure above 65 mmHg, and normalization of hematocrit. Lactated Ringer's solution was preferred as initial resuscitation fluid based on evidence suggesting potential advantages over normal saline in acute pancreatitis.Pain management utilized multimodal approaches including opioid analgesics, with patient-controlled analgesia systems when appropriate, supplemented by non-opioid adjuncts when feasible. Nutritional support was initiated early, with enteral nutrition via nasogastric or nasojejunal tubes preferred over parenteral nutrition based on evidence of reduced infectious complications. Nutrition was started within 48-72 hours of admission in hemodynamically stable patients. Prophylactic antibiotics were not routinely administered to patients with sterile necrosis, while those with confirmed or strongly suspected infected necrosis received broad-spectrum antimicrobials adjusted based on culture results and local resistance patterns.Timing of intervention represented a critical decision point. Current evidence supports delayed intervention, typically after 4 weeks when walled-off necrosis has matured, except in cases of abdominal compartment syndrome, uncontrolled bleeding, or overwhelming sepsis unresponsive to intensive care measures. This approach was followed in the majority of cases, though clinical judgment was applied individually. Patients undergoing minimally invasive interventions typically had these performed 3-6 weeks after disease onset, while open operations when performed occurred across a wider timeframe depending on indication.Percutaneous drainage procedures were performed by interventional radiologists using ultrasound or CT guidance. Under local anesthesia with conscious sedation, pigtail catheters of 10-14 French caliber were placed directly into fluid collections or necrotic cavities. Drains were irrigated regularly and left in place until output decreased to minimal levels and clinical improvement was sustained. Serial imaging monitored collection resolution. Some patients required multiple drains in different locations or sequential drain placements as collections evolved.Video laparoscopic interventions were conducted in the operating room under general anesthesia. Standard laparoscopic access was established, typically with three to four ports depending on specific requirements. The abdomen was systematically explored, inflammatory exudate was aspirated, and lavage with warmed saline was performed until effluent cleared. In cases requiring debridement, necrotic tissue was gently separated from viable pancreas and surrounding structures, though aggressive necrosectomy was avoided due to bleeding risk. Drains were placed in the lesser sac and other relevant spaces and brought out through separate sites.Open surgical procedures involved various incision types based on necrosis location and extent. Upper midline laparotomy provided excellent exposure for pancreatic head and body necrosis, while bilateral subcostal incisions offered superior access to the entire gland. Upon entering the abdomen, extensive inflammatory changes including turbid ascites, saponification of omentum and mesenteric fat, and retroperitoneal inflammatory edema were typically encountered. The gastrocolic ligament was divided to access the lesser sac, allowing visualization of the pancreas. Necrotic tissue was meticulously debrided using combination of blunt dissection, irrigation, and gentle curettage, preserving viable pancreatic tissue and carefully protecting vascular structures including the splenic vessels and superior mesenteric vessels.Postoperative management following surgical interventions required intensive monitoring and support. Patients typically remained in intensive care for several days, with ongoing attention to fluid balance, hemodynamic stability, respiratory support as needed, and pain control. Drains were managed with regular flushing protocols and removed when output criteria were met. Nutritional support was continued or reinitiated as soon as gastrointestinal function allowed. Serial laboratory monitoring tracked inflammatory markers, organ function, and metabolic parameters. Repeat imaging was performed to assess treatment response and detect complications.
3. Results and Discussion
- The comparative analysis of mortality rates among the studied subgroups revealed statistically significant differences that have important clinical implications. The overall mortality rate in Group A (aseptic pancreatic necrosis) was 10 patients out of 107, representing 9.3% of this population. This relatively favorable outcome reflects the less severe disease course typically associated with sterile necrosis compared to infected necrosis, assuming appropriate management is provided. However, mortality still occurred in nearly one in ten patients, underscoring that aseptic necrosis remains a serious condition with life-threatening potential.
|
|
|
4. Conclusions
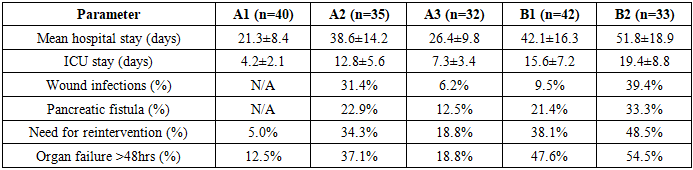
- 1. Minimally invasive surgical interventions represent effective treatment options for severe acute pancreatitis, successfully implemented in 51.4% of patients in this series. Video laparoscopic lavage serves as the primary minimally invasive approach for aseptic pancreatic necrosis, while percutaneous drainage under ultrasound or computed tomography guidance provides effective treatment for pancreatic abscess and infected fluid collections. These techniques, when appropriately applied, achieve source control while minimizing surgical trauma and associated complications.2. Minimally invasive interventions demonstrate clear advantages over open surgical operations in patients with aseptic severe acute pancreatitis, supported by statistically significant reduction in mortality rates (6.2% versus 17.2%, p<0.05). This substantial 11 percentage point difference in mortality, combined with additional benefits including shorter hospital stays, reduced intensive care requirements, and lower complication rates, establishes minimally invasive approaches as the preferred intervention when surgical management becomes necessary for aseptic pancreatic necrosis. Conservative management without surgical intervention remains appropriate for carefully selected patients with limited necrosis who demonstrate clinical improvement with supportive care, achieving favorable outcomes with 5.0% mortality in this series.3. In infected pancreatic necrosis, minimally invasive interventions similarly provide superior outcomes compared to open surgical approaches, with mortality rates of 19.1% versus 27.3% respectively (p<0.05). While this 8.2 percentage point difference appears smaller than observed in aseptic necrosis, it remains clinically meaningful and statistically significant. The higher overall mortality in infected versus aseptic necrosis (19.1% versus 6.2% for minimally invasive approaches; 27.3% versus 17.2% for open surgery) confirms infection as a critical determinant of prognosis independent of surgical technique.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML