-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3681-3685
doi:10.5923/j.ajmms.20251510.85
Received: Oct. 5, 2025; Accepted: Oct. 22, 2025; Published: Oct. 31, 2025

Modified Approach to Treatment Options in Metastatic Bone Involvement
Polatova Djamilya Shogayratovna1, Abdusattorov Oybek Kobilovich2, Savkin Aleksandr Vladimirovich3, Batirov Fares Batirovich4
1DcS, Professor, Head of the Department of Oncology and Medical Radiology, Tashkent State Medical University, Heads the Scientific and Practical Medical Center for Pediatric Oncology, Hematology, and Immunology, Uzbekistan
2Phd Student, Tashkent State Medical University, First-class physician, Surgeon in the 2nd Department of Pediatric Surgical Oncology, Scientific and Practical Medical Center for Pediatric Oncology, Hematology, and Immunology, Uzbekistan
3PhD, Assistant Professor, Department of Oncology and Medical Radiology, Tashkent State Medical University, Head of the 2nd Department of Pediatric Surgical Oncology, Scientific and Practical Medical Center for Pediatric Oncology, Hematology, and Immunology, Uzbekistan
4Second-Year Master's Student in General Oncology of the Department of Oncology and Medical Radiology, Tashkent State Medical University, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Metastatic bone involvement (MBI) is a frequent complication in cancer patients, significantly affecting their quality of life and prognosis. Accurate assessment of bone involvement and fracture risk is crucial for optimal treatment planning. Methods: A retrospective analysis of 76 patients with MBI was conducted from 2015 to 2022 at the Musculoskeletal Tumors department of RSNPMSCOiR clinic. Patients were categorized based on pathological fractures, and a modified computational model was used to evaluate fracture risk. Imaging techniques such as CT, MRI, and PET were employed for accurate assessment. Results: The study confirmed that timely intervention, including palliative surgery and radiation therapy, plays a vital role in improving outcomes for MBI patients. Patients with high fracture risk required preventive osteosynthesis or, in some cases, therapeutic resection. The computational model provided valuable predictions regarding the extent of bone damage and fracture risk. Conclusion: A multidisciplinary approach is essential for managing MBI. The mathematical model developed in this study serves as an effective tool for personalized treatment planning, optimizing the balance between over-treatment and under-treatment.
Keywords: Metastatic bone involvement, Fracture risk, Orthopedic surgery, Palliative care, Bone metastasis management, Prognostic model, Multidisciplinary approach, Long bone metastasis, Treatment strategies
Cite this paper: Polatova Djamilya Shogayratovna, Abdusattorov Oybek Kobilovich, Savkin Aleksandr Vladimirovich, Batirov Fares Batirovich, Modified Approach to Treatment Options in Metastatic Bone Involvement, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3681-3685. doi: 10.5923/j.ajmms.20251510.85.
1. Introduction
- In 2020, approximately 20 million cases of malignant neoplasms were diagnosed globally, and it is estimated that bone metastases are present in 15% of these patients. The frequency of bone metastases varies depending on the type of primary cancer: 70 to 95% in multiple myeloma, 65 to 90% in prostate cancer, and approximately 65 to 75% in breast cancer [1]. Bone tissue is a highly vascularized and innervated structure that undergoes continuous remodeling, regulated by specific hormones, growth factors, and cytokines. The unique bone environment, which includes bone cells (osteocytes, osteoclasts, and osteoblasts), the mineralized bone matrix, and various growth factors, provides a favorable microenvironment for the growth of metastatic tumor cells. Metastatic bone involvement (MBI) is classified into three types based on the nature of bone destruction: osteolytic, osteoblastic, and mixed. Osteolytic bone metastases are typically observed in lung cancer, kidney cancer, and multiple myeloma [5], where bone destruction is induced by the secretion of osteolytic factors from the tumor, such as PTHrP and IL-11, leading to increased bone resorption [2]. Osteoblastic bone metastases, primarily associated with prostate cancer, are mediated by signaling through endothelin 1, GDF15, and Wnt [2]. Mixed bone lesions are frequently observed in metastatic breast cancer [2,3].Currently, one of the most critical and challenging areas in clinical oncology is the treatment of patients with MBI. According to the malignant neoplasm registry in Uzbekistan, the incidence has increased over the past decade, yet thanks to advancements in treatment, the mortality rates have remained almost unchanged, with a slight trend towards reduction. After the lungs and liver, the skeleton ranks third in terms of frequency of metastatic involvement. In modern oncological practice, there is a clear trend where the effectiveness of treatment for patients with malignant tumors is not only determined by the success of the treatment for the primary lesion but also by the effective management of distant metastases, as secondary bone tumors negatively impact the quality of life and reduce its duration. The main complications of MBI include severe pain syndrome, pathological bone fractures or the risk of their occurrence, which significantly impair the quality of life and, in certain cases, may lead to disability. In most patients with MBI, surgical interventions are aimed at palliative care [4]; however, in certain cases, when there is solitary metastatic involvement, radical resections with reconstructive and plastic components may be considered.The treatment of patients with MBI requires a multidisciplinary approach that incorporates all available methods of treatment in oncological practice. Within the multidisciplinary team, orthopedic surgeons play an essential role in enhancing the quality of life for cancer patients. Surgical intervention in MBI is aimed at reducing pain, eliminating or preventing pathological fractures, restoring limb function, and improving the patient’s functional independence. In some clinical cases, in the presence of resectable solitary metastases in the skeleton, surgical treatment may serve a therapeutic function, aiming to prolong life expectancy [13]. The most commonly employed methods in the treatment of metastases in long bones are endoprosthetics and intramedullary osteosynthesis [19]. When various parts of the skeleton are affected, appropriate oncological orthopedic care improves the patient's quality of life and facilitates subsequent specialized therapies (chemotherapy, targeted therapy, radiation therapy, etc.) in comfortable conditions for the patient. Pelvic MBI significantly reduces the quality of life, not only causing intense pain but also severely limiting mobility, often leading to disability. The current surgical methods can prevent pathological fractures, alleviate pain, and improve the quality of life. Additionally, an alternative treatment option for this group of patients may include radiation therapy, which provides significant pain reduction [14].Thus, decision-making for these patients is often empirical or based on the clinical variants of the disease. Indeed, optimal therapeutic guidelines or standardized prognostic systems for managing patients with MBI have not yet been developed. Considering this, the aim of this study is to select the most appropriate treatment option for patients with MBI through a comprehensive assessment of prognostic factors.
2. Materials and Methods
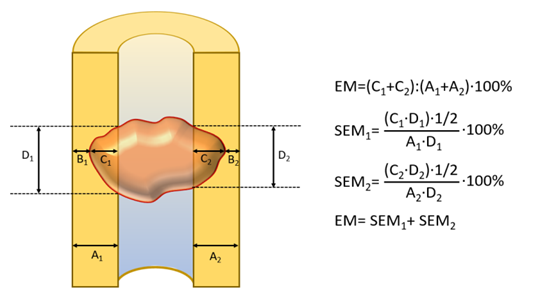
- We conducted a comprehensive retrospective analysis of clinical data from 76 patients with metastatic bone involvement (MBI) who were hospitalized and treated in the "Musculoskeletal Tumors" department of the RSNPMSCOiR clinic from 2015 to 2022. The study included patients who received palliative treatment for MBI complicated by pathological fractures, the risk of their occurrence, or severe pain syndrome due to tumor-induced skeletal changes. Patients with MBI of the skull, hands, and feet were excluded from the study. In order to select an adequate treatment method, all patients underwent a complete comprehensive examination, which included computed tomography (CT), bone scintigraphy, positron emission tomography (PET), magnetic resonance imaging (MRI), ultrasound of the affected area, and regional lymph nodes.Undoubtedly, when determining the risk of pathological fractures, it is essential to accurately assess the extent of bone involvement by the metastatic tumor. In this study, we developed a modified computational model for assessing the degree of damage to long bones. Accordingly, the degree of bone involvement on a longitudinal cross-section is determined by dividing the total size of the cortical layer affected by the tumor by the total size of the anatomically intact cortical layer in the horizontal plane at the largest tumor diameter, and multiplying by 100 (Figure 1). It is important to note that, in addition to the overall degree of damage to the cortical part of the bone, a separate calculation for each side may allow us to predict which side is more affected and estimate the fracture angle. This formula reveals that when calculating the level of involvement, the part of the tumor located in the trabecular part of the bone is not taken into account, because, in long bones, the cortical layer bears most of the load.
3. Results
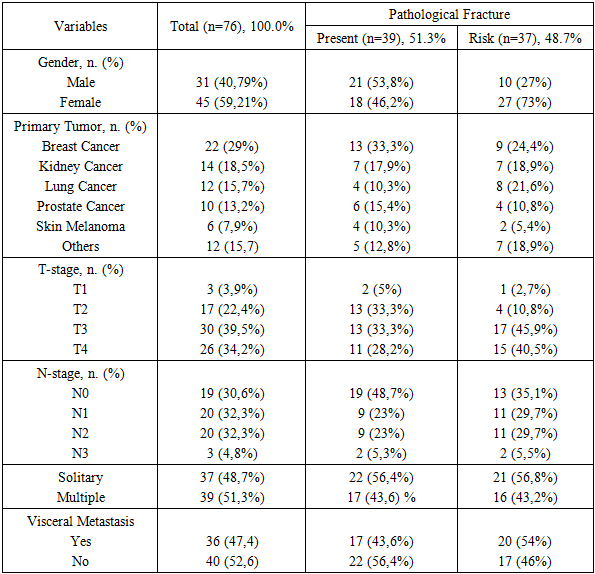
- Patients underwent various types of palliative treatment. Among them, 31 were men (40.79%) and 45 were women (59.21%). The average age of the patients was 57.5 years (ranging from 25 to 76 years). The majority of patients, 68 (89.5%), presented with complaints of pain syndrome of varying severity, while only 8 (10.5%) patients did not report any pain in the area of the metastatic bone lesion. The patients in the overall study group were divided into two groups based on the presence or absence of pathological fractures. Pathological fractures were observed in 39 (51.3%) of the 76 patients, while the remaining patients were assessed for fracture risk (Table 1). Breast cancer is the most common primary cancer that metastasizes to bone (29%). Following that, kidney cancer, lung cancer, and prostate cancer are the most common diseases causing bone damage. Despite the fact that metastatic bone lesions are more common in women (59.21%), pathological fractures are more frequent in men (53.8%) [7].
|
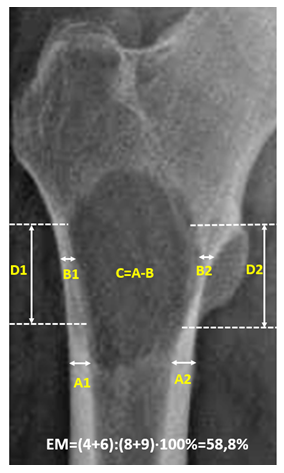 | Figure 2. Osteolytic type of metastatic involvement in the proximal region of the right femur (standard radiograph: high risk of pathological fracture, with cortical involvement greater than 50%) |
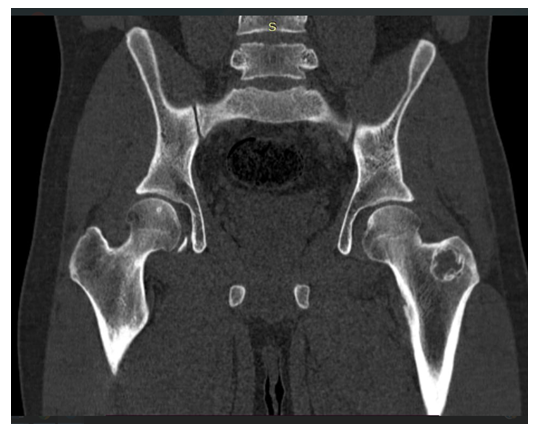 | Figure 3. Metastatic involvement of the greater trochanter of the left femur (computed tomography: low risk of pathological fracture, with cortical involvement less than 50%) |
4. Discussion
- Currently, in clinical practice, the assessment of the actual extent of tumor spread and the risk of pathological fractures in patients with metastatic bone disease (MBD) is carried out empirically based on visual examination results. Accordingly, accurate prediction of clinical outcomes and fracture risk in patients with metastatic involvement of long bones using highly prognostic factors can help prevent both over-treatment and under-treatment. Research findings have confirmed this, and the use of this mathematical model holds significant practical value in evaluating the precise level of bone involvement by the metastatic tumor, fracture risk, and the correct determination of treatment methods. Patients at high risk for fractures due to metastatic involvement of long bones require timely preventive osteosynthesis and, in some cases, therapeutic resection [10]. Modern approaches to treating metastatic cancer have expanded the use of external beam radiation therapy, chemotherapy, immuno-oncology and targeted therapies, hormonal therapy, or drugs that inhibit bone resorption in patients with low fracture risk [8,9]. However, in many cases, it is quite difficult to distinguish between low- and high-risk lesions based on clinical and radiological manifestations of bone metastases.According to clinical studies, patients with low risk of pathological fractures should be considered for single-fraction radiation therapy (8Gy) or therapeutic resection with good overall functional status (ECOG-PS), whereas patients at high risk should undergo multifractional radiation therapy with a high cumulative dose or stabilizing operations (Eggermont F, van der Linden Y, Verdonschot N, et al. A Patient-Specific Fracture Risk Assessment Tool for Femoral Bone Metastases: Using the Bone Strength (BOS) Score in Clinical Practice. Cancers (Basel). 2022;14(23):5904. Published 2022 Nov 29. doi:10.3390/cancers14235904).Currently, modern digital technologies and artificial intelligence programs are employed to help identify the mechanical, biological, and chemical properties of bone in patients with MBD [11]. The mechanistic model we recommend is an additional diagnostic strategy that is simple, easy to implement, and cost-effective, providing practical support to clinicians and radiologists in managing these patients [12]. Additionally, in certain cases, it becomes challenging to visually assess the level of bone involvement in osteoblastic and mixed bone metastases, which can, to some extent, be used to implement a general bone strength indicator. The density of osteoblastic metastases on CT is higher than the actual bone density, which can reduce the reliability of prognostic models when assessing the risk of pathological fractures in patients in this cohort. There are instances when, despite the large size of the metastatic tumor, the cortical portion of the bone may be minimally affected, allowing patients to avoid unnecessary surgical treatment. According to the developed formula, the higher the cortical involvement index, the higher the risk of pathological fractures, and the lower the index, the lower the risk.
5. Conclusions
- The fundamental principles of treatment for metastatic bone involvement (MBI) include pain management, the maintenance or improvement of quality of life, early mobilization, the creation of a stable orthopedic structure for bone replacement or augmentation, and the prevention of disease progression, when possible. In most cases, surgical intervention is typically recommended to prevent impending fractures or to stabilize pathological fractures. However, the performance of high-tech surgeries and the use of groundbreaking medications can yield favorable oncological outcomes in the treatment of patients with MBI. Therefore, a delicate balance is required to avoid both excessive and insufficient treatment of these lesions, particularly in procedures that require longer recovery times or result in higher morbidity. Pathological bone fractures due to metastatic involvement lead to a rapid decline in patients' functional status and a sharp reduction in survival. Consequently, it is crucial to assess the risk of pathological bone fractures and promptly apply the necessary therapeutic measures. In cases of tumors with high or moderate fracture risk, it is important to assess the feasibility of preventive and, in some instances, therapeutic surgical options. The mathematical model developed in this study can provide significant practical assistance in making the right decisions in complex clinical situations that arise in patients with MBI.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML