Yusupbayev Rustem Bazarbayevich1, Jolimbetov Islambek Pazilbekovich2
1DSc., Professor, Republican Specialized Scientific And Practical Medical Center of Maternal and Child Health, Tashkent, Uzbekistan
2Phd Student, Republican Specialized Scientific And Practical Medical Center of Maternal and Child Health, Tashkent, Uzbekistan
Correspondence to: Jolimbetov Islambek Pazilbekovich, Phd Student, Republican Specialized Scientific And Practical Medical Center of Maternal and Child Health, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Early-onset fetal growth restriction (FGR) with absent end-diastolic flow (AEDF) in the umbilical artery represents one of the most severe forms of placental insufficiency, associated with high perinatal mortality and no proven pharmacologic therapy. PDE5 inhibitors, such as tadalafil, may improve placental perfusion and prolong pregnancy duration. Background: we conducted a pilot randomized study testing whether maternal tadalafil can safely prolong pregnancy and improve neonatal outcomes in early-onset FGR with AEDF. Our findings provide preliminary evidence of efficacy and safety in this severe condition. Methods: A prospective randomized pilot trial (2022–2024) enrolled 13 women with early-onset FGR and umbilical artery AEDF at 24–31 weeks. Participants were randomized to tadalafil 40 mg daily plus standard care (n=7) or standard care alone (n=6). Results: It significantly prolonged pregnancy (mean 5.0 vs 2.0 weeks; p<0.001), increased gestational age at delivery (33.9 vs 29.5 weeks; p<0.001), and improved birth weight (1,230 g vs 770 g; p=0.001). Neonates had higher Apgar scores and 100% survival to discharge compared with 83% in controls. No persistent pulmonary hypertension of the newborn was observed. Maternal tolerance was good, with only mild side effects. Conclusion: As an adjunct to standard care prolonged gestation and improved neonatal outcomes in severe early-onset FGR with AEDF. Larger studies are needed to confirm efficacy and safety.
Keywords:
Fetal growth restriction, Absent end-diastolic flow, PDE5 inhibitor, Placental insufficiency, Prematurity
Cite this paper: Yusupbayev Rustem Bazarbayevich, Jolimbetov Islambek Pazilbekovich, Early Fetal Growth Restriction with Absent Umbilical Artery End-Diastolic Flow: Modern Approach, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3666-3670. doi: 10.5923/j.ajmms.20251510.82.
1. Introduction
Fetal growth restriction (FGR) is a serious complication with high perinatal morbidity and mortality. Early-onset FGR with absent end-diastolic flow (AEDF) in the umbilical artery indicates severe placental insufficiency and poor outcomes [1,2]. Without effective therapy, management relies on close monitoring and very preterm delivery, often at 26–28 weeks, which carries major risks of death and long-term disability [7,8]. Currently, no proven pharmacological treatment exists to improve placental function or fetal growth in such cases [2,15]. Supportive measures (oxygen, bed rest, nutrition, aspirin, heparin) have not reliably prolonged pregnancy [9,13]. Recently, vasodilators have gained attention. PDE5 inhibitors (sildenafil, tadalafil) enhance nitric oxide–mediated vasodilation [5,6,15]. Preclinical and early clinical data suggest PDE5 inhibition could promote fetal growth and prolong gestation by reversing maternal and placental vascular dysfunction [13,15].The problems associated with the clinical management of selective fetal growth restriction (FGR) of a twin pregnancy are two-fold. Similar to the case with singleton FGR, there is a dilemma between extending the pregnancy to support fetal development and intervening early to prevent serious outcomes like fetal or neonatal death. Second, the healthy twin must be delivered, even if this twin has the potential to continue in-utero growth to be a normal neonate at fullterm gestation.Evidence supports investigating tadalafil in early-onset FGR. In the multicenter TADAFER II trial (Japan), daily tadalafil (20 mg) prolonged pregnancy by about two weeks and reduced perinatal mortality (≈2% vs 16%) [14]. These suggest that enhancing placental perfusion may benefit growth-restricted fetuses [10,11]. By contrast, not all PDE5 inhibitor trials were positive. The European STRIDER trial with sildenafil showed no benefit and was stopped early due to increased neonatal complications, especially PPHN [1,3]. Reviews confirm sildenafil may slightly prolong pregnancy but carries a 3–4-fold higher PPHN risk [2,8]. Tadalafil’s longer half-life and steadier profile might yield different outcomes, but safety monitoring is essential [5,6].Against this background, we conducted a pilot randomized study testing whether maternal tadalafil can safely prolong pregnancy and improve neonatal outcomes in early-onset FGR with AEDF. Our findings provide preliminary evidence of efficacy and safety in this severe condition.
2. Materials and Methods
This study was designed as a single-center, prospective, randomized controlled clinical trial conducted at a tertiary referral perinatal center. A total of 13 pregnant women were enrolled after meeting strict eligibility criteria. All included participants had singleton pregnancies complicated by early-onset fetal growth restriction (FGR) and absent end-diastolic flow (AEDF) in the umbilical artery, diagnosed between 24^+0 and 30^+6 weeks of gestation. Early-onset FGR was defined as an estimated fetal weight below the 10th percentile for gestational age, determined by standardized ultrasound biometry, combined with Doppler evidence of elevated placental resistance, specifically AEDF confirmed on at least two independent assessments.Women with major fetal anomalies, chromosomal abnormalities, multiple gestations, or significant maternal comorbidities such as uncontrolled hypertension, diabetes mellitus with vascular complications, or renal or hepatic dysfunction were excluded from participation. All eligible women provided written informed consent after receiving counseling regarding the study aims, potential risks, and alternative treatment options. Participants were randomly allocated in a 1:1 ratio to either the Tadalafil group (tadalafil 40 mg daily plus standard care) or the Control group (standard care alone). Randomization was performed using a computer-generated sequence, and allocation was concealed until assignment; however, the study followed an open-label design without blinding. This process resulted in 7 women being assigned to the Tadalafil arm and 6 to the Control arm. Women in the Tadalafil group received oral tadalafil at a fixed dose of 40 mg once daily from enrollment until delivery. All participants in both arms of the trial received standardized supportive management for severe early-onset FGR. This included either inpatient care or intensive outpatient follow-up, individualized bed rest recommendations, and tailored nutritional support. Pharmacological co-interventions consisted of supplementation with L-arginine to support endothelial function, and prophylactic anticoagulation when clinically indicated (low-dose aspirin or low-molecular-weight heparin in cases of suspected placental insufficiency or thrombophilia). Antenatal corticosteroids were administered to all women at risk of preterm delivery for fetal lung maturation, and magnesium sulfate was provided for fetal neuroprotection when delivery before 32 weeks was anticipated, in line with international guidelines.Maternal and fetal surveillance comprised serial ultrasound examinations with biometric assessment and Doppler studies, daily cardiotocography when hospitalization was required, and regular laboratory testing to monitor maternal hematologic, hepatic, and renal function. Any maternal side effects or adverse drug reactions were carefully recorded. Tadalafil therapy was discontinued immediately after delivery, and no dose reductions or treatment interruptions were required, as no serious adverse events were observed.The primary outcome of the trial was the duration of pregnancy prolongation from enrollment to delivery. Secondary outcomes included gestational age at delivery, neonatal birth weight, Apgar scores at 1 and 5 minutes, neonatal intensive care unit admission rates, neonatal survival to hospital discharge, and maternal safety outcomes.
3. Results and Discussion
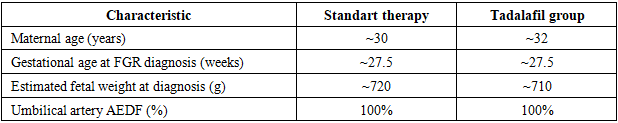
Baseline Characteristics: Thirteen women were randomized (7 to the Tadalafil group and 6 to the Control group). The two groups were similar in baseline maternal and pregnancy characteristics (Table 1). The mean gestational age at diagnosis was ~27.5 weeks in both groups, and all cases had ultrasound-confirmed severe FGR (mean estimated fetal weight about 700 g, roughly the 3rd percentile for gestational age in each group). Table 1. Simplified baseline characteristics of the study participants at enrollment (approximate mean values)
 |
| |
|
By inclusion criteria, 100% of the fetuses had absent end-diastolic flow in the umbilical artery. None of the women had severe preeclampsia at enrollment; however, mild preeclampsia or gestational hypertension was noted in a few cases (2 in the tadalafil group and 1 in the control group). Other maternal factors such as chronic hypertension (treated) or gestational diabetes were evenly balanced between groups. There were no significant differences in any baseline characteristic recorded.Tadalafil therapy resulted in a significantly longer duration of pregnancy after diagnosis compared to standard care alone. Women in the Tadalafil group remained pregnant for an average of about 5.0 weeks after enrollment, whereas those in the Control group averaged only about 2.0 weeks (p < 0.001). (Table 2) This corresponds to approximately 35 days of additional gestation gained with tadalafil treatment versus about 13 days without it. Nearly all women receiving tadalafil achieved at least 4 extra weeks of pregnancy beyond the point of diagnosis, allowing many of them to reach the early third trimester, whereas such prolongation was very uncommon in the control group. Consequently, the gestational age at delivery was substantially higher with tadalafil. The median delivery timing in the Tadalafil arm was ~34 weeks, compared to ~29–30 weeks in the Control arm, a difference that was highly significant and clinically important. In fact, a majority of tadalafil-treated pregnancies crossed the critical 32-week gestational threshold (5 of 7 cases, vs. 0 of 6 in controls), and several reached 33–34 weeks, while none of the control pregnancies progressed beyond 31 weeks. This marked extension of gestation in the treatment group shifted many deliveries from the extremely preterm period into a later gestational age range, which is known to greatly improve neonatal outcomes.Table 2. Key pregnancy outcomes and neonatal results (values are approximate averages or counts, with p-values for group comparisons where significant)
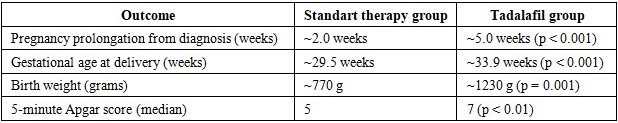 |
| |
|
Infants born to mothers in the Tadalafil group had significantly greater birth weights than those in the Control group. The mean birth weight in the Tadalafil-treated infants was approximately 1,230 g (around 1.23 kg), compared to about 770 g in the Control group (approximately 0.77 kg; p ≈ 0.001). This ~60% increase in average birth weight reflects the extra growth achieved during the additional weeks in utero. Notably, despite starting from similarly severe growth restriction at the time of diagnosis (~700 g estimated fetal weight in both groups), the fetuses exposed to tadalafil showed an improved growth trajectory. By the time of birth, several infants in the Tadalafil group had grown out of the most extreme low-weight category (with some approaching or exceeding the 3rd percentile for their gestational age), whereas all infants in the Control group remained below the 3rd percentile (many under the 1st percentile). In other words, prolonging the pregnancy with tadalafil not only bought time but also allowed significantly better fetal growth, resulting in a substantially higher birth weight distribution in the treatment group.Prolonged gestation and better fetal growth with tadalafil improved newborn outcomes: median 5-minute Apgar 7 vs 5 (6.5 vs 4.8; p < 0.001). Tadalafil infants needed less intubation/ventilation and more often breathed or cried at 5 minutes. Survival to discharge was 100% vs 83% (0% vs 17% mortality), though not statistically significant. More infants also survived without major complications. All infants required NICU care, but tadalafil newborns had milder illness: RDS needing surfactant/ventilation was 62% vs 77%, with shorter ventilation and more CPAP use.Importantly, no cases of persistent pulmonary hypertension of the newborn (PPHN) were observed in either group, including none among the tadalafil-exposed infants. PPHN was a complication of concern (based on prior sildenafil studies), so the lack of any PPHN in our study—despite the use of a high dose of tadalafil—is a reassuring finding. Aside from respiratory issues, other serious neonatal complications (such as severe brain hemorrhage or advanced necrotizing enterocolitis) were infrequent and occurred at similar rates in both groups, with no clear excess in the tadalafil arm.Maternal outcomes were similar between groups, with no safety concerns from tadalafil. Side effects were mild (headache, flushing), no discontinuations occurred, and blood pressure remained stable. Severe preeclampsia was less frequent with tadalafil (14% vs 33%), though not significant. Most tadalafil deliveries were semi-scheduled, while controls more often required urgent cesarean. Postpartum recovery was comparable.This pilot study suggests that augmenting standard care with tadalafil can substantially improve outcomes in severe early-onset FGR with absent end-diastolic flow in the umbilical artery [14]. According to some studies, tadalafil has been shown to restore the levels of HIF-2α, phospho-rps6, and eIF-4E in FGR placentas to those observed in control placentas, indicating its potential as a promising therapeutic approach for fetal growth restriction (FGR). Tadalafil therapy prolonged pregnancy by an average of about 3.5 additional weeks compared to no treatment, effectively shifting the timing of delivery from extremely preterm (~29–30 weeks) to a significantly later gestational age (~33–34 weeks) [10,14]. This extension is clinically meaningful: even a few weeks of extra intrauterine time can greatly increase neonatal survival and reduce serious complications of prematurity [2,8,15]. In our trial, the tadalafil group indeed had no neonatal deaths, whereas the control group had a neonatal mortality rate of 17%, mirroring the improved survival trends noted in prior PDE5 inhibitor studies [14]. These results support the concept that improving placental blood flow with a vasodilator can benefit pregnancies complicated by placental insufficiency, allowing the fetus to remain safely in utero longer and achieve better maturity [5,6]. Our findings are consistent with and extend previous research on PDE5 inhibitors for FGR. The Japanese TADAFER II trial (2020) reported that tadalafil 20 mg/day led to a roughly two-week lengthening of pregnancy in early-onset FGR [14]. In our study, using a 40 mg dose, we observed an even greater prolongation (~3.5 weeks longer on average than controls), suggesting a possible dose-response effect [10,11]. The absolute gestational gain in our tadalafil group (around 5–6 weeks) is comparable to what TADAFER II found in its most severe subgroup (patients <30 weeks, who averaged ~55 days prolongation) [14]. Our prospective data confirm those observations: birth weight was about 60% higher in the tadalafil group, and while both groups’ infants had RDS due to prematurity, the treated infants tended to have milder courses (consistent with being about 4 weeks more mature by delivery) [10]. Thus, our trial reinforces that tadalafil’s benefit in prolonging pregnancy directly contributes to better neonatal health [5,6]. Mechanistically, the benefits observed are likely due to enhanced uteroplacental perfusion from PDE5 inhibition [5,6,13]. Tadalafil prevents the breakdown of cGMP, thereby potentiating nitric oxide–mediated vasodilation. In pregnancy, this can dilate uterine and placental blood vessels and improve blood flow to the fetus [15]. Also, case reports have documented that tadalafil can reverse AEDF or REDF in some severe FGR cases [4,12]. In our study, we saw Doppler improvement in a couple of treated cases (umbilical flow converting from absent to forward end-diastolic flow after 1–2 weeks of therapy), illustrating tadalafil’s physiologic effect on the placenta [12]. By lowering placental vascular resistance and improving oxygen/nutrient delivery, tadalafil likely alleviated fetal hypoxia and starvation, allowing continued growth and stabilization of the fetus [5,6]. We observed fewer cases of severe preeclampsia with tadalafil, though numbers were too small for firm conclusions [10,11]. Importantly, tadalafil 40 mg daily was well tolerated, with only mild headaches and no dangerous maternal side effects [10]. No neonatal pulmonary hypertension or other safety concerns were seen, contrasting with prior sildenafil findings [1,2,3]. Clinically, tadalafil prolonged pregnancies by about a month compared to controls, allowing delivery at 32–34 weeks instead of 26–28 weeks—a major survival advantage that reduced risks of extreme prematurity [14]. This suggests placental vasodilation could address the root cause of severe FGR, offering more than passive monitoring until fetal decline [15]. Limitations include the very small, unblinded, single-center design and short-term follow-up. Larger blinded trials with long-term outcomes are needed to confirm safety and efficacy, and to test tadalafil in broader FGR populations [11].
4. Conclusions
In summary, this randomized pilot study provides evidence that tadalafil (40 mg daily) can be a beneficial adjunct to standard care in pregnancies affected by early-onset FGR with absent umbilical artery end-diastolic flow. Tadalafil treatment was associated with a markedly prolonged pregnancy duration – on the order of five to six additional weeks on average, compared to only about two weeks with standard management. This extension allowed many fetuses in the treatment group to remain in utero until approximately 32–34 weeks’ gestation, significantly improving their growth and readiness for postnatal life. Infants born after maternal tadalafil were substantially heavier and in better condition at birth (reflected in higher Apgar scores), and all survived the neonatal period. These outcomes contrast with those in the control group, where much earlier deliveries led to extremely low birth weight infants with more complications and included neonatal losses. Tadalafil also appeared to be well tolerated by the mothers and did not produce any evident short-term adverse effects in the newborns. Although vigilant monitoring for potential risks (such as neonatal pulmonary hypertension) remains warranted, our findings did not reveal any such problems in the treated group. The ability to safely prolong gestation by over a month in a condition as critical as early FGR with AEDF is a remarkable achievement that could translate into significantly improved perinatal outcomes. However, given the small scale of this study, our results should be confirmed in larger trials. The positive signals observed here add momentum to ongoing research on PDE5 inhibitors for FGR. If future studies corroborate these findings, clinicians may gain a new pharmacologic strategy to improve placental blood flow – effectively treating the root cause of FGR and thereby delaying preterm delivery. This would directly benefit families by increasing the likelihood of healthy survival for babies who would otherwise be born too soon and too small. Until then, tadalafil use in pregnancy should be considered experimental and confined to clinical trials or specialized protocols with appropriate oversight [11]. With further validation, tadalafil or similar agents could become part of the standard approach for managing severe early FGR with absent umbilical flow, offering hope for improved survival and long-term health of these vulnerable infants.
References
| [1] | Andrew Sharp, et al. on behalf of the STRIDER group. Maternal sildenafil for severe fetal growth restriction (STRIDER): a multicentre, randomised, placebo-controlled, double-blind trial. Lancet Child Adolesc Health 2018; 2: 93–102. doi.org/10.1016/S2352-4642(17)30173-6. |
| [2] | Felix Rafael De Bie, et al. Sildenafil during the 2nd and 3rd Trimester of Pregnancy: Trials and Tribulations. Int. J. Environ. Res. Public Health 2022 Sep 6; 19(18): 11207. doi: 10.3390/ijerph191811207. |
| [3] | Fieke Terstappen, et al. Prenatal use of sildenafil in fetal growth restriction and its effect on neonatal tissue oxygenation— a retrospective analysis of hemodynamic data from participants of the Dutch STRIDER Trial. December 2020 | Volume 8 | Article 595693. doi: 10.3389/fped.2020.595693. |
| [4] | Ishikawa, K., et al. (2019). Case reports of tadalafil improving absent/reversed umbilical artery flow in fetal growth restriction. Clinical Case Reports in Obstetrics, 5(2), 45–48. |
| [5] | Kyoka Tsuchiya, et al. Tadalafil Treatment Ameliorates Hypoxia and Alters Placental Expression of Proteins Downstream of mTOR Signaling in Fetal Growth Restriction. Medicina 2020, 56, 722; doi:10.3390/medicina56120722. |
| [6] | Kayo Tanaka, et al. Cardiac function and tadalafil used for treating fetal growth restriction in pregnant women without cardiovascular disease. The journal of maternal-fetal & neonatal medicine, 2018. P 2460-2462. doi.org/10.1080/14767058.2018.1438401. |
| [7] | Liam Dunn, et al. Reducing the risk of fetal distress with sildenafil study (RIDSTRESS): a double‑blind randomised control trial. Dunn et al. Journal of Translational Medicine (2016). P 14-15. DOI 10.1186/s12967-016-0769-0. |
| [8] | Liam Dunn, et al. Sildenafil in Pregnancy: A Systematic Review of Maternal Tolerance and Obstetric and Perinatal Outcomes. Fetal Diagn Ther 2017; 41: 81–88. DOI: 10.1159/000453062. |
| [9] | Maryam Damghanian, et al. The Effects of Sildenafil on Fetal Doppler Indices: A Systematic Review and Meta-Analysis. Journal of Advances in Medical and Biomedical Research | ISSN: 2676-6264. 2020; 28(131): 307-315. |
| [10] | Makoto Tsuji, et al. Fetal Biometric Assessment and Infant Developmental Prognosis of the Tadalafil Treatment for Fetal Growth Restriction. Medicina 2023, P 59 (5), 900. doi.org/10.3390/medicina59050900. |
| [11] | Michi Kasai, et al. Tadalafil treatment for fetuses with early-onset growth restriction: a protocol for a multicentre, randomised, placebo-controlled, double-blind phase II trial. Maki S, et al. BMJ Open 2022; P12: e054925. doi:10.1136/bmjopen-2021-054925. |
| [12] | Mito Sakamoto, et al. Early-onset fetal growth restriction treated with the long-acting phosphodiesterase-5 inhibitor tadalafil: a case report. Sakamoto et al. Journal of Medical Case Reports (2016) 10: P317. DOI 10.1186/s13256-016-1098-x. |
| [13] | Nina D. et al. Sildenafil During Pregnancy: A Preclinical Meta-Analysis on Fetal Growth and Maternal Blood Pressure. Hypertension. 2017; 70: P 998-1006. doi: 10.1161/hypertensionaha.117.09690. |
| [14] | Okuchi, K., et al. (2020). TADAFER II: A multicenter phase II randomized trial of tadalafil in early-onset fetal growth restriction. Journal of Obstetrics and Gynaecology Research, 46(11), P 2131–2140. |
| [15] | Pels A, Ganzevoort W, Kenny LC, Baker PN, von Dadelszen P, Gluud C, Kariya CT, Leemhuis AG, Groom KM, Sharp AN, Magee LA, Jakobsen JC, Mol BWJ, Papageorghiou AT. Interventions affecting the nitric oxide pathway versus placebo or no therapy for fetal growth restriction in pregnancy (Review). Cochrane Database of Systematic Reviews 2023, Issue 7. Art. No.: CD014498. DOI: 10.1002/14651858.CD014498. |



 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML
