-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3590-3592
doi:10.5923/j.ajmms.20251510.66
Received: Sep. 26, 2025; Accepted: Oct. 21, 2025; Published: Oct. 28, 2025

Dynamics of Cytokine Changes in the Treatment of Respiratory Allergies in Children
Matniyozova Zainab Tukhtaboevna
Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: Matniyozova Zainab Tukhtaboevna, Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study analyses cytokine and interleukin levels in children with respiratory allergies. Particular attention is paid to the problem of immunological disorders in children with allergic diseases. Serum levels of IL-8, IL-1β, anti-inflammatory IL-4 and IFNα production were examined in children with respiratory allergies (bronchial asthma, sinusitis). A significant decrease in IFNα levels and an increase in IL-6, IL-8 and IL-4 concentrations were found, which in turn confirms impaired immune reactivity and decreased local and systemic immunity in patients with respiratory allergies. The results obtained determine the need to use immunocorrective measures in the complex therapy of these patients.
Keywords: Cytokines, Respiratory allergies, Children
Cite this paper: Matniyozova Zainab Tukhtaboevna, Dynamics of Cytokine Changes in the Treatment of Respiratory Allergies in Children, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3590-3592. doi: 10.5923/j.ajmms.20251510.66.
1. Introduction
- Recent decades have been characterised by a steady increase in the prevalence and severity of allergic diseases, the pathogenesis of which involves immune system cells, regulatory molecules (cytokines and chemokines) and corresponding receptors. An imbalance in the levels of cytokines involved in allergic inflammation plays an important role in the pathogenesis of respiratory allergies in children. In particular, impaired production of interferon alpha leads to recurrence of respiratory allergies [3,7]. The effector phase of immediate-type allergic reactions involves mast cells, basophils, eosinophils, neutrophils, endothelial and epithelial cells, and platelets, whose activation triggers the synthesis and secretion of a number of cytokines: interleukins-1, 6, 8, 12 (IL-1, 6, 8, 12), tumour necrosis factor-a (TNFa) and gamma interferon (IFNy) [6]. The local effect of cytokines in allergic reactions initiates inflammation, determines increased permeability, vasodilation, exudate accumulation, induces the expression of adhesion molecules on endothelial cells, and promotes the migration of leukocytes into tissues. They regulate cell interactions in allergic reactions, ensure the maturation of the precursors of the main cells involved in allergic inflammation, their proliferation, differentiation, activation, and mobilisation to the site of the allergic reaction, and participate in the regulation of the form and strength of the specific response to the allergen. Allergic diseases are classified as immune-mediated diseases. They are characterised by impaired immune regulation, chronic inflammation and tissue damage [8]. Despite advances in immunology, the cytokine mechanisms of allergic diseases are still not fully understood, and further research is needed on the role of monocytes and cytokines in the pathogenesis of allergic inflammation [1,4,9].Interleukin (IL) 1 is produced mainly by activated macrophages, sometimes by epithelial, endothelial, glial cells, fibroblasts, and keratinocytes. There are two known forms of IL-1: alpha and beta. An increase in the level of IL-1 beta in blood serum is an extremely important indicator in diseases based on inflammation. During exacerbations of bronchial asthma and other allergic reactions, the production of IL-1 beta by peripheral blood mononuclear cells increases. In various infectious diseases, the severity of the process correlates with the level of IL-1 beta production [2,5].IL-4 (B-cell stimulating factor) is produced by activated type 2 T-helper cells. Its main function is to switch immunoglobulin (Ig) G1 synthesis to IgG4 and IgE synthesis. It has been shown that the IL-4 content in the blood of children with allergic diseases correlates with clinical manifestations and IgE levels [10].Research objective: to study the cytokine profile in respiratory allergies in children.
2. Materials and Methods
- The study analysed systemic and local immunity indicators, cytokine and interleukin levels in 20 children with respiratory allergies, who constituted the main group, and 20 practically healthy children in the control group. Clinical, laboratory, and immunological indicators were measured, and local and systemic immune disorders against the background of respiratory allergies were assessed. Statistical data processing was performed using the Statistica software package with Student's t-test. IL-4, IL-6, IL-8, TNFa and IFNy in blood serum were determined using enzyme-linked immunosorbent assay according to the generally accepted method. General clinical research methods were used in this study, and medical records were analysed.
3. Results and Discussion
- In children in the main group, high levels of IL-8 (155.58±19.06 versus 23.50±1.9 pg/ml in the control group) and TNFa, low levels of IFNy (31.11±3.05 pg/ml) and no significant differences in serum IL-2 levels between the study and control groups. The rate and intensity of changes in serum IL-8 and TNFa concentrations reflected the degree of activity and severity of allergic inflammation. Changes in serum IL-4 and IFNy depending on the severity and period of allergic diseases were insignificant. When analysing the IL-6 content in the serum of children with bronchial asthma, it was noted that the serum IL-6 level in this pathology significantly exceeded that of healthy children (21.12±0.61 vs. 5.12±0.55 pg/ml). The worsening of asthma was accompanied by an increase in serum IL-6 levels, especially during periods of exacerbation (47.64±2.55 pg/ml). In children with mild clinical course during persistent remission, serum IL-6 levels did not exceed those of healthy peers (6.78±1.22 and 5.03±0.54 pg/ml, p>0.05). In mild cases, the increase in serum IL-6 levels was moderate (from 8.85±1.40 to 9.70±1.68 pg/ml, p>0.05). In moderate asthma during remission, the level of IL-6 in blood serum was recorded at 12.01±0.77 pg/ml. Activation of the inflammatory process at the time of exacerbation of the disease was accompanied by an increase in IL-6 content to 19.88±1.47 pg/ml.
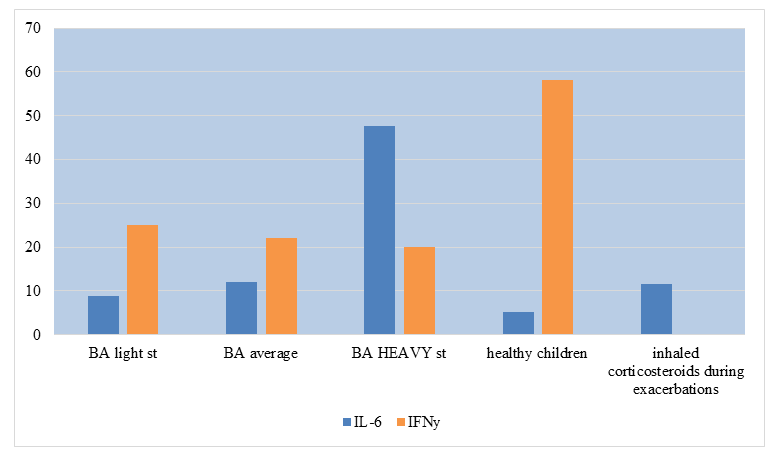 | Figure 1. Cytokine levels in bronchial asthma |
4. Conclusions
- The results of the study of IL-4, IL-8, TNFa, and INFy allow us to recommend these cytokines as markers of respiratory allergies, as immunological criteria for the effectiveness, adequacy, and timeliness of anti-inflammatory and immunocorrective therapy. The dynamics of cytokine content in blood serum reflects the current state of the immune system and local defence and, in combination with other indicators, has diagnostic value in determining the location, severity and degree of activity of allergic inflammation.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML