-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3587-3589
doi:10.5923/j.ajmms.20251510.65
Received: Sep. 22, 2025; Accepted: Oct. 21, 2025; Published: Oct. 28, 2025

Age-Related Features of the Manifestations and Diagnosis of Mycoplasma Pneumonia in Children
Yuldasheva G. G.1, Zhalilov Zh. A.2
1PhD, Associate Professor of Paediatrics, Bukhara State Medical Institute, Bukhara, Uzbekistan
2Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: Yuldasheva G. G., PhD, Associate Professor of Paediatrics, Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

M. pneumoniae is one of the most discussed respiratory problems in paediatrics. This is due to the widespread prevalence of mycoplasma infection in children and adolescents and diagnostic errors. The high probability of diagnostic errors when using clinical signs and the lack of an ideal laboratory diagnostic method often lead to the excessive prescription of macrolides and, as a result, to an increase in the resistance of respiratory infection pathogens to antimicrobial drugs. This article presents the results of clinical and laboratory studies of mycoplasma pneumonia in different age groups of children. It has been established that mycoplasma pneumonia most commonly occurs in young children, with no obvious clinical symptoms and a prolonged course.
Keywords: Mycoplasma pneumonia, Young children, Diagnosis, Immunoenzymatic method
Cite this paper: Yuldasheva G. G., Zhalilov Zh. A., Age-Related Features of the Manifestations and Diagnosis of Mycoplasma Pneumonia in Children, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3587-3589. doi: 10.5923/j.ajmms.20251510.65.
1. Introduction
- M. pneumoniae was first isolated in tissue culture from the sputum of a patient with primary atypical pneumonia. In 1961, it was proven that the ‘Eaton pathogen’ belongs to the Mycoplasma species. In 1963, Chanock succeeded in culturing the Eaton pathogen in a cell-free mammalian medium and proposed the taxonomic designation M. pneumoniae. M. pneumoniae is a unique organism that does not have a cell wall under any circumstances and does not need a host cell for replication. This organism causes a variety of clinical manifestations, ranging from self-limiting to life-threatening. The severity of the disease appears to depend on the degree of protection provided by the body [2,8]. M. pneumoniae accounts for 10.6–17.0% of community-acquired cases and 3.0–20.8% of hospital-acquired cases of community-acquired pneumonia, respectively, and the frequency of hospitalisation in intensive care units is relatively low (2–3.6%). Arnold et al. showed that M. pneumoniae is the most common causative agent of atypical pneumonia, accounting for 11–15% of community-acquired pneumonia cases worldwide [5].M. pneumoniae is an important causative agent of community-acquired pneumonia in children, but the epidemiology and clinical manifestations of M. pneumoniae infection in infants have been described in only a few studies. Family outbreaks and outbreaks in closed communities — in barracks, kindergartens, schools, and universities — are often described, lasting several months and recurring every 3–5 years, as prolonged contact creates conditions for the persistence of the pathogen [1,3,5]. Pneumonia caused by M. pneumoniae manifests itself in several ways, such as fever, cough, wheezing, diarrhoea, vomiting, and other non-specific symptoms. However, it is impossible to diagnose M. pneumoniae based solely on these signs and symptoms, and laboratory analysis is more important for detecting M. pneumoniae infection. Community-acquired M. pneumoniae epidemics are usually seen in school-aged children and young adults. However, a growing body of research indicates that M. pneumoniae infection is common in children aged 1 to 5 years and that the clinical characteristics differ between younger and older patients. M. pneumoniae infection in young children is receiving increasing attention. The most common problem faced by paediatricians is the difficulty of differentiating between mycoplasma and mild pneumococcal pneumonia in an outpatient setting [4,6]. For a long time, diagnosing mycoplasma infection was only possible in specialised laboratories due to the difficulties involved in culturing the pathogen that causes mycoplasmosis. The development and use of fundamentally new, modern, and highly sensitive detection methods, such as polymerase chain reaction (PCR) and enzyme-linked immunosorbent assay (ELISA), have contributed to progress in the study of this problem. However, the characteristics of the clinical manifestations of ‘atypical pneumonia’ in relation to the immunological indicators of the disease in young children have not been sufficiently studied [5,7,8].Research objective: to study the incidence and clinical and laboratory characteristics of M. pneumoniae among the paediatric population.
2. Materials and Methods
- A retrospective study of 124 medical records was conducted to examine the demographic, clinical, and microbiological characteristics of patients with lower respiratory tract infections (LRTI) admitted to the ODMPC during the period 2023-2024. Children were included in the study if they had the following symptoms: (a) fever, cough and shortness of breath (clinical data); abnormal breathing sounds, wheezing or moist rales (auscultatory data); and/or focal/segmental consolidation, interstitial changes or pleural effusion (radiographic data) confirming lower respiratory tract infections. IgM on admission or during follow-up, or ≥4-fold increase in IgG titre. The exclusion criteria were children with severe concomitant diseases, such as neoplasms, renal or hepatic dysfunction, immunosuppression, chronic lung or cardiovascular diseases, genetic or neurological disorders, and chronic metabolic diseases. Serum IgG and IgM levels for M. pneumoniae were determined using a commercial kit (Virion-Serion, Germany). The result was considered positive if the IgM titre was ≥1.1 or if the IgG titre exceeded 4 times the normal level.
3. Results and Discussion
- Of the 124 individuals examined, 24 were diagnosed with M. pneumoniae infection. The test results showed that 12 of them were aged 1 to 3 years; 9 were aged 3 to 6 years; and 3 were aged 6 to 9 years. M. pneumoniae infection mainly occurred in September-October. The presence of fever with a maximum temperature >39.0°C for ≥3 days was more common in the 1-3 age group (P< 0.05). Laboratory tests showed that infants aged 6-9 years had higher peripheral white blood cell counts (P = 0.04) and neutrophil counts (P = 0.02) and higher CRP levels (P = 0.05). Furthermore, the median length of hospitalisation for infants aged 1-3 years was shorter than in the other two groups.
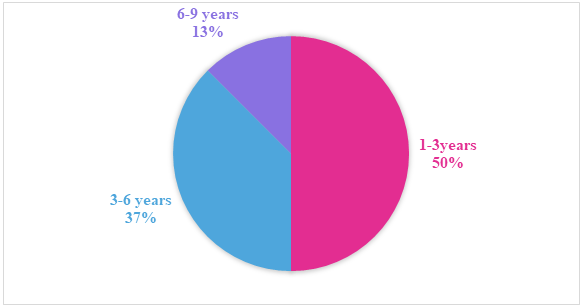 | Figure 1. Prevalence of mycoplasma pneumonia among the study group of children |
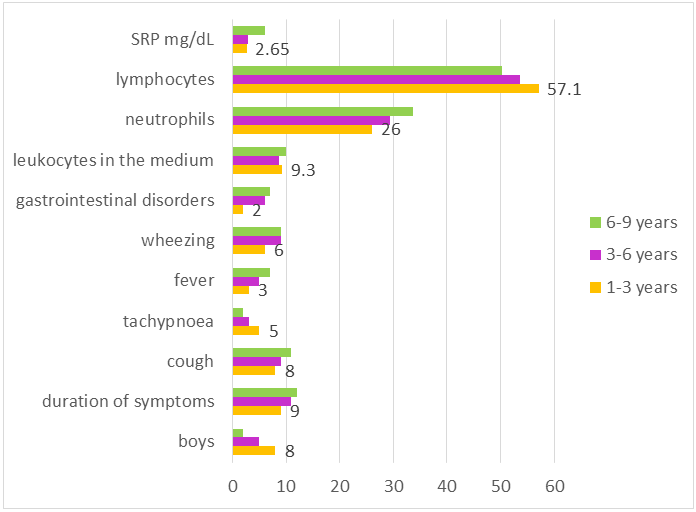 | Figure 2. Average clinical and laboratory parameters in the study groups |
4. Conclusions
- M. pneumoniae respiratory tract infection is characterised by mild clinical manifestations, the leading symptom of which is cough. Its diagnosis and treatment require non-standard, non-routine approaches. These features necessitate increased attention from specialists, careful collection of medical history, detailed physical examination, and knowledge necessary for the selection of antimicrobial therapy effective against both typical and atypical pathogens.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML