-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3568-3573
doi:10.5923/j.ajmms.20251510.61
Received: Sep. 19, 2025; Accepted: Oct. 17, 2025; Published: Oct. 28, 2025

Comprehensive Diagnostics of Cognitive and Emotional Disorders in Children and Adolescents with Type 1 Diabetes
Alidjanova Durdona Abdullajonovna
Doctor of Medical Sciences, Associate Professor, Department of Neurology, Pediatric Neurology, and Medical Genetics, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Alidjanova Durdona Abdullajonovna, Doctor of Medical Sciences, Associate Professor, Department of Neurology, Pediatric Neurology, and Medical Genetics, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This article contains data from the analysis of neuropsychological, laboratory, and neuroimaging examination methods related to the issue of cerebral complications in type 1 diabetes in children. It presents data from our own studies on psychological testing, methods for assessing situational (SA) and personal (PA) anxiety, as well as an analysis of the content of neuropeptides and major brain metabolites. According to the results obtained, it can be concluded that the proposed set of diagnostic measures using modern research methods in the diagnosis of cognitive and emotional changes at early stages of this pathology is justified and appropriate, as they provide a real opportunity to monitor changes in brain tissue and visualize brain metabolism deviations that lead to cerebral disorders at a stage when clinical manifestations are not yet observed.
Keywords: Cognitive deficit, Children and adolescents, Diabetes mellitus, Neuron-specific proteins, Proton magnetic resonance spectroscopy
Cite this paper: Alidjanova Durdona Abdullajonovna, Comprehensive Diagnostics of Cognitive and Emotional Disorders in Children and Adolescents with Type 1 Diabetes, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3568-3573. doi: 10.5923/j.ajmms.20251510.61.
1. Introduction
- Diabetes mellitus (DM) is classified as an autoimmune pathology of the endocrine system. The development of DM occurs due to an absolute or relative insulin deficiency, induced by the disintegration of pancreatic beta cells. Today, DM represents a serious problem for the global medical community, as it continues to rank among the leading causes of reduced quality of life and early disability due to long-term complications [1]. Among the wide range of complications of type 1 DM in children, changes in the brain take a special place, primarily manifesting as cognitive impairments (CIs). Close attention to cognitive function in diabetes is associated not only with the normal daily functioning and social adaptation of the child but also with regular adequate self-monitoring of glycemia, which directly affects the overall course of the primary disease [2]. Currently, the early diagnosis of cognitive impairment in patients with diabetes mellitus is one of the priority areas in modern neuropathology [3]. In the diagnosis of higher cortical function disorders, such as memory, attention, and thinking deficits, neuropsychological testing methods hold a dominant position. However, it must be taken into account that these methods still provide a subjective assessment, with both false-positive and false-negative results, and are unable to offer complete reliable information on all criteria of cognitive impairment [4]. Therefore, the issue of adequate and early diagnosis of these disorders, with objective criteria that most accurately indicate the localization of the pathological focus causing cognitive changes, is considered quite relevant not only for neurologists but also for specialists in other areas of medicine. According to this, the primary task today is to identify the risks of developing cognitive dysfunctions long before they arise. The development and implementation of a clear algorithm for predicting cognitive deficit at the stage when there are no clinical signs can address this task and guide specialists in prescribing appropriate preventive therapy, which, in turn, will help maintain higher cortical functions at a satisfactory level for a fairly long period [5].Neurospecific proteins are tissue-specific for the nervous system and, histo-genetically, are part of neurons and glial cells. In blood serum, low concentrations of these proteins are considered normal; this occurs due to the natural death of neurons, as well as pathological changes. Therefore, elevated levels of neurospecific proteins are regarded as markers of various pathological processes in the brains of adults and children, among which epilepsy, traumatic brain injury (TBI), diabetes, consequences of hypoxia, autism, and Parkinson's disease are somewhat better studied. Among the total number of known neurospecific proteins, the most studied are protein S-100 and neuron-specific enolase (NSE), which, according to scientists, provide greater information about the state of the pathological process and may be useful in studying the pathogenesis of neurological dysfunction, including in children and adolescents with type 1 diabetes [8].The use by specialists in practical activities of a method such as proton magnetic resonance spectroscopy (¹H-MRS) has provided a means for non-invasive and in vivo analysis of metabolic abnormalities occurring in the brain in various neurological pathologies. The basis of this neuroimaging technique is the so-called 'chemical shift' of the resonance frequencies of different chemical compounds. To date, two methods of magnetic resonance (MR) spectroscopy have been developed: single-voxel and multi-voxel spectroscopy. The first spectroscopy method provides a rapid analysis of the biochemical composition of a limited volume in the area of interest in the brain. The second method is based on the use of the so-called color mapping, which covers a fairly large area of the brain with a multi-voxel volume, allowing for the simultaneous focus on 16 to 64 voxels, including both the damaged brain tissue and the contralateral hemisphere, with demonstration at various anatomical levels [9]. The main metabolites studied in MR spectroscopy are N-acetylaspartate (NAA), choline (Cho), and creatine (Cr). In addition to assessing their absolute concentrations, their ratios are also determined, namely N-acetylaspartate to creatine (NAA/Cr); choline to creatine (Cho/Cr); and choline to N-acetylaspartate (Cho/NAA) [10]. According to experts working on cognitive disorders across various nosological entities, the sensitivity, specificity, and significant prognostic value for detecting cognitive deficits using MR spectroscopy are 89.8%, 88.2%, and 95.3%, respectively [11]. Numerous scientific studies confirm the high significance of the aforementioned methods in studying cognitive deviations associated with various diseases in both adults and children. However, despite this, there remain many questions that require more precise definitions and clarifications. This circumstance primarily concerns childhood and adolescence, when the issue of early diagnosis of cognitive disorders is particularly acute and of vital importance [12].Research objective: To study the role of biomarkers in the earliest diagnosis of cognitive deficit in children and adolescents with type 1 diabetes.
2. Materials and Methods
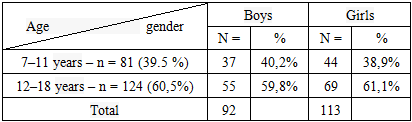
- An examination was conducted on 205 children aged 7 to 18 years with type 1 diabetes mellitus (T1DM), including 92 boys (45%) and 113 girls (55%). Children aged 7 to 11 years (mean age 9.0±1.6 years) accounted for 81 patients (39.5%), while 124 patients aged 12 to 18 years (mean age 14.7±1.8 years) were examined (60.5%) (Table 1).
|
3. Results and Discussion
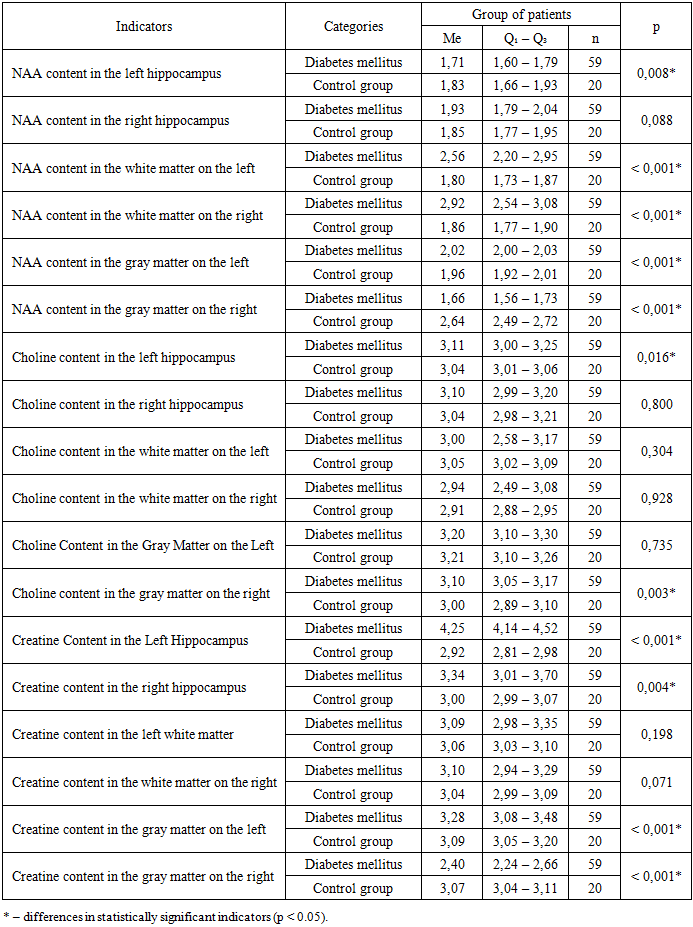
- In the comparative assessment of HbA1c values considering the duration of the disease, we found that in the first 1.5–2 years from the onset of diabetes, glycated hemoglobin levels were significantly lower, averaging 8.5%. In the group with a disease duration of 3 to 6 years, or more precisely by the middle of the third year of diabetes, carbohydrate metabolism indicators worsened, and their average values were significantly higher than in children with a disease duration of up to 3 years – 9.4% (p < 0.001). In patients with a longer duration of diabetes, the trend toward worsening HbA1c levels persisted and was on average significantly higher – 10.4% (p < 0.001) than in groups with shorter disease duration.During the analysis of individual tasks of the MoCA test, it was determined that patients with type 1 diabetes performed significantly worse on the 'clock' (p<0.001), 'attention' (p<0.001), 'phrase repetition' (p<0.001), and 'delayed recall' (p<0.001) tasks compared to the control group. The anxiety test results demonstrated the presence of moderate situational anxiety in 120 (58.5%) patients and high situational anxiety in 85 (41.4%) children. Regarding trait anxiety, it was detected at a low level in 4 (2%) patients, at a moderate level in 70 (34%) cases, and at a high level in 131 (64%) patients. Anxiety testing results, taking into account the duration of type 1 diabetes, showed that a moderate level of situational anxiety in the group of children with a disease duration of less than 3 years was observed in 62.8% of cases, with a duration of 3 to 6 years in 49.3% of children, and for more than 6 years in 63.3% of patients. A high level of stress (ST) in patients with a disease duration of less than 3 years was observed in 37.2% of cases, from 3 to 6 years in 50.7% of children, and in the group with a duration of more than 6 years in 36.7% of patients. According to the personal anxiety scale, a moderate level in children with a disease duration of less than 3 years was determined in 37.2% of episodes, from 3 to 6 years in 28.4% of patients, and in those with a longer period of pathology in 36.7% of children. A high degree of personal anxiety in this category of patients was identified in 60.3%, 70.1%, and 61.7% of cases, respectively (Fig. 1).
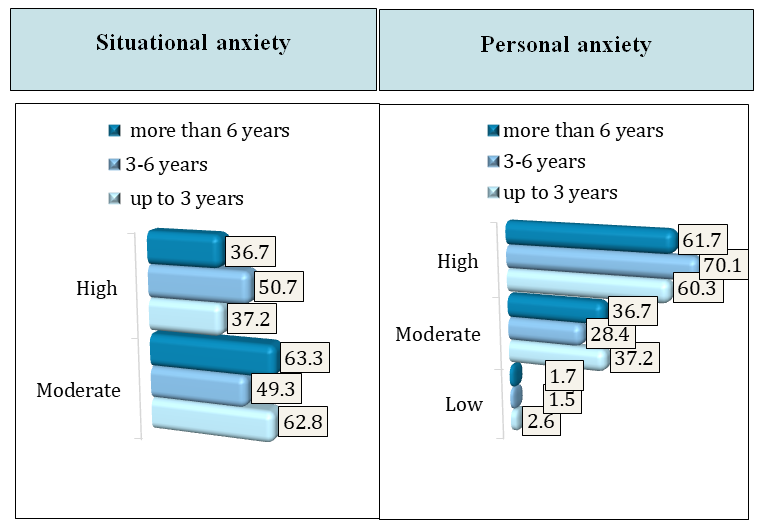 | Figure 1. Levels of situational and personal anxiety in children and adolescents with type 1 diabetes |
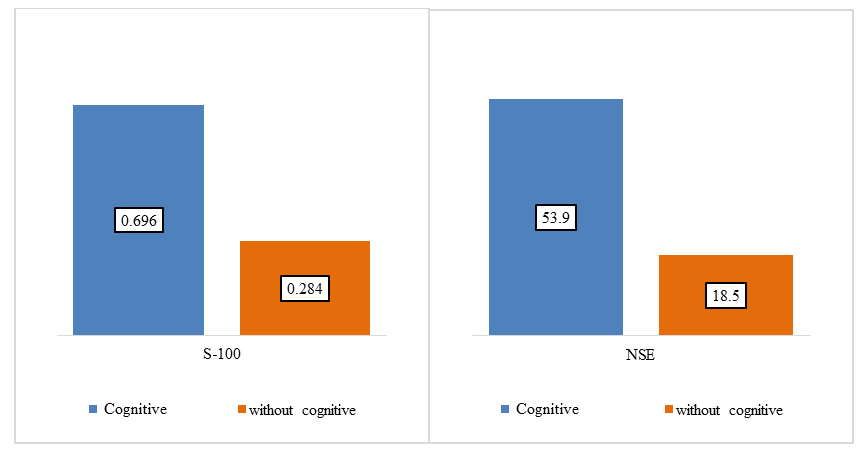 | Figure 2. Analysis of S-100 and NSE proteins depending on the presence of cognitive impairments |
|
4. Conclusions
- Conducting PMRS studies and determining the concentration of neuron-specific proteins made it possible to localize areas of the brain responsible for decreased cognitive and emotional functions in children and adolescents with type 1 diabetes. In addition, it allowed for obtaining more precise information on metabolic changes in these brain regions. Overall, these methods enabled the dynamic assessment of patients with type 1 diabetes, the detection of cerebral changes at a stage when clinical manifestations are not yet present, as well as the monitoring of potential disease progression and the adequacy of the selected therapy to correct higher cortical function deviations.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML