-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3551-3555
doi:10.5923/j.ajmms.20251510.57
Received: Sep. 21, 2025; Accepted: Oct. 16, 2025; Published: Oct. 28, 2025

Association of CALCA and ACE Gene Polymorphisms with Migraine Susceptibility: A Clinical-Genetic Study in a Central Asian Cohort
Xolmatov R. I., Abdukadirov U. T., Kasimova S. A., Abbosova I. A., Sotvoldiyev M. M.
Department of Neurology, Andijan State Medical Institute, Andijan, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

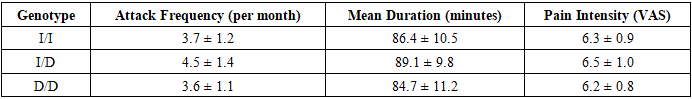
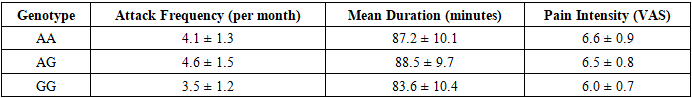
Background: Migraine is a complex neurovascular disorder influenced by genetic and environmental factors. Among candidate genes, CALCA (encoding calcitonin gene-related peptide, CGRP) and ACE (angiotensin-converting enzyme) are implicated in vascular tone regulation and pain sensitization. Objective: To investigate the association of CALCA (A>G) and ACE (I/D) polymorphisms with migraine clinical characteristics in a Central Asian population. Methods: A cross-sectional study was conducted on 60 clinically confirmed migraine patients (47 females, 13 males; mean age 34.4 ± 1.02 years). Genotyping was performed using PCR and agarose gel electrophoresis. Allele frequencies were compared with gnomAD data. Statistical analyses included χ², ANOVA, and Pearson correlation. Results: For ACE, genotype distribution was: I/I – 30%, I/D – 38.3%, D/D – 18.3%; Hardy–Weinberg equilibrium was preserved (χ² = 0.52, P > 0.05). For CALCA, allele frequencies were A – 65.5%, G – 34.5%. A allele carriers showed a tendency toward more severe migraine attacks, while I/D heterozygotes had higher attack frequency (4.5 ± 1.4), though not statistically significant (P > 0.05). No associations were found with age or sex. Conclusion: CALCA and ACE polymorphisms may contribute to migraine pathogenesis, particularly through vascular and neuropeptide-mediated pathways, although larger studies are needed for confirmation.
Keywords: Migraine, CALCA, ACE, CGRP, Genetic polymorphism, Neurovascular disorders
Cite this paper: Xolmatov R. I., Abdukadirov U. T., Kasimova S. A., Abbosova I. A., Sotvoldiyev M. M., Association of CALCA and ACE Gene Polymorphisms with Migraine Susceptibility: A Clinical-Genetic Study in a Central Asian Cohort, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3551-3555. doi: 10.5923/j.ajmms.20251510.57.
1. Introduction
- Migraine is a highly prevalent and disabling neurological disorder that affects approximately 12–15% of the global population, with a markedly higher prevalence among women compared to men [1]. It is characterized by recurrent attacks of severe headache, often accompanied by nausea, photophobia, phonophobia, and, in some cases, aura symptoms such as visual or sensory disturbances. Despite its high prevalence and significant socioeconomic burden, the exact pathophysiology of migraine remains incompletely understood. Current evidence suggests that migraine is a complex, multifactorial disorder involving neurovascular, neuroinflammatory, and genetic mechanisms [2].Among the genetic determinants implicated in migraine, the CALCA and ACE genes have received particular attention. The CALCA gene encodes calcitonin gene-related peptide (CGRP), a neuropeptide with potent vasodilatory and pro-inflammatory properties. CGRP plays an essential role in migraine pathogenesis by mediating trigeminovascular activation, promoting neurogenic inflammation, and sensitizing nociceptors [3]. Elevated plasma CGRP levels have been consistently observed during migraine attacks, and pharmacological blockade of CGRP or its receptor has shown remarkable clinical efficacy in reducing migraine frequency and severity[4]. Importantly, the CALCA A>G polymorphism has been hypothesized to influence CGRP expression, with the A allele potentially associated with higher peptide levels and more severe clinical manifestations [5].The ACE gene, encoding angiotensin-converting enzyme, represents another candidate gene implicated in migraine susceptibility. ACE regulates the renin–angiotensin system by converting angiotensin I into angiotensin II, a potent vasoconstrictor. Elevated ACE activity may contribute to cerebrovascular dysfunction, endothelial impairment, and altered cerebral blood flow, all of which are relevant to migraine pathophysiology (Peres et al., 2001). The well-characterized insertion/deletion (I/D) polymorphism in intron 16 of the ACE gene has been shown to modulate enzyme activity, with the D allele associated with higher ACE activity and increased vascular tone. Several studies have reported a higher prevalence of the D/D genotype among migraine patients, suggesting that ACE polymorphisms may contribute to disease risk [6].Notably, previous investigations have also highlighted possible sex-specific genetic associations in migraine. Given the influence of sex hormones, particularly estrogen, on vascular reactivity and CGRP activity, genetic variants in CALCA and ACE may exert differential effects in women and men [8]. However, findings across studies remain inconsistent, and population-specific differences in genetic architecture may partly explain these discrepancies. In particular, there is a paucity of data from Central Asian populations, where unique genetic backgrounds and environmental exposures may shape migraine susceptibility differently compared to European or East Asian cohorts.Therefore, the present study aimed to investigate the distribution of CALCA (A>G) and ACE (I/D) polymorphisms in a cohort of Central Asian migraine patients and to explore their potential associations with clinical features such as attack frequency, duration, and pain intensity. By integrating molecular genetic data with clinical phenotyping, we sought to contribute to a better understanding of the genetic underpinnings of migraine in this population and to provide insights relevant for future personalized therapeutic strategies.
2. Methods
- This investigation was designed as a cross-sectional clinical-genetic study and was conducted at the Department of Neurology, [Institution name], during the year 2023. The study protocol was reviewed and approved by the Ethics Committee of the Ministry of Health (approval number: [insert number]), and all procedures complied with the Declaration of Helsinki. Written informed consent was obtained from all participants prior to enrollment. A total of 60 unrelated patients diagnosed with migraine according to the International Classification of Headache Disorders, 3rd edition (ICHD-3, 2018), were recruited. The cohort included 47 females (78.3%) and 13 males (21.7%), with a mean age of 34.4 ± 1.02 years. Patients with secondary headaches were excluded. Demographic and clinical data were collected using structured questionnaires and clinical interviews, including headache frequency, duration, intensity, aura symptoms, and comorbidities. Migraine characteristics were assessed by trained neurologists, with headache frequency measured as the number of attacks per month, duration as average minutes per attack, and pain intensity recorded using the Visual Analogue Scale (VAS, 0–10). Associated symptoms such as nausea, vomiting, photophobia, phonophobia, and aura were documented. Additional standardized tools included the Hospital Anxiety and Depression Scale (HADS) and the SF-36 quality of life questionnaire.Peripheral venous blood samples (5 ml) were collected from each participant in EDTA tubes. Genomic DNA was extracted using the phenol–chloroform method and quantified spectrophotometrically. The ACE intron 16 I/D polymorphism was genotyped by PCR using sequence-specific primers (Forward: 5’-CTGGAGACCACTCCCATCCTTTCT-3’; Reverse: 5’ GATGTGGCCATCACATTCGTCAGAT-3’). PCR conditions included initial denaturation at 95°C for 5 min, 35 cycles at 94°C for 30 s, 58°C for 30 s, and 72°C for 1 min, with final extension at 72°C for 7 min. Amplification products were separated by 2% agarose gel electrophoresis and visualized under UV light. The presence of a 490 bp band indicated the I allele, while a 190 bp band indicated the D allele. The CALCA A>G polymorphism was analyzed by PCR-RFLP, with PCR products digested using [enzyme name] at 37°C for 16 hours and separated on a 3% agarose gel. AA genotypes showed an undigested fragment, AG heterozygotes yielded two bands, and GG homozygotes showed a digested fragment only. Negative controls were included in each PCR run, and 10% of samples were re-genotyped for quality control, yielding 100% concordance.Allele frequencies observed in this cohort were compared with publicly available datasets, including the Genome Aggregation Database (gnomAD v3) and the 1000 Genomes Project, using the Ensembl API. Statistical analyses were performed using SPSS version 26.0. Genotype and allele frequencies were calculated by direct counting, and Hardy–Weinberg equilibrium was tested by χ² analysis. Associations between genotypes and categorical variables such as sex were evaluated using χ² tests, while comparisons of continuous variables such as age, migraine frequency, duration, and pain intensity across genotypes were assessed using one-way ANOVA. Correlations between genotypes and clinical features were further evaluated using Pearson correlation coefficients, with a p-value < 0.05 considered statistically significant.
3. Results
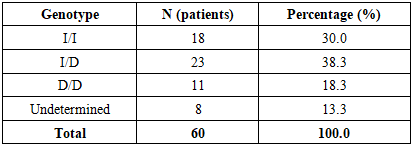
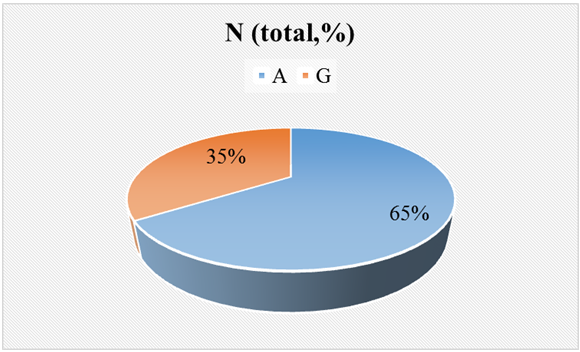
- A total of 60 migraine patients (47 females, 13 males; mean age 34.4 ± 1.02 years) were successfully genotyped for ACE I/D and CALCA A>G polymorphisms.The genotype frequencies of the ACE polymorphism were as follows: I/I in 18 patients (30.0%), I/D in 23 patients (38.3%), and D/D in 11 patients (18.3%). In 8 patients (13.3%), genotyping results were inconclusive due to technical limitations. The observed distribution conformed to Hardy–Weinberg equilibrium (χ² = 0.52, P > 0.05), indicating no deviation from population expectations. The heterozygous I/D genotype was the most common, accounting for nearly two-fifths of the cohort (Table 1, Figure 1).
|
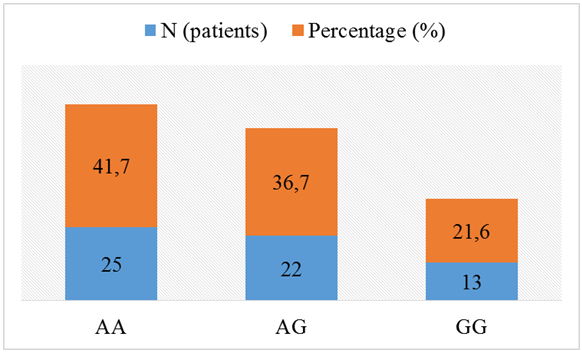 | Figure 1. CALCA Genotype and Allele Frequencies |
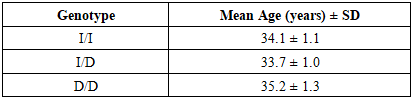
 | Figure 2. Mean Age Across ACE Genotypes |
|
|
|
4. Discussion
- The present study investigated the distribution of ACE (I/D) and CALCA (A>G) polymorphisms in a cohort of Central Asian patients diagnosed with migraine and examined their potential associations with clinical features such as attack frequency, duration, and pain intensity. The findings demonstrated that the heterozygous ACE I/D genotype was the most common in this cohort, while the CALCA A allele was more prevalent than the G allele, with AA being the most frequent genotype. Importantly, genotype distributions were consistent with Hardy–Weinberg equilibrium, and allele frequencies showed general alignment with global data but also revealed some population-specific tendencies.Our results regarding the predominance of the ACE I/D genotype are consistent with previous studies in European and Asian populations, where I/D heterozygosity was frequently observed [5,10]. The observation that the D/D genotype was less common aligns with data from neighboring Eurasian populations, suggesting potential regional genetic similarities. However, unlike some studies reporting a significant association between the D allele and migraine risk, especially migraine with aura [6,8], our analysis did not reveal statistically significant correlations between ACE genotypes and clinical outcomes such as attack frequency, duration, or pain severity. This discrepancy may be explained by the relatively small sample size of our cohort, ethnic background differences, or environmental modifiers of migraine expression.With respect to CALCA, the predominance of the A allele in our study corresponds with findings in East and South Asian cohorts, where the A allele has also been reported as more frequent [4]. Interestingly, our study identified a trend toward higher pain intensity among carriers of the A allele, which is biologically plausible given that the A allele has been hypothesized to increase calcitonin gene-related peptide (CGRP) expression. CGRP is a key mediator in migraine pathophysiology, and elevated levels of this neuropeptide have been observed during migraine attacks [2]. Although this trend did not reach statistical significance, it is noteworthy, as it aligns with pharmacological evidence showing that CGRP antagonists and monoclonal antibodies targeting the CGRP pathway are effective treatments for migraine [1].Sex-based analysis did not show significant differences in genotype distribution between male and female participants. This result contrasts with some prior studies that suggested possible sex-specific genetic associations, particularly due to hormonal influences on vascular tone and CGRP activity [9]. The lack of significant sex-related differences in our cohort may again reflect sample size limitations or indicate that genetic effects in this population are not strongly modulated by gender.A key strength of this study is that it provides the first dataset on ACE and CALCA polymorphisms in a Central Asian migraine cohort, thereby filling an existing gap in the literature.
5. Conclusions
- This study represents one of the first investigations of ACE (I/D) and CALCA (A>G) polymorphisms in a Central Asian migraine cohort. The results demonstrated that the heterozygous ACE I/D genotype was the most frequent, while the CALCA A allele predominated over the G allele. Although no statistically significant associations were found between genotypes and clinical characteristics such as attack frequency, duration, or pain intensity, trends were observed in which the ACE I/D and CALCA A allele carriers tended to report more severe clinical features. These findings are biologically plausible given the known roles of angiotensin-converting enzyme and calcitonin gene-related peptide in vascular function and migraine pathophysiology.The study underscores the importance of exploring genetic markers in diverse populations, as allele frequencies and genotype distributions may vary across ethnic groups. While our results were broadly consistent with global data, the slight differences observed highlight the need for population-specific genetic studies.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML