-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3545-3550
doi:10.5923/j.ajmms.20251510.56
Received: Oct. 2, 2025; Accepted: Oct. 23, 2025; Published: Oct. 28, 2025

Optimization of Treatment and Prevention of Chronic Generalized Catarrhal Gingivitis in Patients with Bronchial Asthma
Gulnoza Tursunova1, Dildora Taylakova2
1Department of Pathological Physiology, Bukhara State Medical Institute, Bukhara, Uzbekistan
2Department of Therapeutic Dentistry, Bukhara State Medical Institute, Bukhara, Uzbekistan
Correspondence to: Gulnoza Tursunova, Department of Pathological Physiology, Bukhara State Medical Institute, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Bronchial asthma is associated with chronic inflammation and altered oral homeostasis, leading to increased susceptibility to periodontal diseases. This study evaluated the relationship between asthma and chronic generalized catarrhal gingivitis (CGCG), with a focus on the impact of inhaled corticosteroids (ICS) and the efficacy of complex therapy using mineral and osteotropic agents. A total of 144 patients were examined and divided into main, comparison, and control groups. The inclusion of mineral therapy and osteotropic agents significantly improved periodontal indices: PMA decreased by 66.7%, CPI by 61.6%, and OHI-S by 61.9% after 12 months. Correlation analysis revealed a moderate positive association between ICS dose and periodontal deterioration (r = 0.61). These results confirm the adverse impact of prolonged ICS use on periodontal health and highlight the importance of combined therapeutic approaches to maintain oral health in asthma patients.
Keywords: Bronchial asthma, Gingivitis, Inhaled corticosteroids, Mineral therapy, Osteotropic agents
Cite this paper: Gulnoza Tursunova, Dildora Taylakova, Optimization of Treatment and Prevention of Chronic Generalized Catarrhal Gingivitis in Patients with Bronchial Asthma, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3545-3550. doi: 10.5923/j.ajmms.20251510.56.
Article Outline
1. Introduction
- According to the World Health Organization, severe periodontal disease affects approximately 19% of the global adult population, representing over one billion cases worldwide [1]. This high prevalence underscores the importance of exploring comorbidities such as bronchial asthma that may exacerbate periodontal inflammation [6,24,25].The development of chronic generalized catarrhal gingivitis (CGCG) in patients with bronchial asthma is associated with the effects of inhaled corticosteroids (ICS), which lead to a reduction in local immunity and impairment of periodontal tissue regeneration [26–28]. This work considers the effectiveness of complex therapy, including mineral therapy and osteotropic drugs.The study evaluated the effectiveness of such therapy compared with standard treatment. The obtained data demonstrated that additional use of mineral therapy and osteotropic medications made it possible to achieve a significant reduction in inflammation: the PMA index decreased by 66.7%, CPI by 61.6%, and OHI-S by 61.9% after 12 months of follow-up [31].The results show that standard therapy of CGCG in patients with bronchial asthma is not sufficiently effective. Inclusion of mineral therapy and osteotropic drugs in the treatment regimen promotes a decrease in inflammatory processes, accelerates periodontal tissue recovery, and improves oral hygiene [29–31]. Such an approach may enhance the effectiveness of dental care and improve patients’ quality of life.
2. Literature Review
- The relevance of studying the oral mucosa in patients with bronchial asthma is determined by the high prevalence of this disease and its systemic effects on the body [6–8]. Numerous studies confirm a strong relationship between chronic respiratory diseases and pathological changes in periodontal tissues [9,10,24,25]. Particular attention is paid to the mineral composition of oral fluid and its impact on the development of CGCG in asthmatic patients [2–5].One of the key aspects is the level of ionized calcium in oral fluid. The study by Romanenko I.G., Kaladze K.N., and Poleshchuk O.Y. demonstrated reduced ionized calcium concentration in children with CGCG and bronchial asthma [24,25]. ICS administration was shown to reduce calcium levels in oral fluid, worsening metabolic processes in periodontal tissues [26,27]. Calcium deficiency increases vascular permeability, decreases connective tissue strength, and intensifies inflammatory changes [29].The relationship between asthma and periodontal tissue status was also analyzed in the study by Avdeev O.V. and Vydoinyk O.Y., which revealed immunological peculiarities of inflammation in these patients [25]. The authors reported marked alterations in the oral microflora, with predominance of Porphyromonas gingivalis, Tannerella forsythia, and Prevotella intermedia [24]. Dysbiotic changes aggravate inflammation, necessitating antibacterial therapy and immunomodulatory drugs in comprehensive treatment [30,31].In recent years, considerable attention has been paid to alternative methods of CGCG management in asthmatic patients. Kozyreva Z.K. and Gontarev S.N. demonstrated the efficacy of combined phytotherapy and sulfide mineral waters, showing improvement in gingival blood supply, reduction of inflammation, and normalization of the oral fluid mineral composition [31,32]. The use of phytotherapeutic agents partly compensates for calcium deficiency and minimizes the negative effects of long-term ICS therapy [28,29,33].Thus, patients with bronchial asthma are at high risk of developing CGCG due to mineral metabolism disturbances, dysbiotic changes in oral microflora, and systemic inflammatory responses [24,26,29]. Current evidence supports the necessity of a complex treatment approach, including correction of salivary mineral composition, antibacterial therapy, and phytotherapy [29–31].
3. Materials and Methods
- To study the relationship between bronchial asthma and CGCG, methods of clinical periodontal assessment, analysis of oral fluid mineral composition, and microbiological profiling were employed [24,25,31]. It was confirmed that asthmatic patients are at higher risk of gingival inflammatory diseases due to impaired mineral metabolism and altered oral microflora [9,10,26,29].For objective diagnosis, clinical indices PMA, CPITN, and OHI-S were used to determine the severity of periodontal lesions and treatment needs [24,25]. Studies demonstrated that asthmatic patients exhibit higher index values compared to healthy individuals, reflecting more severe gingival inflammation and lower oral hygiene levels [6,9,10].The mineral composition of oral fluid was assessed by ionometry, revealing a decrease in ionized calcium in asthma patients [2,3,4,5]. ICS administration reduced salivary calcium, weakening periodontal tissue resistance to inflammation [26–28].PCR-based microbiological analysis showed increased P. gingivalis, T. forsythia, and P. intermedia in asthma patients, indicating dysbiotic changes [24,25,30]. These findings emphasize the need for a comprehensive approach combining anti-inflammatory and antibacterial therapy with mineral correction [29–31].
4. Results
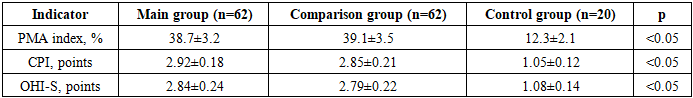
- The study involved 144 patients with CGCG and bronchial asthma. Patients were randomly assigned to two groups:• Main group (n=62): received standard treatment plus mineral therapy and osteotropic drugs.• Comparison group (n=62): received standard treatment only.• Control group (n=20): healthy individuals without asthma or periodontal disease.The mean age was 44.1±3.2 years. Groups were comparable in age and sex (p>0.05). All asthma patients had persistent moderate-to-severe disease and were on baseline ICS therapy.At baseline, periodontal inflammation was more pronounced in asthmatic patients, as indicated by significantly higher PMA, CPI, and OHI-S compared to controls (p<0.05). No significant difference was found between the main and comparison groups initially.
|
|
|
5. Discussion
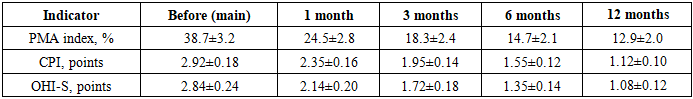
- This study revealed a significant association between bronchial asthma and periodontal inflammation. Asthmatic patients with CGCG had higher PMA (38.7±3.2%), CPI (2.92±0.18), and OHI-S (2.84±0.24) compared to controls (p<0.05). This demonstrates the negative impact of asthma and ICS on periodontal tissues, likely linked to immune dysregulation and local drug effects.Both groups improved with treatment, but patients receiving mineral therapy and osteotropic drugs had superior outcomes. By 12 months, PMA decreased by 66.7%, CPI by 61.6%, and OHI-S by 61.9%. These improvements reflect not only reduced inflammation but also structural periodontal recovery and improved hygiene.
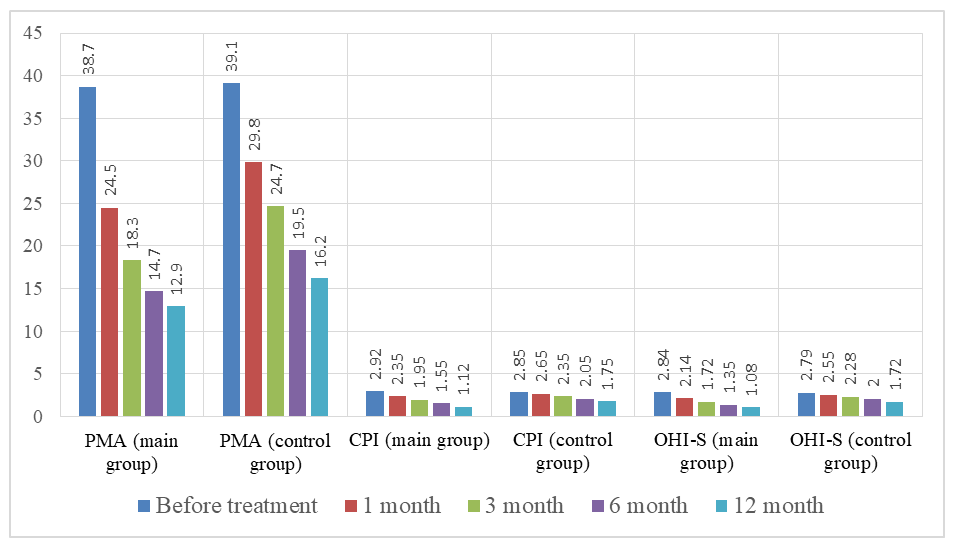 | Graph 1. Development of the RMA, CPI, and OHI-S indices |
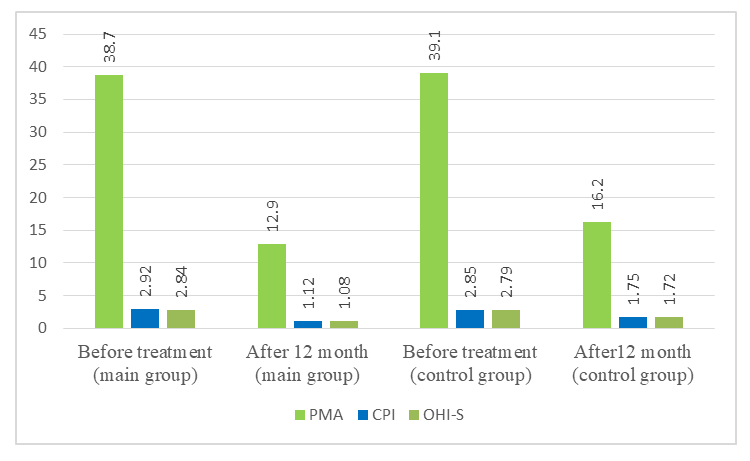 | Graph 2. Comparison of treatment effectiveness |
6. Conclusions
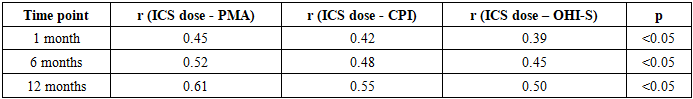
- The study revealed a systemic relationship between bronchial asthma and chronic generalized catarrhal gingivitis, demonstrating that long-term inhalation therapy with corticosteroids exerts a pronounced negative effect on periodontal tissues [2]. Patients with bronchial asthma exhibited significantly more severe gingival inflammation, as confirmed by elevated periodontal indices and alterations in the microbiological profile of the oral cavity.Standard therapy for chronic generalized catarrhal gingivitis proved insufficiently effective in this patient group. Even with conventional treatment, it was not possible to achieve substantial reduction of inflammation or stabilization of periodontal tissue status. Incorporating mineral therapy and osteotropic agents into the therapeutic protocol resulted in a marked improvement in clinical parameters, leading to sustained regression of inflammation, strengthening of connective tissue, and normalization of the mineral balance in oral fluid.Correlation analysis confirmed a direct association between the dosage of inhaled corticosteroids and the severity of gingival inflammation, indicating the cumulative adverse effect of such therapy on the periodontium. A reduction in ionized calcium levels in oral fluid, observed during the study, further highlighted the importance of maintaining mineral balance in patients with bronchial asthma.The findings suggest that inflammatory diseases of the oral mucosa in patients with bronchial asthma require a revision of standard treatment approaches. Restoring mineral balance and the use of osteotropic agents not only enhance therapeutic efficacy but also provide relapse prevention, making this method a promising direction in dentistry for this patient population.
7. Final Remark
- This comprehensive study of chronic generalized catarrhal gingivitis in patients with bronchial asthma identified previously underestimated mechanisms of the adverse impact of long-term inhalation corticosteroid therapy on periodontal tissues. It was established that classical treatment methods in this patient group do not provide sufficient efficacy, which can be explained by their systemic inflammatory response, alterations in the oral microbial composition, and disturbances of mineral metabolism.The inclusion of mineral therapy and osteotropic agents in the treatment regimen demonstrated a substantial improvement in periodontal tissue condition, as confirmed by dynamic changes in periodontal indices and biochemical analyses. The pronounced reduction in inflammation, normalization of mineral metabolism parameters, and improved oral hygiene status in patients receiving this therapy confirm its necessity in clinical practice.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML