Botirov Doniyor Abdusaidovich1, Eshbaev Erkin Abdukhalimovich2, Allaberganov Dilshod Shavkatovich3, Shukurova Yulduz Utkurovna4
1Independent Researcher, Department of Pathological Anatomy, Samarkand State Medical University, Samarkand, Uzbekistan
2Doctor of Medical Sciences, Professor, Department of Pathological Anatomy, Tashkent State Medical University, Republican Pathology and Anatomy Medical Center, Tashkent, Uzbekistan
3Candidate of Medical Sciences, Associate Professor, Department of Pathological Anatomy, Tashkent State Medical University, Republican Pathology and Anatomy Medical Center, Tashkent, Uzbekistan
4Candidate of Medical Sciences, Department of Clinical Sciences, Alfraganus University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
In the immunohistochemical examination of the myocardium in chronic ischemic heart disease (CIHD), the following markers were primarily used: Desmin, SA sarcomeric actin, and the myogenic regulatory factor MyoD1. The desmin marker used in this study represents a protein structure forming part of the intermediate filaments of myocardial myocytes and acts as a structural element determining muscle contractility, detectable in both skeletal and cardiac muscles. This intermediate filament protein is primarily expressed in the cytoplasm of cardiomyocytes, where it appears as an intense yellow staining, reflecting homogeneous expression within healthy muscle components. It is characterized by a uniform yellowish staining pattern in the cytoplasm.
Keywords:
Immunohistochemistry, Morphology, Chronic ischemic heart disease
Cite this paper: Botirov Doniyor Abdusaidovich, Eshbaev Erkin Abdukhalimovich, Allaberganov Dilshod Shavkatovich, Shukurova Yulduz Utkurovna, Immunohistochemical Age-Related Characteristics of Myocardium in Chronic Ischemic Heart Disease, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3539-3544. doi: 10.5923/j.ajmms.20251510.55.
1. Introduction
Ischemic heart disease ranks first among all diseases worldwide. Globally, about 4.1 billion people suffer from various nosological forms of chronic ischemic heart disease. This demonstrates that despite numerous proposed recommendations, the problem remains unresolved and highly diverse [1,2]. For instance, in the USA and Europe, approximately 18.3 million deaths per year are attributed to myocardial infarction, whereas in the Russian Federation and CIS countries, this figure averages 10.8 million, with the highest rate recorded in 2022 [3,4,5]. Globally, around 45.4 million deaths were caused by chronic ischemic heart disease. Although foreign and domestic researchers have studied the morphological and clinical aspects of different forms of CIHD, the diversity of etiological factors contributing to this condition and the corresponding morphological alterations continue to evolve annually [6,7,8]. The pathogenesis of ischemic heart disease often involves indirect factors-various harmful habits and lifestyle patterns prevalent among different ethnic and regional populations-that influence the myocardial structure in distinct ways. For example: In the USA and Europe, obesity is a major contributing factor to CIHD. In China and Japan, despite high levels of physical activity, emotional stress often leads to inadequate coronary perfusion and myocardial ischemia [9,10,11]. In Siberia and the Far East, deficiency of vascular-strengthening nutrients (such as Vitamin C) contributes to interstitial edema and dystrophic myocardial changes due to acute metabolic disturbances [12,13,14,15]. Thus, identifying specific substrates that indicate which myocardial substructures are initially affected during the disease process is of crucial importance for understanding the pathomorphological and immunohistochemical characteristics of chronic ischemic heart disease.Research Aim To study the morphological and immunohistochemical characteristics of the myocardium in chronic ischemic heart disease, focusing on age-related changes and the expression patterns of intermediate filament markers.
2. Materials and Methods
The research material consisted of 132 autopsy cases of individuals who died from acute myocardial infarction due to chronic ischemic heart disease (CIHD), obtained from the Republican Pathological Anatomy Center. Among them, 85 were male and 47 were female. The collected myocardial tissue samples were processed for morphological examination and subsequent immunohistochemical analysis.
3. Results and Discussion
In the studied cases of chronic ischemic heart disease, Desmin marker expression showed a low positive reaction in 73.8% of samples. This finding, from a clinico-morphological perspective, indicates excessive proliferation of fibrous structures within the myocardial stroma, conduction pathway blockade, and disruption of intercellular connectivity among cardiomyocyte bundles. At the cellular level, Desmin exhibited weak cytoplasmic staining, mainly in the perinuclear regions, presenting as a light golden homogeneous protein substrate-a sign of significantly reduced synthesis of intermediate filaments during chronic hypoxia. In the control group, Desmin expression was strongly positive in 83.16% of cases, showing intense cytoplasmic staining. This contrast underscores the sharp decline in intermediate filament synthesis in cardiomyocytes during chronic myocardial ischemia, especially with advancing age. According to the WHO age classification, the 45–59-year age group showed the highest rate of decreased Desmin expression, correlating with marked degenerative changes in the myocardium. Among younger individuals (18–44 years), a high positive reaction in 71.16% of cases indicated preserved morphological adaptation and active intracellular filament synthesis. In the 45–59-year group, 26.2% of samples demonstrated a negative Desmin reaction, while in the 60–74-year group, 55.61% showed low positive and 44.39% negative reactions. These data confirm a progressive decline in Desmin expression with age, reflecting the degenerative and fibrotic remodeling of myocardial tissue characteristic of chronic ischemic heart disease.Table 1. Desmin Marker Expression in Chronic Ischemic Heart Disease Across Different Age Groups (%)
 |
| |
|
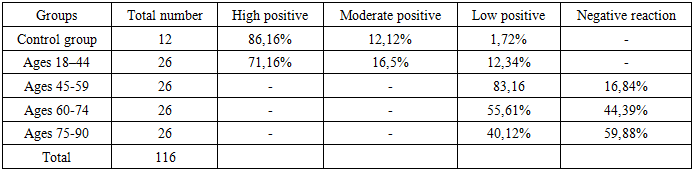
 | Figure 1. Heart of a 44-year-old male cadaver. Autopsy protocol №149-V. High positive Desmin expression. In the transverse section of cardiomyocytes, the cytoplasm shows intense yellow homogeneous protein substrates. Stain: DAB chromogen.Magnification: ×10×40 |
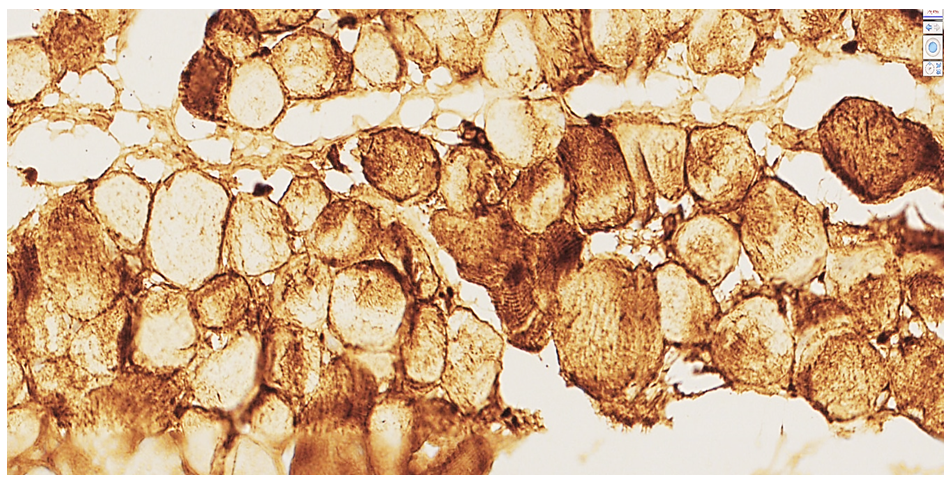
 | Figure 2. 52-year-old male cadaver. Autopsy protocol №103-V. Low positive Desmin expression. In the transverse section of cardiomyocytes, the cytoplasm shows light yellow homogeneous protein substrates with weak staining intensity. Stain:DAB chromogen.Magnification: ×10×40 |
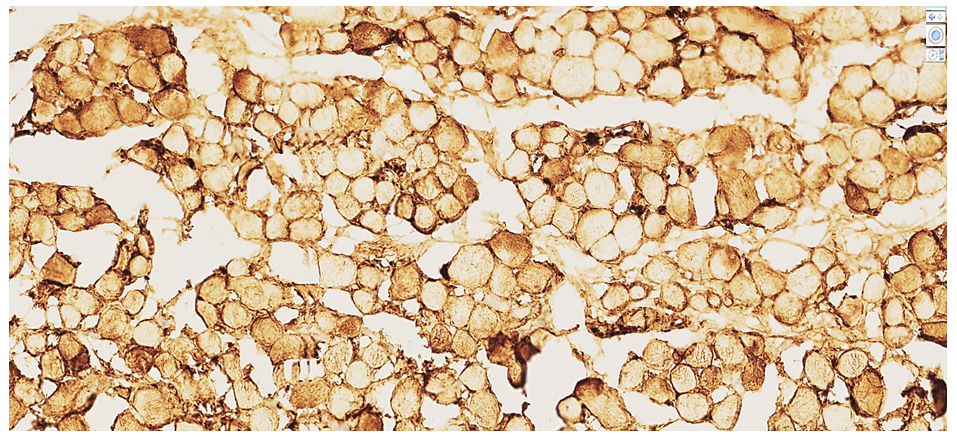
 | Figure 3. 69-year-old male cadaver. Autopsy protocol №93-V. Negative Desmin reaction. The cardiomyocytes appear homogeneous and light yellow in color, with intermediate filaments virtually undetectable. Foci of lipomatosis and sclerosis are observed between the cardiomyocyte bundles |
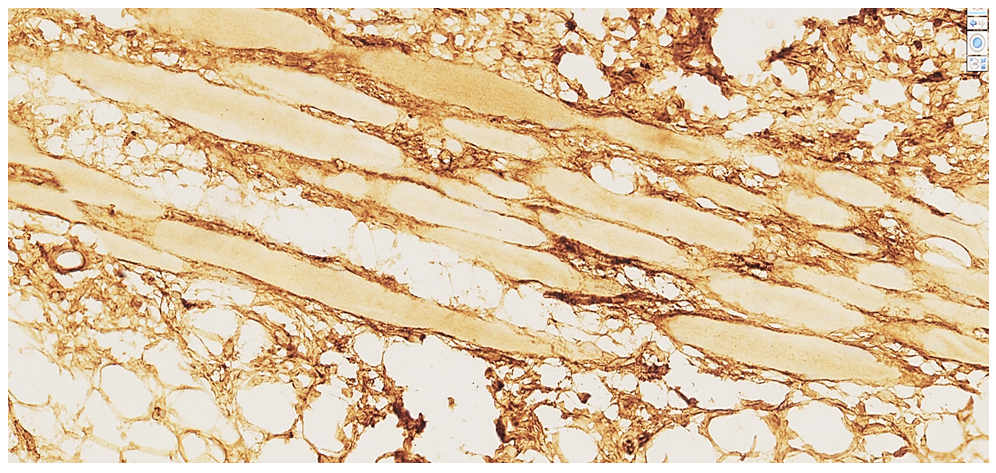
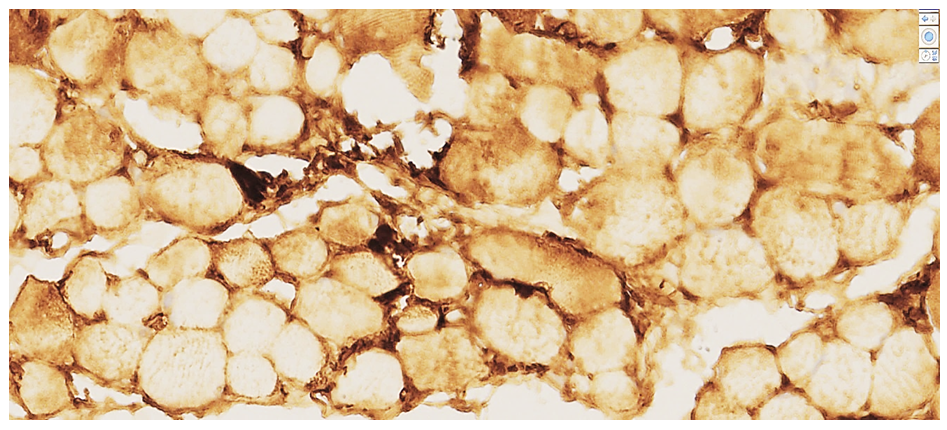 | Figure 4. Heart of a 44-year-old male cadaver. Autopsy protocol №149-V. Moderate positive MyoD1 expression. Along the perimeter of the cardiomyocyte bundles, fibroblast nuclei and cardiomyoblast cells exhibit intense golden-yellow staining. Transformation of fibroblasts within fibrotic (scarred) areas is clearly observed. Stain: DAB chromogen. Magnification: ×10×10 |
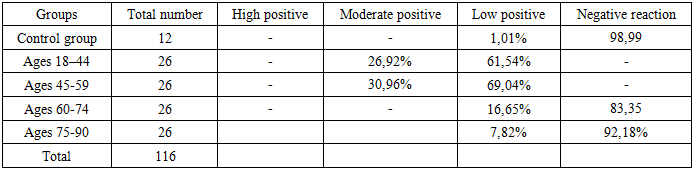
In patients aged 75–90 years with chronic ischemic heart disease (CIHD), the macroscopic reduction in heart size was mainly due to pronounced atrophic and sclerotic changes, reflecting age-related biological aging processes. Considering the reduced morphofunctional indices of the myocardium in this age group, comparative analysis was carried out relative to the control group. Such alterations indicate that in CIHD, cardiomyocytes, which are the structural subunits of the myocardium, undergo atrophic and sclerotic degeneration accompanied by cellular disorganization. These changes result in a marked decline in contractile activity, primarily due to impaired coronary perfusion and capillary insufficiency within the myocardium. The next stage of the immunohistochemical study focused on the MyoD1 myogenic regulatory protein marker. This protein is primarily associated with the proliferative activity of cardiomyocytes during the embryonic period and is widely used in practical pathology for the diagnosis of myogenic malignant tumors. In ischemic heart disease, MyoD1 serves as a marker of reparative regeneration, indicating the transformation of mesenchymal cells located around necrotic cardiomyocyte areas.Specifically, in chronic ischemic heart disease, the MyoD1 marker was used to assess long-term reparative capacity in cardiomyocytes. Moreover, MyoD1 expression reflects the transformation and differentiation of stem cells located in the endocardium and pericardium, as well as partial metaplasia of fibroblasts into myocytes in scarred myocardial regions.According to Zhao B., Chen S., Liu J. et al., activation of the H9c2 gene results in a sharp decrease in myogenin concentration within cardiomyocytes, promoting the restructuring of cardiomyoblasts and the formation of cardiomyogenic foci at the periphery of myocardial necrosis zones. Therefore, in ischemic heart disease, positive MyoD1 reactions should not be interpreted as indicators of tumor presence; rather, they reflect the transformation of fibroblasts into myocytes around foci of myocardial sclerosis. From a clinico-morphological standpoint, the expression of MyoD1 supports the concept that lipid-soluble vitamins may stimulate myocardial repair, as noted by the aforementioned authors.Thus, in our study, the application of the MyoD1 marker proved to be a crucial criterion for identifying mesenchymal cell metaplasia during reparative regeneration in chronic ischemic heart disease. In our findings: Among 18–44-year-olds, out of 26 cases, 7 (26.92%) showed moderate positive expression, 16 (61.54%) demonstrated low positive reaction, and 3 (11.53%) were negative. In the 45–59-year age group, MyoD1 expression increased by approximately 15%, with 8 cases (30.96%) showing moderate positive reaction, and 18 cases (69.04%) displaying low positive reaction. This pattern confirms that reparative regeneration activity in CIHD was higher in the 45–59-year group compared to younger individuals. From a clinico-morphological perspective, this observation aligns with the relatively stable hemodynamic state seen between ages 45–55, a period often associated with greater psychological and physical balance, healthier lifestyle adjustments, and reduction of harmful habits, which together contribute to improved myocardial adaptation and repair potential.Table 2. MyoD1 Marker Expression in Chronic Ischemic Heart Disease Across Different Age Groups (%)
 |
| |
|
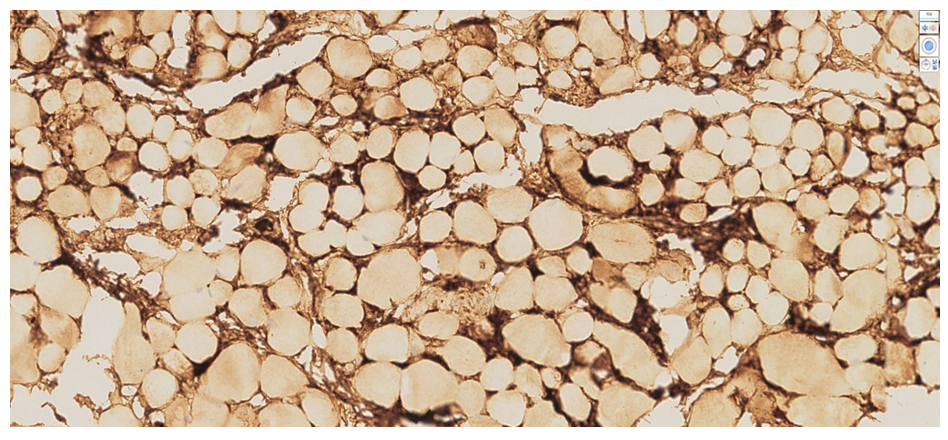 | Figure 5. Heart of a 55-year-old male cadaver. Autopsy protocol No. 209-V. Moderate positive expression of the MyoD1 marker. Numerous fibroblast nuclei and cardiomyoblast cells between the cardiomyocyte bundles are stained dark golden-yellow, indicating an ongoing reparative regeneration process. Staining: DAB chromogen. Magnification: 10×10 |
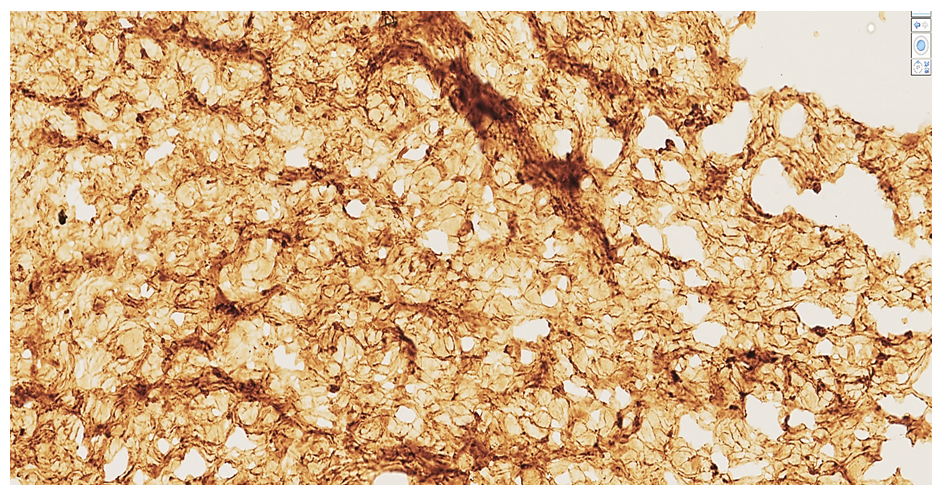 | Figure 6. Heart of a 72-year-old male cadaver. Autopsy protocol No. 251-V. Low positive expression of the MyoD1 marker. In the connective tissue and interstitial areas, preserved myofibrillar structures and dark golden-yellow stained regions resemble cardiomyoblast cells involved in an incomplete reparative process. Staining: DAB chromogen. Magnification: 4×10 |
 | Figure 7. Heart of a 69-year-old male cadaver. Autopsy protocol No. 93-V. Negative reaction for the Desmin marker. The cardiomyocyte cytoplasm appears homogeneous light yellow, with intermediate filaments almost undetectable. Between the cardiomyocyte bundles, foci of lipomatosis and sclerosis are observed. Staining: DAB chromogen. Magnification: 10×10 |
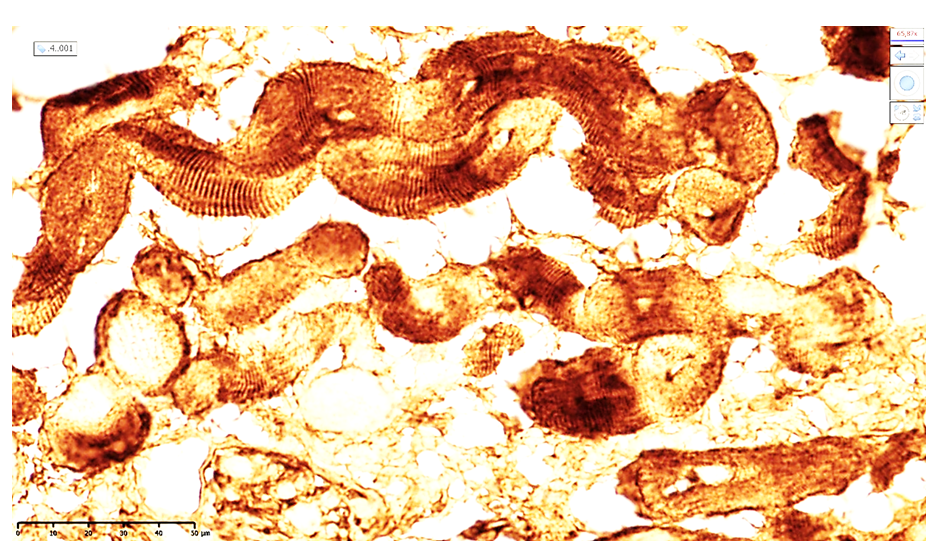 | Figure 8. Heart of a 60-year-old male cadaver. In the wall of the left ventricular apex, moderate positive expression of the AS (sarcomeric actin) marker is observed. Cardiomyocytes exhibit third-degree wavy contracture, with a reduced number of clearly visible Z-lines in the cytoplasm. The stroma shows massive sclerosis and atrophic cardiomyocytes. Staining: DAB chromogen. Magnification: 20×10 |
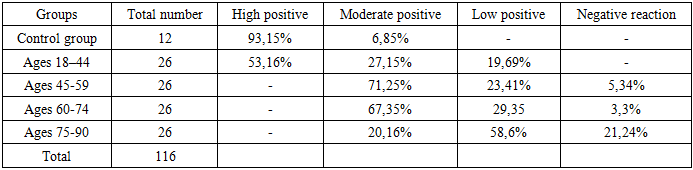
In patients aged 60–74 years with chronic ischemic heart disease, a marked decrease in reparative regeneration of cardiomyocytes—the fundamental functional subunits of the myocardium—is observed. This process is reflected by low-positive or negative MyoD1 marker expression, which morphologically corresponds to a reduction in the number of stem cells within the myocardial stroma, endocardium, and pericardium. The myocardial stroma is predominantly composed of coarse fibrous structures, with a significant reduction of parenchymal elements. These changes are manifested by cardiomyocyte bundles of varying sizes and partial replacement of the conduction pathways by fibrotic tissue. From a morphofunctional perspective, these alterations indicate a pronounced disruption of myocardial structural integrity, decreased electrical conductivity, and progressive weakening of cardiomyocyte activity. Clinically, this explains the reduced reparative regeneration index and poor response to symptomatic therapy in patients aged 60–74 years. A similar tendency is observed in the 75–90-year age group, characterized by the predominance of low or negative MyoD1 expression, confirming the near-complete exhaustion of myocardial regenerative capacity in chronic ischemic processes.Morphological and Immunohistochemical Evaluation of Actin Expression in Chronic Ischemic Heart Disease. Subsequent examinations based on actin complex staining within the sarcomere structure provide the ability to assess the transverse striations of the Z-complex, which represents the fundamental functional unit of both skeletal and cardiac muscle fibers. This approach allows for the early detection of destructive changes in the actin component of the myocardium during the progression of chronic ischemic heart disease (CIHD).Under normal physiological conditions, the actin–myosin complex, which serves as the functional substrate of cardiomyocytes, demonstrates regular transverse linear striations in microscopic examination. However, in cases of ischemic heart disease, fragmentation and destruction of this complex are clearly observed. The defective structural state of the Z-complex in cardiomyocytes in chronic ischemic heart disease contributes to varying degrees of progressive contraction (contracture) of myocardial fibers.The actin marker, a globular protein belonging to the microfilament class, exhibits positive expression through staining of thin, elongated filamentous protein complexes within the cytoskeleton.In our study, among individuals aged 18–44 years diagnosed with ischemic heart disease, high-level positive actin expression was detected in approximately 53.16% of autopsy cases that demonstrated macroscopic myocardial infarction or post-infarction cardiosclerosis. Compared to the control group, expression of the AS (sarcomeric actin) marker decreased by 35–37%. In the control group, 9 out of 12 cases (93.15%) showed strong positive expression, which provided the basis for evaluating actin structure according to its texture and staining intensity.These findings make it possible to assess the morphofunctional characteristics of the actin–myosin complex in ischemic heart disease, particularly the presence and degree of myocardial contracture, as well as interposition and structural alterations of intercalated discs. In post-infarction cardiosclerosis among patients aged 18–44 years, focal endocytomyolysis of the actin component within the actin–myosin complex was observed. This finding, together with angular deformation of cardiomyocytes, indicates the occurrence of cellular contracture processes.Immunohistochemically, microscopic examination of cardiomyocytes in this age group revealed sarcomeric actin structures within the cytoplasm showing uniform dark brown staining intensity. However, the presence of cystic vacuole-like spaces and oval foci of degeneration in between these regions indicates the onset of destructive intracellular changes. In these areas, where contractility is absent, wave-like deformations of the myofibrils are visible, which clinically and morphologically correspond to focal fibrillation and contracture zones within the myocardium.Table 3. AS -Marker Expression in Chronic Ischemic Heart Disease Across Different Age Groups (%)
 |
| |
|
Immunohistochemical Evaluation of Sarcomeric Actin (AS) Expression in Chronic Ischemic Heart Disease Across Different Age Groups: Group II (Ages 45–59 years). These changes indicate a decrease in the myocardial contractility coefficient and the development of fibrillation phenomena. From a clinicomorphological perspective, chronic ischemic heart disease (CIHD) in this group is characterized by disruption of synchronous myocardial contractions, leading to the emergence of ectopic impulse-generating foci that manifest as various types of arrhythmias. Morphological investigations confirmed that these arrhythmias correspond to simultaneous contraction of some cardiomyocyte bundles and relaxation of others. Between the cardiomyocyte bundles, coarse fibrous connective tissue was observed, impeding impulse conduction and confirming the described electrophysiological disturbances. In this group, moderate positive AS expression was identified in 71.25% of cases, low positive reaction in 23.41%, and negative expression in 5.34%. These findings confirm an age-dependent decrease in sarcomeric actin protein and a decline in myocardial contractility, both clinically and morphologically evident. In individuals aged 60–74 years, low positive AS expression was noted. This corresponded to atrophic and sclerotic changes in cardiomyocyte bundles and a marked reduction in Z-line density, accompanied by parallel loss of actin structures. Most cardiomyocytes contained homogeneous protein substrates, with evidence of fragmentation and destruction of the actin–myosin complex and contracture of a small number of fibers. Morphological adaptation in this group was characterized by high contractility in the remaining preserved actin–myosin complexes, reflecting compensatory hyperfunction. However, most cardiomyocyte bundles showed contracture, wave-like deformation, and cystic dilation of degenerated actin filaments. Within a 200× visual field, active cardiomyocyte bundles were observed in approximately 45–56% of cases. Among the 26 examined cases, 67.35% showed moderate positive expression, indicating a 2.5-fold reduction in the morphofunctional activity of cardiomyocytes compared with younger groups. Clinically and pathologically, these data suggest that in death-causing mechanisms, myocardial failure is primarily due to cardiogenic shock rather than pulmonary edema. This necessitates individualized therapeutic approaches and careful consideration of cardiotropic drug use during resuscitation. In 29.35% of cases, sarcomeric actin expression was low positive, correlating with higher age-related mortality, while 3.3% exhibited negative immunoreactivity. Microscopically, wave-like deformation of high-contractility cardiomyocytes was observed alongside irregular coarse fibrous formations in interstitial zones, confirming the subcompensation stage of myocardial function. Hypertrophy of the remaining functional cardiomyocytes was evident. Compared with the control group, sarcomeric actin expression decreased 2.5-fold, with a corresponding decline in the number of contractile cardiomyocytes. These changes were accompanied by an increase in connective tissue components between actin-deficient cardiomyocytes. In 60–74-year-olds, only morphofunctionally preserved contractile cardiomyocytes displayed low-grade positive AS expression, while sclerotic and atrophic cells demonstrated negative reactions. The myocardial stroma consisted predominantly of coarse fibrous connective tissue, contributing to deformation of fiber bundles and functional insufficiency. Stromal actin proteins were mainly localized around blood vessel walls and within myofascicular membranes. Group IV (Ages 75–90 years). In this group, myocardial tissue consisted largely of cardiomyocytes of varying, mostly small, atrophic sizes. Low AS marker expression confirmed a sharp decline in actin filament content. Among 26 examined cases, 20.16% demonstrated moderate positive expression, whereas nearly 80% of CIHD cases were dominated by cardiogenic shock as the terminal mechanism. This emphasizes that, during autopsy, attention should focus not only on pulmonary edema but also on the tonic state of the myocardium. Low positive actin expression was identified in 58.6% of cases, while negative expression was found in 21.24%, representing a 4.5-fold reduction compared with the control group.
4. Conclusions
Based on immunohistochemical studies of the myocardium in chronic ischemic heart disease, the following trends were established. Desmin marker: Compared with the control group, Desmin reactivity in the 45–59 and 60–74-year age groups decreased 2–4.5-fold, confirming an age-related decline in reparative regeneration capacity of the myocardium. This highlights the need for age-specific individualized therapeutic approaches. MyoD1 marker: The highest synthesis of myogenin was observed in patients aged 45–59 and 60– 74 years, exceeding the control group by 5–10-fold, while in 75–90-year-olds, a near-complete deficiency of myogenin stimulatory activity was detected. Clinically and morphologically, this indicates a sharp decline in myocardial viability and regenerative potential. AS (sarcomeric actin) marker: Similar to the above, actin protein expression was highest in the 18–44-year group, nearly comparable to controls. In contrast, 45–59 and 60–74-year groups showed 2.5–4.5-fold lower expression, accompanied by fibrillation phenomena and dominance of cardiogenic shock as terminal events. Collectively, these results demonstrate that age-related structural and functional deterioration of the myocardial cytoskeleton underlies impaired contractility, increased arrhythmogenicity, and reduced regenerative response in chronic ischemic heart disease.
References
| [1] | Ackerman M.J., Priori S.G., Willems S., et al. HRS/EHRA Expert Consensus Statement on the State of Genetic Testing for the Channelopathies and Cardiomyopathies. // Heart Rhythm. – 2011. – Vol. 8(8). – P. 1308–1339. |
| [2] | Bokeria O.L., Biniashvili M.B. Molecular genetic aspects of sudden cardiac death. // Forensic medical examination. - 2013. - No. 5. - P. 14-19. |
| [3] | Smolensky A.V., Lyubina B.G. Diagnostics of sudden cardiac death in young people. - M.: Medicine, 2002. - 134 p. |
| [4] | Zhou SF, Liu RX, Luo HW, Li H, Guan XK, Yin LL, Li L, Hu DP. [Effect of Zishen Huoxue Recipe on Pathomorphology in Coronary Heart Disease Rats with Shen Deficiency Blood Stasis Syndrome]. Zhongguo Zhong Xi Yi Jie He Za Zhi. 2016 Jan; 36(1): 85-9. |
| [5] | Rosenberg VD, Nepomnyashchikh LM. Pathomorphological peculiarities of coronary artery ectasias and their role in the pathogenesis of sudden cardiac death. // Bull Exp Biol Med. 2004 Nov; 138(5): 515-21. |
| [6] | Aktürk E, Aşkın L, Nacar H, Taşolar MH, Türkmen S, Çetin M, Bozkurt M. Association of serum prolidase activity in patients with isolated coronary artery ectasia. Anatol J Cardiol. 2018 Feb; 19(2): 110-116. doi: 10.14744/AnatolJCardiol.2017.8160. Epub 2018 Jan 17. |
| [7] | Giannopoulos AA, Buechel RR, Kaufmann PA. Coronary microvascular disease in hypertrophic and infiltrative cardiomyopathies. // J Nucl Cardiol. 2023 Apr; 30(2): 800-810. |
| [8] | İrgi T, Baycan ÖF, Güvenç TS, Özcan FB, Atıcı A, Yılmaz Y, Çalişkan M. Concomitant amyloidosis is the primary cause of endothelial and coronary microvascular dysfunction in carpal tunnel syndrome. // Am Heart J Plus. 2024 Apr 15; 41: 100393. |
| [9] | Patel AR, Rodriguez Lozano PF. Impact of coronary artery disease on the myocardium: importance of ischemia, infarction, and contractility. //JACC Cardiovasc Imaging. 2022 Aug; 15(8): 1423-1426. |
| [10] | Chandarana H, Srichai MB. Evaluation of myocardial abnormalities and ischemia. // Radiol Clin North Am. 2010 Jul; 48(4): 771-82. |
| [11] | Bettencourt N, Ferreira ND, Leite D, Carvalho M, Ferreira WDS, Schuster A, Chiribiri A, Leite-Moreira A, Silva-Cardoso J, Nagel E, Gama V. CAD detection in patients with intermediate-high pre-test probability: low-dose CT delayed enhancement detects ischemic myocardial scar with moderate accuracy but does not improve performance of a stress-rest CT perfusion protocol. // JACC Cardiovasc Imaging. 2013 Oct; 6(10): 1062-1071. |
| [12] | Heusch G, Skyschally A, Kleinbongard P. Coronary microembolization and microvascular dysfunction. // Int J Cardiol. 2018 May 1; 258: 17-23. |
| [13] | Liu JF, Wang BW, Hung HF, Chang H, Shyu KG. Human mesenchymal stem cells improve myocardial performance in a splenectomized rat model of chronic myocardial infarction. // J Formos Med Assoc. 2008 Feb; 107(2): 165-74. |
| [14] | Hao M, Wang R, Wang W. Cell Therapies in Cardiomyopathy: Current Status of Clinical Trials. Anal Cell Pathol (Amst). 2017; 2017: 9404057. |
| [15] | Katarzyna R. Adult Stem Cell Therapy for cardiac repair in patients after acute myocardial infarction leading to ischemic heart failure: An Overview of Evidence from the Recent Clinical Trials. Curr Cardiol Rev. 2017; 13(3): 223-231. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML









