-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3523-3531
doi:10.5923/j.ajmms.20251510.52
Received: Sep. 29, 2025; Accepted: Oct. 21, 2025; Published: Oct. 28, 2025

Metabolic Profile of Military Personnel as a Predictor of Immune Response Under Physical Load
Pulatova Zarina Aliyevna, Burankulova Nigora Mikhaylovna
Associate Professor, PhD, Military Medical Academy of the Armed Forces of the Republic of Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Regular physical activity influences the immune system both acutely and chronically. In this study, we assessed key metabolic and nutritional biomarkers – including glucose, lipid profile, iron stores, and vitamin D status – in different categories of military servicemen to evaluate their impact on immune responsiveness to physical exertion. A total of 257 healthy male servicemen were divided into four groups (young conscripts, contract soldiers, high physical load units, and officers/instructors). Baseline biochemical indicators were measured for all participants and analyzed alongside immunological parameters. Results showed that serum lipid levels (total cholesterol and low-density lipoprotein cholesterol) increased progressively with age and service duration (from conscripts to officers), accompanied by a higher frequency of subclinical dyslipidemia. High-load personnel and officers also exhibited less favorable high-density lipoprotein and triglyceride profiles compared to younger servicemen. Ferritin levels indicated latent iron deficiencies in a subset of younger troops, while vitamin D deficiency or insufficiency was widespread in all groups, with only about one-quarter of servicemen having optimal 25(OH)D levels. These metabolic trends correlated with immune findings: servicemen with less favorable metabolic profiles tended to have higher baseline inflammatory indexes. Our findings suggest that the metabolic profile of servicemen – including dyslipidemia, iron status, and vitamin D levels – may serve as important predictors of the magnitude and balance of immune responses to physical stress.
Keywords: Exercise immunology, Military personnel, Metabolic syndrome, Vitamin D, Immune readiness, Lipid profile, Inflammation
Cite this paper: Pulatova Zarina Aliyevna, Burankulova Nigora Mikhaylovna, Metabolic Profile of Military Personnel as a Predictor of Immune Response Under Physical Load, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3523-3531. doi: 10.5923/j.ajmms.20251510.52.
1. Introduction
- It is known that regular physical exercise exerts complex effects on both innate and adaptive immunity. Moderate-intensity exercise has been demonstrated to enhance immune surveillance and reduce the risk of infection, whereas excessive workloads can transiently suppress certain immune functions [1] [2]. For example, the classic “J-shaped” curve hypothesis proposed by Nieman suggests that while moderate exercise lowers infection risk, very intense or prolonged exercise may increase short-term susceptibility to infections, especially upper respiratory tract infections in endurance athletes. Recent research has refined this view: many apparent post-exercise immune deficits may actually reflect other stressors (lack of sleep, energy deficit, extreme environment) rather than exercise alone [3]. Indeed, current consensus holds that an active lifestyle generally benefits immune defense, with well-periodized training enhancing immune competence and reducing inflammation in the long term.It is known that the nutritional and metabolic status of an individual significantly modulates immunity. For example, obesity and dyslipidemia are associated with a chronic low-grade inflammatory state that can impair immune response effectiveness. Conversely, maintaining adequate micronutrient levels (such as iron and vitamin D) is critical for optimal immune function. Vitamin D in particular has gained attention as an immunomodulator; deficiency in 25(OH)D is widespread in many populations and has been linked to higher incidence of respiratory infections and poorer immune regulation. Military personnel often face unique challenges, including high physical demands and environmental stressors, which can affect both their metabolic profile and immune status. However, baseline metabolic health metrics (like lipid profile, glucose, iron stores, etc.) are not routinely monitored in young servicemen, even though these factors might explain inter-individual variability in adaptation to training loads.For example, it is known that elevated serum cholesterol and other atherogenic lipid markers can promote systemic inflammation and may predispose individuals to exaggerated immune responses under stress. On the other hand, deficiencies such as low iron stores or hypovitaminosis D could weaken immune defenses and impair recovery. Given the multi-layered interaction between metabolism and immunity, this study aimed to evaluate the biochemical profile of different groups of military servicemen and to determine how these metabolic indicators relate to their immune responses to physical load. We hypothesized that older and more experienced servicemen (e.g. officers) might exhibit less favorable metabolic profiles (due to longer service and lifestyle factors) which could correlate with distinctive immune parameters, compared to younger recruits.
2. Methods
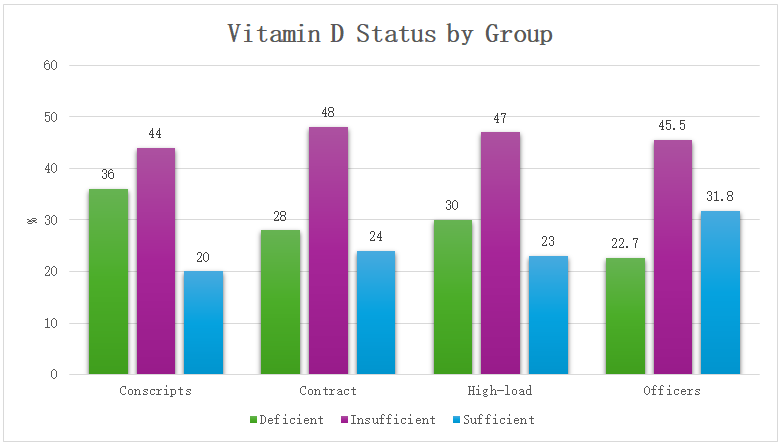
- Study design and participants: This cross-sectional study included 257 male military personnel of the Armed Forces, categorized into four groups based on service status and typical physical load: Group 1 – young servicemen in initial training (n=67, newly enlisted conscripts); Group 2 – active duty soldiers on regular service training (contract servicemen, n=64); Group 3 – high physical load units (mountain troops, reconnaissance and airborne units with intensive exercise routines, n=66); Group 4 – officers and instructors with stable routine activity, serving as a reference group (n=60). All participants were 18–40 years old (mean age 25.5 ± 5.8 years) and clinically healthy at the time of inclusion. Key exclusion criteria were any acute illness in the 4 weeks prior or decompensated chronic conditions. The study was conducted in 2022–2025 under real military training conditions. Written informed consent was obtained from all servicemen, and the protocol was approved by the Defense Health Research Ethics Committee.Anthropometry and physical load assessment: Basic anthropometric measurements were recorded, including body mass index (BMI), waist circumference, and waist-to-height ratio (WHtR). A standardized 20-meter shuttle run test (Léger’s beep test) was used to evaluate fitness and induce acute physical load in a controlled manner. All participants performed the shuttle run to volitional exhaustion in the morning (08:00–10:30) after abstaining from heavy exercise for 24 hours. Heart rate was monitored (chest belt, recorded every second) to obtain peak heart rate (HR_max). Rating of perceived exertion (RPE) was collected 20–30 minutes post-exercise and used to calculate session training load (session-RPE × duration). For all participants, blood samples were taken at three time points relative to the shuttle test: immediately before exercise (T0, baseline), immediately after exercise (T1), and 24 hours post-exercise (T2). This design allowed assessment of acute immune changes and recovery dynamics.Laboratory measurements: Venous blood was drawn from the antecubital vein at each time point under fasting, resting conditions. A complete blood count was performed using an automated hematology analyzer, with manual smear review for differential when needed. Total leukocyte count and differential counts (neutrophils, lymphocytes, monocytes) were recorded. From these, the neutrophil-to-lymphocyte ratio (NLR) was calculated for each sample as an integrated inflammation index. Saliva samples were collected to assess mucosal immunity: unstimulated saliva was obtained over 5 minutes before exercise (T0) and at T1 and T2, then centrifuged and stored at –20 °C. Secretory immunoglobulin A (sIgA) concentration in saliva was measured by ELISA (enzyme-linked immunosorbent assay), and salivary flow rate was noted to compute total sIgA secretion if needed. In a subset of participants (n≈25–30 per group), serum interleukin-6 (IL-6) was measured as a key pro-inflammatory cytokine using a high-sensitivity chemiluminescent immunoassay (Abbott Architect i2000SR). All immunological assays were conducted in the same certified laboratory with proper quality controls in place.Biochemical assays: Fasting blood samples (collected at baseline T0) were also analyzed for metabolic parameters. Serum glucose was measured by the glucose oxidase enzymatic colorimetry method. Lipid profile included total cholesterol (Total-C), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and triglycerides, all determined via enzymatic methods on a Roche Cobas c311 autoanalyzer. High-sensitivity C-reactive protein (hs-CRP) was measured immunoturbidimetrically as a systemic inflammation marker. Iron stores were evaluated by serum ferritin concentration using electrochemiluminescent immunoassay (Cobas e411 analyzer). In a partial sample of participants (n=96), vitamin D status was assessed by measuring 25-hydroxyvitamin D [25(OH)D] levels (chemiluminescent assay, Abbott Architect) with samples stored at –80°C until batch analysis. Vitamin D status was classified as deficient (<20 ng/mL), insufficient (20–30 ng/mL), or sufficient (>30 ng/mL) for descriptive analysis. All assays underwent rigorous internal and external quality control, and results were recorded for statistical analysis.Statistical analysis: Data were analyzed with SPSS 25.0. Continuous variables are presented as mean ± standard deviation (M ± SD). Group comparisons for baseline biochemical parameters were performed by one-way ANOVA with Tukey’s post hoc tests. The distribution of abnormal values (e.g. % of subjects above or below clinical cut-offs) was compared using Chi-square test. Repeated-measures ANOVA was used to assess changes in immune parameters over time (T0, T1, T2) and interactions with group. Pearson correlation analysis was applied to explore associations between baseline metabolic markers and immune indices (e.g. cholesterol vs NLR, 25(OH)D vs sIgA). A significance threshold of p<0.05 was used.
3. Results
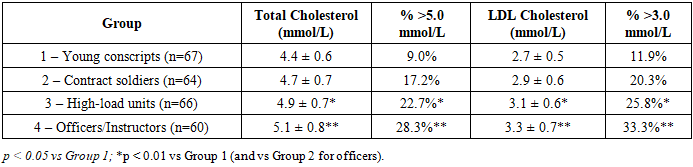
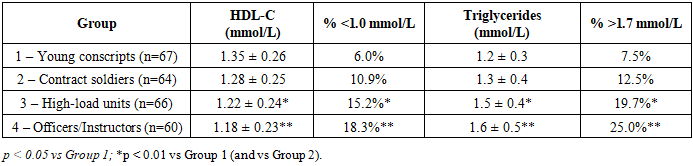
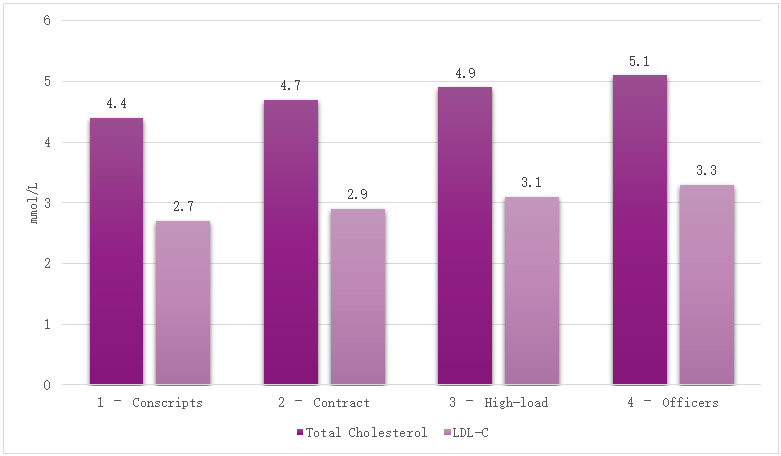
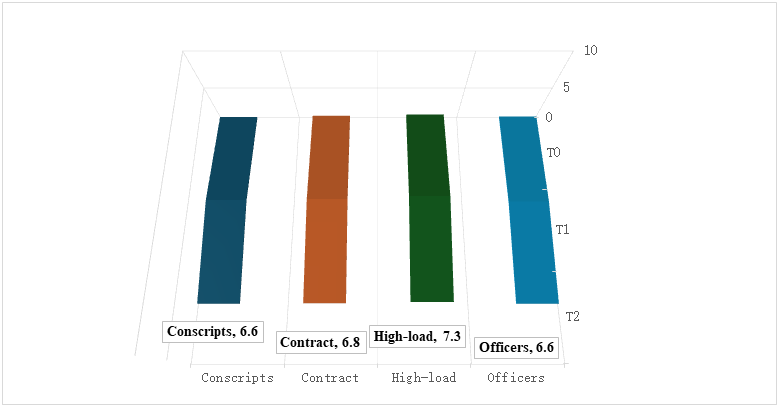
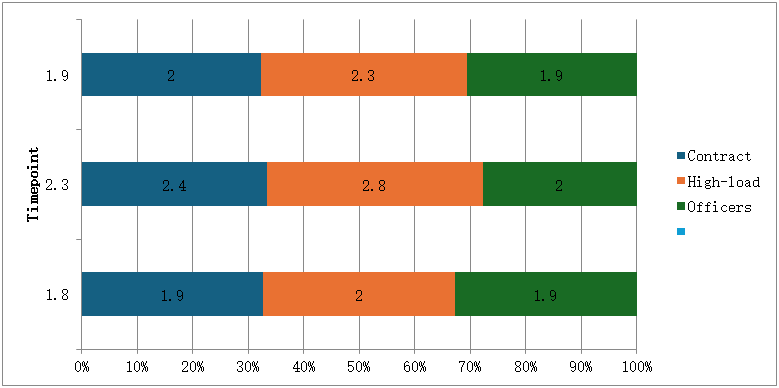
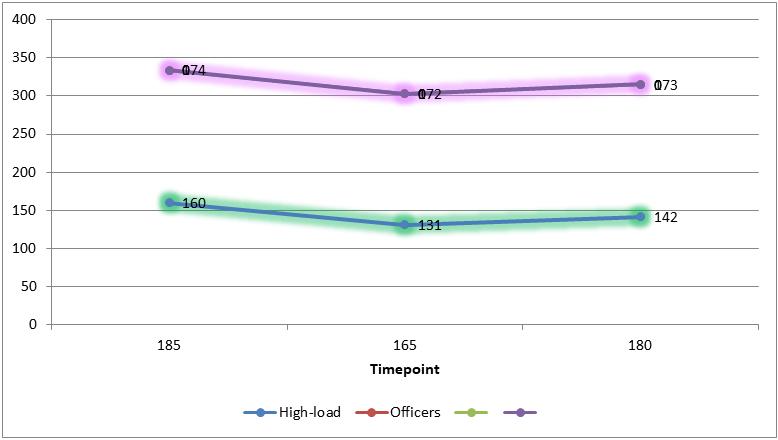
- Baseline Metabolic CharacteristicsIt is known that lipid levels tend to increase with age and prolonged service. For example, Table 1 summarizes the baseline lipid profile across the four groups of servicemen. Total cholesterol (TC) showed a stepwise increase from the youngest group to the officers: mean TC was 4.4 ± 0.6 mmol/L in young conscripts, rising to 5.1 ± 0.8 mmol/L in officers (p<0.01 vs Group 1). Similarly, mean LDL-C increased from 2.7 ± 0.5 mmol/L to 3.3 ± 0.7 mmol/L (p<0.01). The proportion of individuals with borderline-high cholesterol (>5.0 mmol/L) or elevated LDL (>3.0 mmol/L) was significantly greater in the high-load and officer groups than in young servicemen (e.g. one-third of officers had LDL >3.0 mmol/L, vs 11.9% of conscripts). These data indicate an unfavorable shift in lipid profile with increasing service duration and training load intensity.
|
|
|
|
4. Discussion
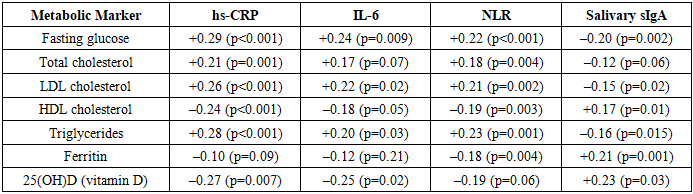
- In this study of healthy military personnel, we found that distinct metabolic profiles correspond to differences in immune status and responses to physical load. The key findings can be summarized as follows: (1) Older and more experienced servicemen (especially those in high-load roles and officers) showed a trend toward metabolic changes such as higher cholesterol, LDL, and triglycerides, along with lower HDL. These changes, while mostly within subclinical ranges, were associated with a pro-inflammatory shift in immune indicators. (2) Micronutrient status varied by group – young recruits had more latent iron deficiency, and vitamin D insufficiency was pervasive in all groups. Importantly, those with poorer vitamin D status exhibited signs of elevated inflammation and weaker mucosal immunity. (3) Acute exercise induced significant immune changes (leukocytosis, neutrophilia, sIgA reduction, IL-6 surge) in all servicemen, but the magnitude and duration of these changes were greatest in individuals from high physical load units. Officers, presumably benefiting from long-term adaptation and possibly healthier lifestyles, had the most stable immune response with minimal perturbation. (4) There were significant correlations linking metabolic risk markers (e.g. LDL, glucose) to higher inflammatory markers at baseline. Collectively, these findings underscore the interplay between metabolic health and immune resilience in physically active populations.Our results are in line with the concept of an “immuno-metabolic” profile influencing exercise outcomes. Prior research has shown that obesity and central adiposity act as chronic inflammatory stimuli, accelerating immunosenescence and impairing vaccine responses [5] [6]. Although our participants were generally young and not obese on average, even modest elevations in BMI or waist circumference in some individuals could have contributed to elevated NLR and CRP. Kondrat’ev et al. (2021) reported that NLR is a reliable marker of systemic inflammation in obesity, and we similarly observed higher NLR in subjects with metabolic irregularities (e.g. dyslipidemia or low HDL often co-occuring with higher BMI). Furthermore, Lazarev et al. (2022) demonstrated strong correlations between visceral fat and inflammatory markers like IL-6 and CRP. Our finding that waist/height ratio correlated more strongly with CRP and NLR than BMI supports the idea that central fat distribution is critical in immune modulation, even in a non-obese range.One novel aspect of this study is highlighting the high prevalence of vitamin D insufficiency in military personnel and its possible impact on immunity. Nearly 75% of tested servicemen had suboptimal 25(OH)D levels, which is consistent with other reports of widespread vitamin D deficiency in Central Asian populations despite abundant sunlight. Gorelov et al. (2023) in a systematic review confirm that vitamin D plays an important role in preventing respiratory infections. In our data, lower vitamin D was associated with higher CRP/IL-6 and lower sIgA, suggesting a more inflammation-prone and infection-prone immune status, which aligns with the immunoregulatory function of vitamin D noted in athletes and soldiers by various authors. This finding has practical implications: correcting vitamin D deficiency (through supplementation or diet) could be a simple measure to improve immune readiness in the military, as also recommended by recent military medicine guidelines [7].The acute exercise findings also deserve discussion. We observed that high physical load personnel had an exaggerated leukocyte and cytokine response to the shuttle run compared to others. While a certain level of post-exercise immune activation is normal, the sustained elevation at 24 h in this group might indicate incomplete recovery or an “open window” of increased susceptibility to infections. Vasilenko (2015) and Didenko & Aleksanyants (2015) both documented that intense training blocks can cause transient drops in salivary IgA and spikes in cortisol, correlating with higher incidence of upper respiratory tract infections in athletes [8]. Our data similarly showed a ~18% sIgA drop in high-load servicemen and only partial rebound by next day, which could translate to higher URTI risk. Indeed, there is evidence that soldiers undergoing heavy training (e.g. special forces) report more frequent respiratory illnesses, likely due to this immune suppression combined with operational stress. The more tempered response in officers suggests that long-term adaptation or possibly lifestyle factors (better nutrition, routine) confer a protective effect—this resonates with studies showing that well-conditioned individuals have less dramatic “post-exercise immune depression” [3] and a quicker return to homeostasis.Notably, the groups with more robust immune perturbations (high-load units) were also those with slightly worse metabolic profiles on average (higher LDL, more vitamin D deficiency). This raises the question: are their heightened inflammatory responses a consequence purely of acute exertion, or are they primed by underlying metabolic/inflammatory status? It is plausible that chronic training stress combined with marginal nutrition (e.g. insufficient vitamin D, iron) creates a pro-inflammatory baseline that amplifies acute responses. On the other hand, the officers’ favorable metabolic status (e.g. relatively lower triglycerides and higher vitamin D sufficiency) could be contributing to their stable immune responses. Simpson et al. (2020) emphasize that maintaining good general health and nutrition is key to an effective immune system in athletes [9]. Our findings support that recommendation in a military context: attention to metabolic health — treating dyslipidemia, correcting micronutrient deficiencies, etc. — may improve soldiers’ immunological resilience to training and deployment stress.Some limitations of this study should be noted. The research was cross-sectional in metabolic measurements, so we cannot infer causality between metabolic factors and immune differences. Unmeasured confounders (diet, sleep, genetics) might also influence both metabolism and immunity. The vitamin D and IL-6 data were obtained in subsets of participants, which limits generalization. Additionally, while we stratified by broad service categories, there is individual variability within groups (some conscripts may be very fit, some officers less so). Nevertheless, the significant trends observed suggest real physiological differences that align with group roles. Future longitudinal studies or interventions (e.g. vitamin D supplementation trials, tailored nutrition programs) in military populations could more directly test the causative links between improving metabolic health and enhancing immune function.In conclusion, this study demonstrates that even in young, non-obese military men, there are gradations of metabolic health that correlate with immune readiness. Servicemen with optimum metabolic profiles (healthy lipid levels, adequate iron and vitamin D) tended to exhibit balanced immune responses to physical exertion, whereas those with emerging metabolic risk factors showed heightened inflammation and transient immune suppression after exertion. These findings highlight the importance of monitoring and managing metabolic health as part of military medical readiness programs. Simple measures such as nutritional supplementation (vitamin D, iron when needed) and lifestyle interventions to improve lipid profiles could confer immunological benefits, potentially reducing illness rates and improving performance. In the modern military setting, where personnel are expected to withstand significant physical and environmental stress, maintaining metabolic fitness is as crucial as physical fitness for ensuring an optimal, resilient immune system [10].
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML