-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(10): 3332-3335
doi:10.5923/j.ajmms.20251510.11
Received: Sep. 16, 2025; Accepted: Oct. 8, 2025; Published: Oct. 11, 2025

Changes of Brain Morphogenesis and Cell Density in Rats Born with Metabolic Syndrome
Usmanov R. Dj., Kuziev Z. N.
Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In this study, changes in morphological indicators of brain development and cell density in the prefrontal cortex were analyzed in offspring born to mothers with metabolic syndrome. Total body and brain weight, anatomical dimensions of the hemispheres, and cell density were studied at 3, 7, 14, 21, 30, and 60 days of postnatal ontogenesis. The differences between the control and experimental groups were statistically significant, and cell density in children in the experimental group decreased by 20–25% at 14–60 days (p<0.05). The results of the study confirm the negative impact of metabolic syndrome on the neurodevelopment of the offspring.
Keywords: Metabolic syndrome, Prefrontal cortex, Brain, Cell density, Neurogenesis, Postnatal ontogenesis
Cite this paper: Usmanov R. Dj., Kuziev Z. N., Changes of Brain Morphogenesis and Cell Density in Rats Born with Metabolic Syndrome, American Journal of Medicine and Medical Sciences, Vol. 15 No. 10, 2025, pp. 3332-3335. doi: 10.5923/j.ajmms.20251510.11.
1. Introduction
- Metabolism is one of the most pressing problems of modern medicine today. Metabolic syndrome is a set of symptoms, such as obesity, hypertension, high blood sugar and cholesterol, which significantly increases the risk of developing brain pathology, type 2 diabetes and a number of other diseases. In fact, it is not a disease in itself, but a group of risk factors that often occur together, increasing the likelihood of a serious illness [3,5,9]. The term “metabolic syndrome” was introduced relatively recently - in the 80s of the 20th century. It is one of the main health problems in many countries of the world [6,8]. If previously metabolic syndrome was considered a disease of the elderly, now the percentage of young people suffering from it is increasing. Metabolic syndrome is a metabolic risk factor for the development of vascular diseases that are pathogenetically related and mutually reinforce each other [1,7]. Given this, as well as the high prevalence among the population and the sharp increase in the frequency of metabolic syndrome (including its aging trend) predicted by the WHO, this problem is one of the priority areas of modern medicine [2,4].Taking into account the above, we set out to study the morphological changes in the brain of offspring born to mothers with metabolic syndrome.
2. Materials and Methods
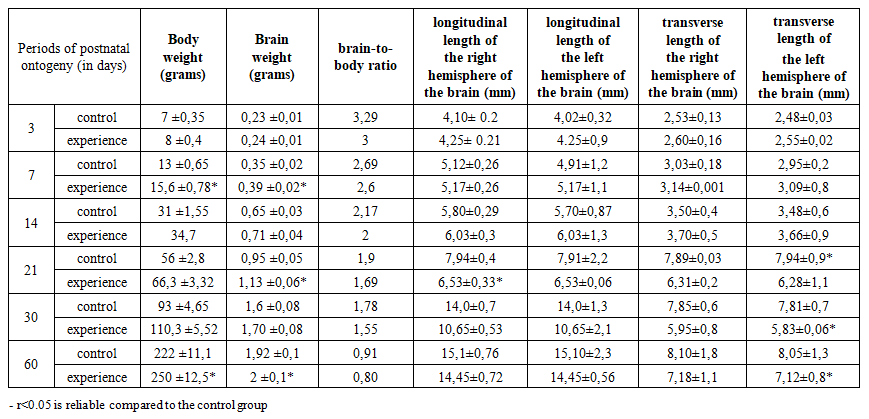
- The studies were conducted from 2024 to 2025 in the vivarium of the Tashkent Medical Academy. Permission was obtained from the Center for Bioethics for conducting experiments and the experiments were conducted in accordance with the requirements of the Center. To achieve the goal of the study, 15 female white rats weighing 160-180 grams, previously unborn, not bred, and a total of 220 (control group - 100, experimental group - 120) rat pups born from them were taken. Experimental animals were kept under normal laboratory diet conditions.To achieve the goal, 120 white laboratory rats were used. White laboratory rats were divided into 2 groups. Group 1 consisted of 7 healthy rats as a control group. Mother rats in the control group were given 1.0 ml of saline solution every morning into the stomach. A subclavian catheter was used as a probe. The rats were anesthetized under ether anesthesia on days 7, 14, 21, 30 and 60 after birth. Group 2 was the experimental group, and 8 white female laboratory rats were kept in specially prepared cages to induce experimental metabolic syndrome. This metabolic syndrome model was continued after the rats became pregnant and gave birth to their offspring. For this purpose, the forebrain was prepared in a frontal direction using standard histological sections, and the cell density in pyramidal layers II–III of the prefrontal cortex was calculated by microscopy. The number of neurons in 5 fields from each sample was counted in an area of 1000 μm², and the mean value ± standard deviation was expressed. A Zeiss microscope and ImageJ software were used for morphometry. The tissues were fixed in 10% neutral formalin solution for 24 hours, embedded in paraffin blocks, and 5 μm-thick sections were prepared using a microkeratome. The sections were stained with hematoxylin-eosin (to assess the general histological structure, tissue and nuclear-cytoplasmic ratio) and Masson trichrome (to determine the density of connective tissue and collagen fibers). Table 1 compares the body weight, brain weight and morphometric parameters of offspring born to control and metabolic syndrome mothers at 3, 7, 14, 21, 30 and 60 days of age. According to the results of the analysis, the body weight of the offspring of the experimental group was higher than that of the control group at all age stages. On the 3rd day of postnatal ontogenesis, the body weight of the control group was 7 g, while in the experimental group it was 8 g; at 30 days, it was 93 g and 110.3 g, respectively.
 | Table 1. Cerebral parameters of laboratory rat pups in the control group |
3. Results and Discussions
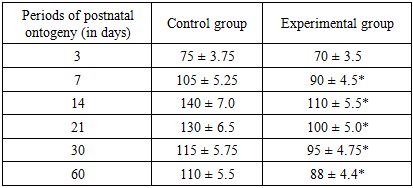
- When comparing the cerebral cortex of the rats in the control and experimental groups, it was found that by the 14th day of the experiment, neurocytes had lost their typical pyramidal shape. These cells had acquired a long shape with no clear boundaries. On the 14th day of the experiment, it was found that the density of neurons in the rats born to mothers with metabolic syndrome was higher than in the control group. When comparing the histological structure of the brains of the offspring born to mothers in the experimental and control groups, it was found that by the 14th day, the cortex was fully formed. In the rats in the experimental group, as in the control group, bipolar cells with 2-4 nuclei were detected in the cerebral cortex. The 2nd and 3rd layers of the cerebral cortex are densely located. At this time, the difference in the brains of the rats in the control and experimental groups is not noticeable. The cortical neurocytes in the experimental group are relatively round and smaller in size than those in the control group. These neurocytes have a small rim in their cytoplasm, and the nucleus is brightly colored and relatively larger in size. On day 14, the prefrontal cortex of the control group is characterized by high cell density, a clear layered structure, and active synaptogenesis.However, in the experimental group of children born to mothers with metabolic syndrome, the cell density remains low, and the number of mature neurons in layers II–III is statistically significantly reduced (p<0.05).In control animals, basophilic substance is formed in the form of flakes in the apical and basal zones of the cytoplasm of large cells, while in experimental animals, the cytoplasm of such cells is diffusely stained with thionin, and Nissl substance is located in the perinuclear area of the cytoplasm.The brains of the offspring born to rats from the control group are fully formed by day 21, the morphological structure of neurocytes is fully formed. In most of the cells of various sizes, both large and small, in different layers of the cortex, basal growths are clearly visible, and the nuclei are mainly round in shape.The neurocytes in the brains of the offspring in the 21-day experimental group were pale in color, with poorly developed cytoplasm, round in shape, and neurocytes were often located perinuclearly, and in some neurocytes they were located in the basal region. Neuroocytes had a rough structure, and the boundaries of the nucleus were clear. By this period of the experiment, a decrease in neurocytes was observed. It was found that the sizes of neurocytes were smaller than those in the control group at this day of the experiment. This was especially clearly reflected in the cytoplasm. The boundaries of the cytoplasm and nucleus were not clear when compared with the control group. The location of the basophilic substance in the cytoplasm of neurocytes located in the hippocampus differed from that in the control group. It was found that the basophilic substance was located in the center of the cytoplasm, and the periphery was more fluid. By the 21st day of the experiment, pathologically changed cells began to be detected in the hippocampus. Here, solitary cells with a whitish cytoplasm and a swollen nucleus could be found.By the 30th day of the experiment, it was found that large and medium-sized neurocytes of the hippocampus were vacuolated, and there was a perinuclear swelling in their cytoplasm. The boundaries of the nuclei of these cells were clearly defined, and the nucleus was visible in the form of a small black dot. At this time, shadow cells of neurocytes were detected. At this time, focal lysis of neurocytes was detected.By the 60th day of the experiment, the structure of neurocytes in the experimental group was different from that in the control group. By this time, cells with vacuolated cytoplasm were found singly, and the cytoplasm of neurocytes mainly had a reticular structure. Basophilic substance was located around the nucleus. The nuclei of these cells were found to be of a fluid color. Their size was increased compared to that in the control group. Chromatin balls were found to be located in most cases on the periphery of the nucleus. By the 60th day, the cellular architecture of the prefrontal layer in the control group was stabilized and layered structures were fully formed. In the experimental group, the cell density is much less compared to the control, there is a violation of the layer structure, a decrease in the number of pyramidal neurons, and an increase in glial elements. This indicates that the process of chronic neuroinflammation continues in the brain tissue.Thus, the results of this study showed that offspring born to mothers with metabolic syndrome experience morphological changes in the cerebral cortex.Table 2 consistent increase in cell density was observed in the control group at different periods of postnatal ontogenesis: on the 3rd day — 75 thousand h/mm², on the 7th day — 105 thousand h/mm², on the 14th day — 140 thousand h/mm², on the 21st day — 130 thousand h/mm², on the 30th day — 115 thousand h/mm² and on the 60th day — 110 thousand h/mm². In the animals of the experimental group (children born to mothers with metabolic syndrome), these indicators were lower: 70, 90, 110, 100, 95 and 88 thousand h/mm², respectively.
|
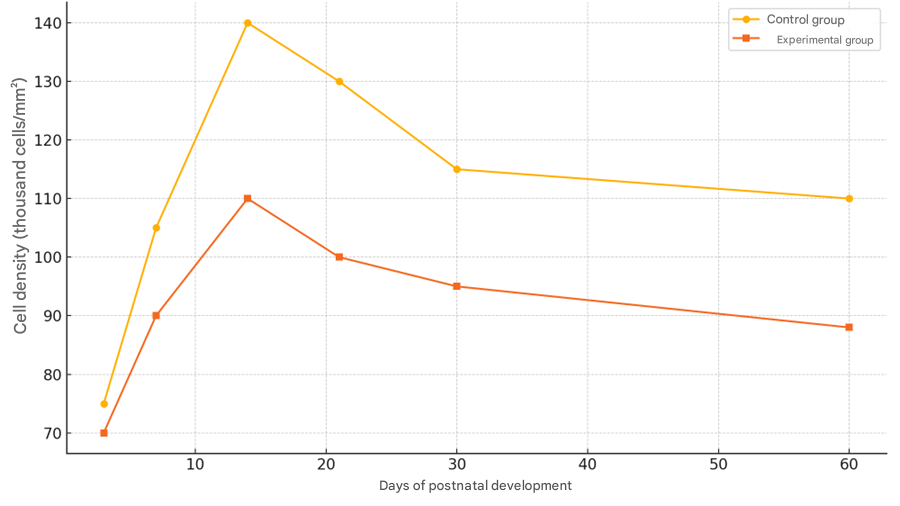 | Figure 1. Dynamics of neuronal growth of cells in the prefrontal layer |
4. Conclusions
- The results of the study clearly confirm that brain morphogenesis delays and disorders may occur in laboratory rat offspring born to mothers with metabolic syndrome. In particular, the statistical and morphometric differences in the ratio of total body weight to brain weight (brain-to-body ratio) and cell density in the prefrontal cortex indicate that the metabolic state of the mother may be associated with neurotrophic changes in the child. At all stages of postnatal ontogenesis (3, 7, 14, 21, 30 and 60 days), the brain dimensions, longitudinal and transverse lengths of the hemispheres, as well as cell density in the experimental group of rat pups were lower than in the control group. In particular, changes were clearly manifested at the 14 and 60-day periods, when a 20–25% decrease in cell number was observed (p<0.05). This indicates a negative effect on the processes of neurogenesis, migration, synaptogenesis and differentiation of neurons.It was also noted that the morphological indicators of the cerebral hemispheres in the experimental group of rat pups deviated from the criteria of normal development. According to the results of morphometry, in children affected by metabolic syndrome, the hemispheres lag behind in anatomical development, a decrease in the number of pyramidal neurons, and an increase in glial infiltration were observed. These changes can lead to a decrease in cognitive abilities, attention deficit, and behavioral problems in children.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML