-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(9): 3192-3196
doi:10.5923/j.ajmms.20251509.78
Received: Aug. 27, 2025; Accepted: Sep. 25, 2025; Published: Sep. 29, 2025

Protein-Energy Malnutrition Problem Among Children in Uzbekistan
Abdullaeva Dilafruz Gayratovna1, Khalilova Nargiza Kurbanovna2
1Doctor of Medical Sciences, Associate Professor, Department of Pediatric Diseases, Tashkent State Medical University, Ministry of Health Republic of Uzbekistan
2Independent Applicant, National Children’s Medical Center, Tashkent, Uzbekistan
Correspondence to: Abdullaeva Dilafruz Gayratovna, Doctor of Medical Sciences, Associate Professor, Department of Pediatric Diseases, Tashkent State Medical University, Ministry of Health Republic of Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Protein-energy malnutrition is a common childhood disorder and is primarily caused by deficiency of energy, protein, and micronutrients. Materials and Methods. In accordance with the principles of Good Clinical Practice, a single-center, non-comparative cross-sectional study was conducted. The study included 85 children scheduled for medium- or large-scale surgical intervention in the surgical department of the National Children’s Medical Center and the clinic of Tashkent State Medical University. Inclusion Criteria: children of both sexes aged from 1 month to 8 years. Results. We studied the level of albumin and prealbumin in the blood serum. Prealbumin is a small protein which has been widely evaluated as a nutritional and a prognostic marker. The small size and concentration of prealbumin in blood proposes challenges on measuring it with high sensitivity and specificity. Conclusion. Thus, adequate correction of nutritional disorders underlies normal growth and development rates of a child, supports learning ability and determines the resistance of a child's body to unfavorable environmental factors. Nutritional support of children is an integral factor in maintaining health and improving the quality of life.
Keywords: Protein-energy malnutrition, Albumin, Prealbumin, Nutritional status
Cite this paper: Abdullaeva Dilafruz Gayratovna, Khalilova Nargiza Kurbanovna, Protein-Energy Malnutrition Problem Among Children in Uzbekistan, American Journal of Medicine and Medical Sciences, Vol. 15 No. 9, 2025, pp. 3192-3196. doi: 10.5923/j.ajmms.20251509.78.
1. Introduction
- Nutritive status is one of the essential pointers for crucial signs of health. Malnutrition is a public health problem of significant significance in growing countries [25]. Protein-Energy Malnutrition (PEM) has been recognized for over 60 years; the term was first used by FAO/WHO nutrition experts. PEM refers to a prolonged state of protein and/or energy deficiency, which may be accompanied by underweight and/or stunted growth. This condition is associated with metabolic disturbances and imbalances in water-electrolyte homeostasis. Furthermore, patients with PEM often experience dysfunctions in the endocrine, immune, digestive, and nervous systems. According to official statistics from the WHO, PEM accounts for about 45% of the causes of child mortality annually. By the end of 2017, about 16 million children under 5 years of age worldwide suffer from acute malnutrition and about 22% of children have chronic protein-energy malnutrition [36].Protein-energy malnutrition (PEM) is a common childhood disorder and is primarily caused by deficiency of energy, protein, and micronutrients. PEM manifests as underweight (low body weight compared with healthy peers), stunting (poor linear growth), wasting (acute weight loss), or edematous malnutrition (kwashiorkor). Case fatality rates among children hospitalized with severe wasting or edema (also known as severe acute malnutrition [SAM]) range from 5% to 30% [32,35].
2. Materials and Methods
- In accordance with the principles of Good Clinical Practice, a single-center, non-comparative cross-sectional study was conducted.
3. Results and Discussion
- Eligibility CriteriaThe study included 85 children scheduled for medium- or large-scale surgical intervention in the surgical department of the National Children’s Medical Center and the clinic of Tashkent State Medical University.Inclusion Criteria: children of both sexes aged from 1 month to 8 years.Study SettingNational Children’s Medical Center and the clinic of Tashkent State Medical University.Study Duration: June 2025 – August 2025.
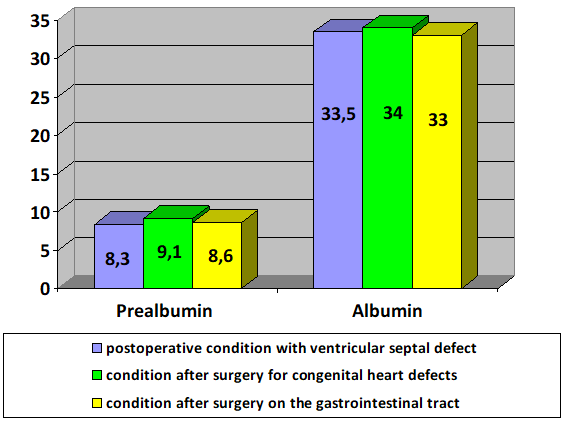 | Figure 1. Before treatment (n=85), % |
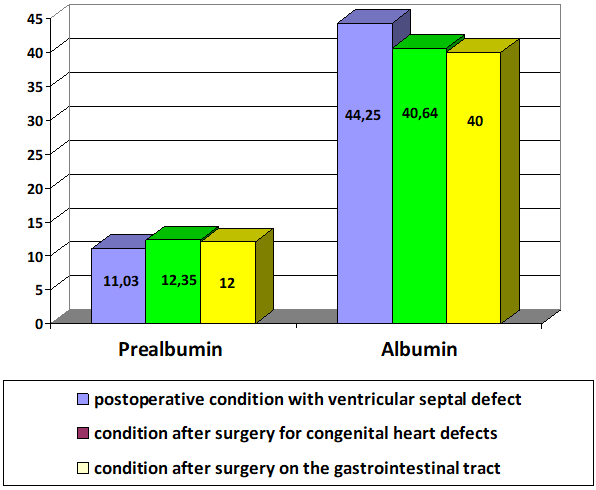 | Figure 2. After treatment (n=85), % |
4. Conclusions
- Thus, adequate correction of nutritional disorders underlies normal growth and development rates of a child, supports learning ability and determines the resistance of a child's body to unfavorable environmental factors. Nutritional support of children is an integral factor in maintaining health and improving the quality of life.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML