-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(9): 3178-3183
doi:10.5923/j.ajmms.20251509.75
Received: Sep. 9, 2025; Accepted: Sep. 25, 2025; Published: Sep. 29, 2025

Asymmetric Dimethylarginine and Metabolic Dysregulation: Novel Insights into Links with Insulin Resistance and Atherogenic Dyslipidemia
Munisakhon M. Makhkamova1, Nargiza M. Nurillayeva2
1PhD Researcher at the Department of “Internal Diseases in Family Medicine and Fundamentals of Preventive Medicine №1” at Tashkent State Medical University, Tashkent, Uzbekistan
2DSc, Professor, Head of the Department of “Internal Diseases in Family Medicine and Fundamentals of Preventive Medicine №1” at Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

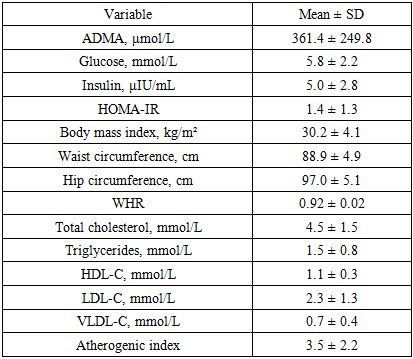
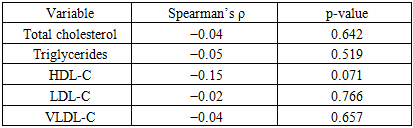
Objective. To examine associations of asymmetric dimethylarginine (ADMA) with metabolic biomarkers reflecting insulin resistance and dyslipidemia in patients with cardiometabolic disorders. Methods. The study included 149 patients. In all participants, ADMA concentrations were measured by ELISA; indices of carbohydrate metabolism (glucose, insulin, HOMA-IR), anthropometrics (BMI, waist circumference, WHR), and lipid profile (total cholesterol, triglycerides \[TG], HDL-C, LDL-C, VLDL-C, atherogenic index) were assessed. Correlation analysis was performed using Spearman’s rank coefficient. Results. The median ADMA level was 241.7 μmol/L (Q1–Q3: 152.1–591.4). Statistically significant positive correlations were observed between ADMA and insulin (ρ=0.18; p=0.026), HOMA-IR (ρ=0.19; p=0.019), body mass index (ρ=0.23; p=0.005), and waist circumference (ρ=0.16; p=0.047). Associations with glucose, hip circumference, and WHR were not statistically significant. No significant correlations were identified within the lipid profile, except for a trend toward an inverse association with HDL-C (ρ=−0.15; p=0.071). Conclusions. These findings support the role of ADMA as a biomarker reflecting the interplay between endothelial dysfunction and components of the metabolic syndrome. The strongest associations were observed with indices of insulin resistance and adiposity.
Keywords: Asymmetric dimethylarginine, Insulin resistance, HOMA-IR, Dyslipidemia, Endothelial dysfunction, Metabolic syndrome, Biomarkers
Cite this paper: Munisakhon M. Makhkamova, Nargiza M. Nurillayeva, Asymmetric Dimethylarginine and Metabolic Dysregulation: Novel Insights into Links with Insulin Resistance and Atherogenic Dyslipidemia, American Journal of Medicine and Medical Sciences, Vol. 15 No. 9, 2025, pp. 3178-3183. doi: 10.5923/j.ajmms.20251509.75.
Article Outline
1. Introduction
- Cardiometabolic disorders converge on a web of metabolic and vascular abnormalities in which insulin resistance, atherogenic dyslipidemia, and endothelial dysfunction reinforce one another to accelerate atherosclerotic risk [1] [16]. (Picture 1) Asymmetric dimethylarginine (ADMA), an endogenous inhibitor of nitric oxide synthase, has been advanced as both a biomarker and mediator within this nexus because it impairs nitric-oxide–dependent endothelial signaling [2]. Foundational clinical and experimental studies have linked higher circulating ADMA to impaired endothelium-dependent vasodilation and to pro-atherogenic vascular phenotypes across diverse populations [3].
 | Picture 1. Risk factors for endothelial cell dysfunction and its pathological consequences. Created with Smart Servier Medical Art |
2. Methods
- This prospective observational study enrolled patients with ischemic heart disease (IHD) presenting with stable angina of functional class I–II. Follow-up was conducted from January 1, 2024 to June 1, 2025 at the Multidisciplinary Clinic of the Tashkent State Medical University (TSMU) and the Republican Specialized Scientific and Practical Medical Centre of Cardiology (RSSPMC Cardiology). The study cohort comprised 149 patients with IHD, functional class I–II. A control group included 20 apparently healthy individuals without clinical or instrumental signs of cardiovascular disease.At the initial evaluation, medical history and physical examination were performed, with systematic assessment of IHD symptoms (angina episodes, exercise tolerance). Liver status was evaluated by abdominal ultrasonography or FibroScan. Laboratory testing included routine hematology and biochemistry with alanine aminotransferase (ALT), aspartate aminotransferase (AST), a complete lipid profile, and indices of glucose metabolism. Cardiac function was assessed by a standard 12-lead electrocardiogram (ECG) and transthoracic echocardiography.Inclusion criteria. Confirmed diagnosis of IHD with stable effort angina, functional class I–II. Age 35 to 75 years; Provision of written informed consent to participate. Exclusion criteria. Unstable angina, acute coronary syndrome, or prior myocardial infarction; Chronic heart failure of New York Heart Association (NYHA) class III–IV or left ventricular ejection fraction < 40%; Severe comorbid conditions potentially affecting ADMA levels or endothelial function, including chronic kidney disease with estimated glomerular filtration rate (eGFR)<45 mL/min/1.73 m²; liver cirrhosis or severe chronic liver diseases, active malignancy, or pronounced systemic inflammatory/autoimmune disorders; Decompensated type 1 diabetes mellitus; poorly controlled type 2 diabetes mellitus with HbA1c > 9% at enrollment (such patients required treatment optimization prior to evaluation); Use of agents known to influence ADMA metabolism within 4 weeks prior to study procedures (e.g., high-dose L-arginine, phosphodiesterase-5 inhibitors, etc.).In all participants, general clinical information was obtained and anthropometry was performed: body weight (kg) and height (cm) were measured to calculate body mass index (BMI, kg/m²), waist circumference (cm) and hip circumference (cm) were recorded to derive the waist-to-hip ratio (WHR). Criteria for abdominal obesity were applied as follows: waist circumference ≥94 cm in men and ≥80 cm in women. The atherogenic index (coefficient of atherogenicity) was calculated as (total cholesterol − HDL-cholesterol) / HDL-cholesterol. Fasting venous blood was collected from all subjects for biochemical analyses. Serum ADMA concentration was measured by enzyme-linked immunosorbent assay (ELISA) using a commercial kit (Beijing Ltd., China) and expressed in μmol/L. Plasma glucose was determined by the hexokinase method (mmol/L). Immunoreactive insulin was measured by ELISA (μIU/mL). Insulin resistance was assessed using the HOMA-IR index, calculated as: glucose (mmol/L) × insulin (μIU/mL) / 22.5. The serum lipid profile (mmol/L) included triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and calculated low-density lipoprotein cholesterol (LDL-C). Lipid measurements were performed using enzymatic colorimetric methods on an automated analyzer. All measurements were conducted in a single certified laboratory with adherence to internal and external quality control procedures.Statistical analyses were performed using MS Excel. Correlations were assessed using Spearman’s rank correlation coefficient (ρ). Statistical significance for correlations was set at p<0.05. Where indicated, multiple regression analysis (linear regression) was employed to identify independent predictors of ADMA levels, entering variables that were significant in univariable analyses.
3. Results
- A total of 149 individuals were examined, of whom 55.7% were women and 44.3% were men. The median age was 46 years [38–54]. The median BMI was 30.2 kg/m² [27.8–32.7], corresponding to excess body weight on average in the cohort. The median waist circumference was 88.5 cm [85.8–92.1]; in 57% of participants it exceeded sex-specific thresholds, indicating abdominal obesity. The waist-to-hip ratio (WHR) was 0.914 [0.902–0.929], exceeding the conventional normal values (0.85 for women, 0.90 for men) in most participants.The median serum ADMA concentration was 361.4 ± 249.8 nmol/L. Median fasting plasma glucose was 5.1 mmol/L [4.4–6.4]; in 13% of patients, glycemia exceeded 7 mmol/L, suggestive of impaired glucose tolerance or diabetes. Median immunoreactive insulin was 3.8 μIU/mL [3.1–5.9], and HOMA-IR was 0.95 [0.64–1.57]. Elevated HOMA-IR > 2.5 (insulin-resistance threshold) was observed in 11% of participants. The lipid profile showed overall moderate abnormalities: total cholesterol 4.0 mmol/L [3.5–4.8], triglycerides 1.50 mmol/L [1.10–1.90], HDL-C 1.00 mmol/L [0.95–1.22], and calculated LDL-C 2.22 mmol/L [1.55–3.03]. In 42% of participants, HDL-C was below target (< 1.0 mmol/L in men and < 1.2 mmol/L in women). The atherogenic coefficient ranged from −0.39 to 16.5 (median 3.20 [2.06–4.49]). Values above the recommended limit of 3.0 were recorded in more than half of the cohort (56%), indicating a high prevalence of atherogenic dyslipidemia in the sample.In the correlation analysis, statistically significant associations were identified between ADMA levels and several indices of metabolic status. Serum ADMA concentration showed a significant positive correlation with fasting insulin (Spearman’s ρ=0.18; p=0.026) and with HOMA-IR (ρ=0.19; p=0.019), indicating that higher ADMA levels were associated with greater insulin resistance. Table 1 presents a scatterplot illustrating the relationship between ADMA and HOMA-IR, demonstrating an upward trend in HOMA-IR with increasing ADMA concentration. Positive correlations were also observed between ADMA and measures of general and central adiposity-BMI (ρ=0.23; p=0.005) and waist circumference (ρ=0.16; p=0.047). Thus, individuals with greater excess weight tended to exhibit higher ADMA levels. (Table 1)
|
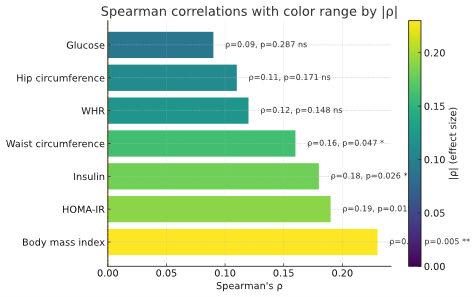 | Figure 1. Correlations of ADMA with metabolic and anthropometric variables |
|
4. Discussion
- In this cohort of patients with stable IHD, circulating ADMA tracked most consistently with indices of insulin resistance and adiposity, while associations with standard lipid parameters were weak. ADMA showed small but statistically significant positive correlations with fasting insulin and HOMA-IR, as well as with BMI and waist circumference, indicating that higher ADMA levels co-occur with greater insulin resistance and central obesity. By contrast, correlations with triglycerides, LDL-C, VLDL-C and the atherogenic index were null, with only a borderline inverse trend for HDL-C. Taken together, these findings suggest that ADMA aligns more closely with the insulin-resistant/obesity phenotype than with conventional cholesterol fractions. Multivariable analyses reinforced this pattern: when BMI and HOMA-IR (or fasting insulin) were modeled together, BMI remained the sole independent predictor of ADMA, pointing to adiposity as the dominant correlate of circulating ADMA in this dataset. A plausible mechanistic interpretation is that excess adiposity-via low-grade inflammation, oxidative stress, and altered DDAH/NO signaling-elevates ADMA and impairs endothelial function, which in turn exacerbates insulin resistance.Several considerations temper inference. The cross-sectional design precludes causal conclusions; medication use and unmeasured confounders (e.g., renal function, systemic inflammation) may attenuate lipid associations; and ELISA-based quantification can differ from chromatographic methods. Unit harmonization for ADMA (μmol/L vs nmol/L) should be verified across text and tables. Clinically, ADMA may have value as an adjunctive marker of cardiometabolic risk-particularly in patients with central obesity and insulin resistance-yet its incremental utility beyond established measures requires prospective validation and, ideally, interventional studies targeting weight reduction, insulin sensitivity, or the DDAH/NO axis.
4.1. Study Limitations
- This analysis is cross-sectional, which precludes establishing causal relationships. It is therefore not possible to determine whether elevated ADMA levels cause insulin resistance and vascular dysfunction or arise secondarily in response to metabolic perturbations. Longitudinal studies are needed to clarify whether ADMA predicts the development of diabetes and atherosclerosis. In addition, the sample comprised a relatively heterogeneous group without strict clinical subclassification; future work should examine associations between ADMA and metabolic markers in more defined cohorts (e.g., patients with metabolic syndrome or with diabetes). Another limitation is the use of ELISA for ADMA measurement-while convenient, chromatographic methods (e.g., HPLC/LC–MS) provide greater analytical specificity. Nevertheless, our findings are broadly consistent with prior studies and are biologically plausible.
5. Conclusions
- In this study, we identified a close relationship between circulating ADMA—an endogenous inhibitor of nitric oxide synthase—and metabolic disturbances characteristic of insulin resistance and dyslipidemia. Higher ADMA concentrations were associated with elevated fasting insulin and HOMA-IR, as well as with measures of adiposity (BMI and waist circumference), reflecting the coupling of endothelial dysfunction with insulin-resistant states. Although direct associations between ADMA and individual lipid parameters were weaker, we observed a trend toward an inverse relationship with HDL-C, consistent with the putative role of ADMA in atherogenesis. These findings add to a growing body of evidence that positions ADMA not merely as a passive marker but as a potential mediator of vascular injury in the context of metabolic disease. Mechanistically, elevated ADMA may contribute to reduced nitric oxide bioavailability, impaired vasodilation, and pro-inflammatory signaling, all of which accelerate atherosclerotic changes. The consistent associations across multiple indices of insulin resistance and adiposity suggest that ADMA could serve as an integrative indicator of cardiometabolic stress.From a practical standpoint, ADMA may be considered a promising biomarker of cardiometabolic risk in patients with insulin resistance and related disorders such as type 2 diabetes or metabolic syndrome. Incorporating ADMA measurement into clinical risk stratification could help identify individuals at heightened risk for early vascular complications. Nevertheless, the observational nature of our study limits inference about causality. Definitive confirmation of a causal role for ADMA will require long-term prospective cohorts and randomized interventional trials aimed at lowering ADMA levels—through lifestyle modification, pharmacologic agents, or targeted enzyme modulation—and evaluating the downstream effects on metabolic and vascular outcomes. Future research should also explore whether genetic or epigenetic determinants of ADMA metabolism influence susceptibility to insulin resistance and cardiovascular disease, which could open avenues for precision-based prevention and therapy.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML