-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(9): 2909-2913
doi:10.5923/j.ajmms.20251509.13
Received: Jul. 24, 2025; Accepted: Aug. 19, 2025; Published: Sep. 8, 2025

The Role of Prenatal Screening Programs and Non-Invasive Prenatal Screening in the Diagnosis of Fetal Chromosomal Abnormalities in the First Trimester
Nigmatulina Ilmira Ildarovna
Center for the Development of Professional Qualifications of Medical Workers, Tashkent, Uzbekistan
Correspondence to: Nigmatulina Ilmira Ildarovna, Center for the Development of Professional Qualifications of Medical Workers, Tashkent, Uzbekistan.
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The aim of the study was to conduct a comparative analysis of the diagnostic efficiency of the “Astraia Obstetrics” combined first trimester prenatal screening program in pregnant women with high and medium risk of fetal chromosomal abnormalities in comparison with non-invasive prenatal testing. The history of early prenatal screening is a fascinating journey of medical achievements and technological advances aimed at identifying possible chromosomal abnormalities and other congenital diseases in the fetus, allowing expecting parents and doctors to prepare for possible problems in advance. The sensitivity of Astraia Obstetrics was determined to be 96.06% with a specificity of 96.1% at high risk; at above-average risk, sensitivity was 87.52% with a specificity of 88.4%. The sensitivity of the non-invasive prenatal test at high risk was 98.24% with a specificity of 98.2%; in the above-average risk group, sensitivity was 95.42% with a specificity of 95.1%.
Keywords: “Astraia” first trimester prenatal screening program, Non-invasive prenatal test, Efficiency
Cite this paper: Nigmatulina Ilmira Ildarovna, The Role of Prenatal Screening Programs and Non-Invasive Prenatal Screening in the Diagnosis of Fetal Chromosomal Abnormalities in the First Trimester, American Journal of Medicine and Medical Sciences, Vol. 15 No. 9, 2025, pp. 2909-2913. doi: 10.5923/j.ajmms.20251509.13.
Article Outline
1. Introduction
- The history of early prenatal screening is a fascinating journey of medical achievements and technological advances aimed at identifying possible chromosomal abnormalities and other congenital diseases in the fetus, allowing expecting parents and doctors to prepare for possible problems in advance [1].The rapid development of prenatal screening occurred in the mid-20th century with the advent of ultrasound. The founder was Ian Donald, who first used ultrasound to visualize the fetus and diagnose abnormalities, albeit only in the form of static images [2]. By the 1970s, thanks to the advent of real-time observation of fetal movements, ultrasound had already become the standard for prenatal diagnostics of fetal pathologies and developmental abnormalities [3]. The measurement of nuchal translucency (NT) began at 11-14 weeks of gestation, and its diagnostic value was proven in the 1990s in terms of increasing the risk of the following pathologies: Down syndrome (DS), Patau syndrome (PS), Edwards syndrome (ES), Turner syndrome, which in turn led to the widespread use of this method in combined first trimester screening [4]. The main purpose of NT measurement is to determine the risk of chromosomal abnormalities (CA) and congenital malformations (CM) in the fetus. Currently, NT screening detects 66.7-92% of CA and CM in early gestation and significantly improves the diagnostic quality of prenatal screening [4].In the 1990s, along with NT determination by ultrasound, the detection of biochemical markers—free beta-human chorionic gonadotropin (β-hCG) and free unconjugated estriol (uE3)—as well as the pregnancy-associated AFP test (α-fetoprotein) associated with pregnancy, led to a significant increase in the accuracy of diagnosing chromosomal abnormalities [5].Modern first trimester prenatal diagnostics is an integral part of obstetric dynamic monitoring, represented by several programs, one of which is Astraia Obstetrics (Astraia software gmbH, Germany), developed relatively recently with the support of the FMF (Fetal Medicine Foundation – FMF) (London, UK), which has undergone several clinical trials on a large number of pregnant women in different countries and is constantly being optimized [6]. According to the “Astraia Obstetrics” program, pregnant women with a risk of 1 ≤ 101 are classified as low risk, while those with a probability of 1 ≥100 are classified as high risk [6].Another leap forward in prenatal screening took place at the turn of the millennium with the advent of non-invasive prenatal testing (NIPT), which is ten times more efficient than conventional biochemical screening. It can be performed before biochemical screening and before the end of pregnancy, but it has almost 100% sensitivity only for Down syndrome; for other chromosomal abnormalities - 92-97%, while the 99% accuracy of NIPT is a population indicator, and not an individual test accuracy [7].The aim of the study was to conduct a comparative analysis of the diagnostic efficiency of the “Astraia Obstetrics” combined first trimester prenatal screening program in pregnant women with high and medium risk of fetal chromosomal abnormalities in comparison with non-invasive prenatal testing.
2. Material and Methods
- Clinical observations were carried out at the Maternal and Child Diagnostic Center “Aliev`s Family” in Tashkent, where 46 pregnant women were examined in the period 2022-2024 after receiving the results of comprehensive prenatal screening under the Astraia Obstetrics program. The age of the patients was 26-38 years (mean age – 32.8±3.89 years) at a period of 11-13+6 days weeks with a crown-rump length (CRL) of the fetus of 45-84 mm. The following research methods were used in our study: • instrumental examination methods (ultrasound with fetal fetometry and Doppler ultrasound of the venous duct and tricuspid valve of the fetal heart); • results of the prenatal screening program "Astraia Obstetrics";• NIPT;• invasive prenatal diagnostics (IPD) (amniocentesis) to determine the karyotype of the fetus using the fluorescence in situ hybridization (FISH) method;• statistical methods for processing the obtained research results.
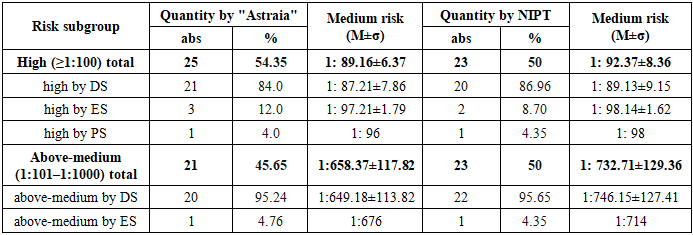
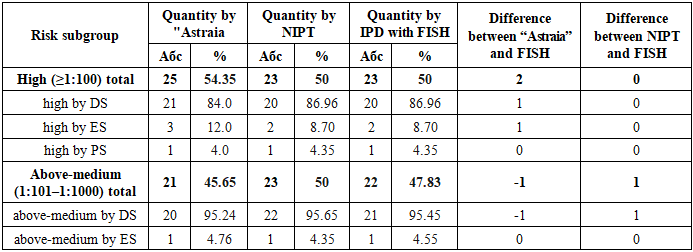
3. Results
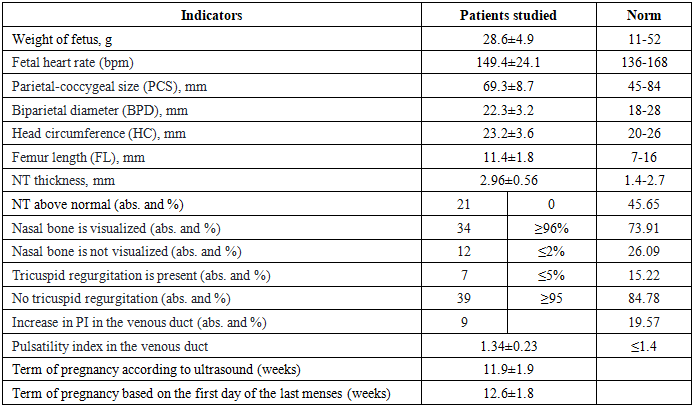
- Ultrasound fetometry of the fetus performed by the transabdominal method at 11-13+6days weeks of gestation in the pregnant women studied allowed us to confirm singleton pregnancy in all cases and various deviations from the norm according to many indicators (Tab. 1).
|
|
|
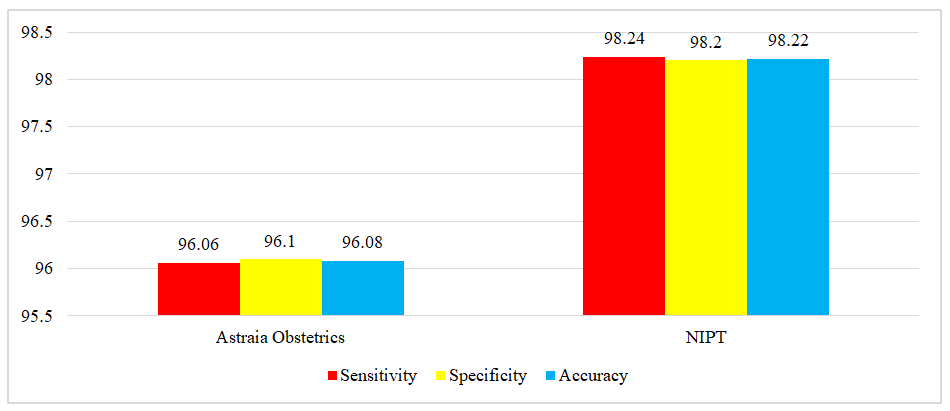 | Figure 1. Diagnostic indicators of the "Astraia" prenatal screening program and NIPT in the high-risk group, % |
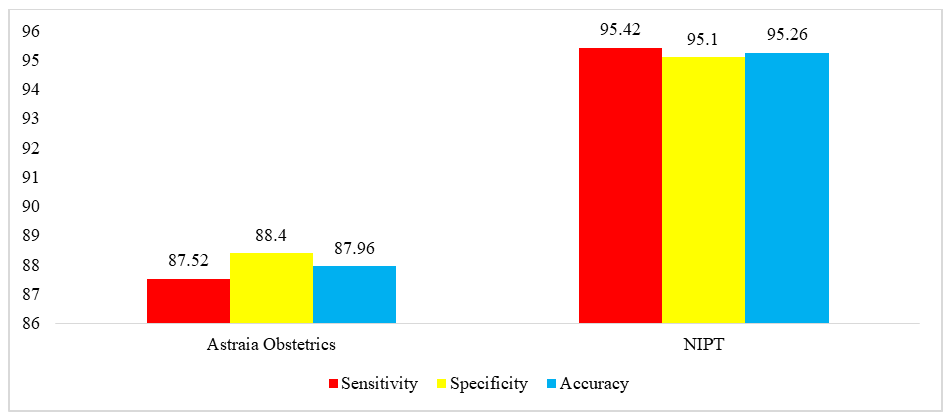 | Figure 2. Diagnostic indicators of the "Astraia" prenatal screening program and NIPT in the above-medium group, % |
4. Conclusions
- It is possible to determine the risk level of fetal chromosomal abnormalities (CA) with high probability by using NIPT at the primary diagnostic stage before or in parallel with FISH results, especially Down syndrome (DS) in women of any age up to 14 weeks. Regression analysis suggests that NIPT results can be used independently for screening the risk of developing fetal CA.The diagnostic accuracy of NIPT for determining the risk of fetal CA development that we have established is an important contribution to the methodology of complex screening studies of pregnant women, especially those with two or more risk factors for this pathology and those located in remote areas, which will be useful for diagnosing CA in the practice of obstetricians and gynecologists.
Conflict of Interests’ Statement
- The author declares no conflict of interest. This study does not include the involvement of any budgetary, grant or other funds. The article is published for the first time and is part of a scientific work.
ACKNOWLEDGEMENTS
- The author expresses their gratitude to the management of the Maternal and Child Diagnostic Center “Aliev`s Family” for the material provided for our study.
Ethical Approval and Consent to Participate
- The Research Ethics Board of our institution does not require review or approval of case reports. Our research was carried out in accordance with the World Medical Association Code of Ethics (Declaration of Helsinki).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML