-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2799-2802
doi:10.5923/j.ajmms.20251508.80
Received: Aug. 8, 2025; Accepted: Aug. 27, 2025; Published: Aug. 30, 2025

Association of Clinical Forms and Hemostatic Disturbances in Pediatric Henoch–Schönlein Purpura
Turdikul A. Bobomuratov, Guloyim S. Avezova, Nafisa S. Sultanova
Tashkent Medical Academy, Tashkent, Uzbekistan
Correspondence to: Guloyim S. Avezova, Tashkent Medical Academy, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background. Henoch–Schönlein purpura (HSP), or IgA vasculitis, is the most common systemic vasculitis in children, characterized by a broad spectrum of clinical manifestations ranging from cutaneous and joint involvement to renal and gastrointestinal complications. Despite its usually self-limiting course, a subset of patients develop severe or recurrent forms, highlighting the need for deeper understanding of clinical patterns and prognostic markers. Objectives. This study aimed to analyze the clinical, laboratory, and biochemical features of pediatric HSP in order to assess the frequency of different disease forms, seasonal distribution, and laboratory abnormalities associated with disease severity. Methods. A total of 223 children diagnosed with HSP were enrolled. Clinical forms were classified into cutaneous, cutaneous-articular, mixed, and nephritic variants. Laboratory analysis included hematological, biochemical, and hemostatic parameters, with emphasis on correlations between disease phenotype and laboratory abnormalities. Data were compared with previous research findings to assess similarities and differences. Results. The cutaneous-articular form predominated (53%), followed by mixed (29%), cutaneous (12%), and nephritic (6%) forms. Abdominal syndrome was observed in 22% and nephritic syndrome in 7% of patients, with disease onset more frequent in autumn and spring. Laboratory abnormalities included leukocytosis (35%), elevated ESR (28%), thrombocytosis (18%), thrombocytopenia (9%), dysproteinemia (58%), increased liver enzymes (48%), and renal impairment (6%). Hemostatic disturbances reflecting hypercoagulability were present in 63% of cases, particularly in nephritic and mixed forms. Conclusions. The findings indicate that severe and recurrent forms of HSP are strongly associated with nephritic and abdominal syndromes, thrombocytopenia, and hypercoagulable states. These results underscore the prognostic importance of laboratory monitoring and support the integration of hematological, biochemical, and hemostatic markers into clinical assessment to predict disease severity and outcomes.
Keywords: Henoch–Schönlein purpura, IgA vasculitis, Children, Clinical forms, Laboratory markers, Nephritis, Hypercoagulability, Prognostic factors
Cite this paper: Turdikul A. Bobomuratov, Guloyim S. Avezova, Nafisa S. Sultanova, Association of Clinical Forms and Hemostatic Disturbances in Pediatric Henoch–Schönlein Purpura, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2799-2802. doi: 10.5923/j.ajmms.20251508.80.
1. Introduction
- Henoch–Schönlein purpura (HSP), or hemorrhagic vasculitis, is the most common systemic vasculitis in children, characterized by damage to the walls of small blood vessels and deposition of immune complexes. The disease usually begins with skin manifestations in the form of vasculitic purpura and may progress with joint, abdominal, or renal syndromes [1,5,9].According to the World Health Organization, vasculitides account for a significant proportion of systemic diseases in children. The annual incidence of HSP in children is estimated at 13–20 cases per 100,000, varying depending on climatic conditions, environmental factors, prevalence of infections, and nutritional influences.The etiology of HSP is multifactorial; in many cases, the disease develops after viral or bacterial infections of the upper respiratory tract. Food and drug allergens, insect bites, hypothermia, and stress factors are also reported as triggers [2,3,10,11,12].In the pathogenesis of HSP, the accumulation of immune complexes in the vascular wall, activation of the complement system, and disturbances in blood rheology and the hemostatic system play a crucial role. Notably, changes in the hemostatic system are of great importance in determining the severity of the disease and the development of complications.Aim of the study: To investigate the clinical and laboratory characteristics of HSP in children, with a particular focus on alterations in hemostatic parameters and their association with the disease course.
2. Materials and Methods
- The study was conducted in the Cardiorheumatology Department of the Multidisciplinary Clinic of Tashkent State Medical University during the years 2022–2024. A total of 223 children diagnosed with Henoch–Schönlein purpura (HSP) who met the clinical criteria were enrolled in the study.The study was designed as a prospective dynamic observation. All children were admitted during the acute phase of the disease and were followed up for a period of 6–12 months. The condition of the patients was evaluated based on clinical, laboratory, and instrumental data.Inclusion criteria: Children under 18 years of age;• Diagnosis of Henoch–Schönlein purpura according to the EULAR/PRINTO/PRES criteria (2010);• Clinical manifestations including cutaneous vasculitic purpura, and at least one of the following: joint, abdominal, or renal syndrome.Exclusion criteria: Other systemic rheumatologic or hematologic diseases;• Congenital forms of coagulopathy;• Renal involvement of different etiology (glomerulonephritis, pyelonephritis, etc.).General characteristics of patients. Among the 223 children included in the study, 118 (53%) were boys and 105 (47%) were girls. The age distribution was as follows: 1–3 years – 42 children (19%), 4–7 years – 112 children (50%), 8–11 years – 47 children (21%), and over 12 years – 22 children (10%).Clinical assessment. The condition of patients was evaluated according to clinical manifestations:ü Cutaneous syndrome (palpable purpura, necrotic elements);ü Articular syndrome (arthralgia, arthritis);ü Abdominal syndrome (abdominal pain, dyspeptic disturbances, gastrointestinal bleeding);ü Renal syndrome (proteinuria, hematuria, features of nephrotic syndrome).Laboratory investigations. All patients underwent standard laboratory evaluations:ü Complete blood count (CBC): hemoglobin, erythrocytes, leukocytes, platelets, erythrocyte sedimentation rate (ESR);ü Biochemical tests: total protein, protein fractions, ALT, AST, creatinine, urea;ü Immunological tests: IgA, IgG, IgM, complement components (C3, C4);ü Hemostasis tests: activated partial thromboplastin time (APTT), prothrombin index (PTI), fibrinogen levels, D-dimer, soluble fibrin monomer complexes (SFMC).Instrumental examinations. Ultrasound of the kidneys and abdominal organs;ü ECG and echocardiography in necessary cases.Treatment protocols. All patients received therapy according to the standard protocol:ü Bed rest and dietary regimen;ü Antiplatelet agents (dipyridamole, curantil);ü Anticoagulants (heparin, dosage depending on the clinical form);ü Pathogenetic therapy (enterosorbents, antihistamines, pentoxifylline);ü In severe cases, glucocorticosteroids (prednisolone at 2 mg/kg/day);ü In several cases, plasmapheresis and infusion of fresh frozen plasma were performed.Statistical analysis. All data were processed using SPSS version 26.0. Results were presented as mean ± standard error. Differences between groups were assessed by χ² test and Student’s t-test. A p-value <0.05 was considered statistically significant.
3. Results
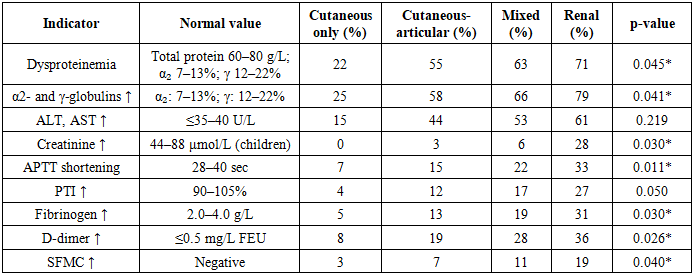
- A total of 223 children diagnosed with Henoch–Schönlein purpura (HSP) were included in the study. The following clinical forms of the disease were observed:• Cutaneous-articular form – 118 children (53%),• Mixed form – 64 children (29%),• Cutaneous form only – 27 children (12%),• Renal form – 14 children (6%).Abdominal syndrome was observed in 49 children (22%), while nephritic syndrome was recorded in 16 children (7%). In most cases, the disease debuted in the autumn (40%) and spring (28%) seasons.The clinical analysis of the enrolled patients revealed that the cutaneous-articular form was the most common clinical variant, occurring in 118 children (53%). This indicates a pronounced tropism of the disease towards vascular walls and small joints. The mixed form was diagnosed in 64 children (29%), suggesting the simultaneous manifestation of several syndromes and a relatively more severe course of the disease. The isolated cutaneous form was identified in 27 patients (12%), representing a milder variant. The renal form was found in 14 patients (6%) and is of particular clinical importance, as such cases have a higher probability of chronic progression and unfavorable outcomes.Analysis of additional syndromes revealed that abdominal syndrome was present in 49 patients (22%). This was mainly associated with mesenteric vascular involvement, which manifested clinically as acute abdominal pain, bloody diarrhea, or symptoms suggestive of peritonitis. Nephritic syndrome was observed in 16 patients (7%), and its presence was recognized as one of the most significant prognostic factors for recurrence and chronicity of the disease.The analysis of seasonal distribution demonstrated that the disease most frequently debuted in autumn (40%) and spring (28%). These findings are likely related to environmental triggers, particularly seasonal infections, allergic reactions, and changes in immune system reactivity. The lower incidence during winter and summer may be explained by reduced exposure to respiratory infections and variations in immunological reactivity.In summary, the findings of this study demonstrated that HSP manifests in diverse clinical forms, with varying frequencies of presentation. The presence of nephritic and abdominal syndromes is closely associated with a more severe disease course and an increased risk of recurrence.Laboratory Findings by Clinical Forms. The detailed assessment of laboratory indicators demonstrated significant variability depending on the clinical form of Henoch–Schönlein purpura.Leukocytosis was detected in 35% of the total cohort, but its frequency differed by clinical form: it was most common in the mixed form (41%) and in the cutaneous-articular form (36%), while much less frequent in the isolated cutaneous variant (18%). Similarly, an elevated ESR (>20 mm/h) was noted in 28% of patients overall, with higher prevalence in mixed (34%) and cutaneous-articular (29%) groups compared to only 12% in the isolated cutaneous form. These findings indicate that systemic inflammatory activation is more pronounced in severe variants of the disease.Normal platelet counts were preserved in the majority (73%), but deviations showed clinically meaningful distributions. Thrombocytosis (>450×10⁹/L) was present in 18% overall, with higher rates in mixed (27%) and cutaneous-articular (22%) forms, compared to 5% in the isolated cutaneous form. Thrombocytopenia (<150×10⁹/L) occurred in 9% overall, but was disproportionately concentrated in the renal form (25%), with lower rates in mixed (8%) and cutaneous-articular (7%) variants. This strongly suggests that thrombocytopenia is associated with renal involvement and worse prognosis.Dysproteinemia was detected in 58% of cases, again most common in the renal (71%) and mixed (63%) groups, less so in cutaneous-articular (55%) and minimal in the cutaneous form (22%). Elevated α₂- and γ-globulins were found in 61% overall, peaking in renal (79%) and mixed (66%) patients, indicating pronounced humoral immune activation. Elevated transaminases (ALT/AST) occurred in 48% of children, more frequently in mixed (53%) and renal (61%) forms than in cutaneous (15%). Elevated creatinine was relatively rare (6%), but strongly associated with the renal form (28%), highlighting its prognostic significance.Coagulation disturbances were among the most clinically relevant findings. A shortened APTT was documented in 19% of all patients, with the highest rates in renal (33%) and mixed (22%) forms. Increased prothrombin index (PTI) was observed in 14%, again more common in renal (27%) and mixed (17%) cases. Elevated fibrinogen (>4.5 g/L) was recorded in 15%, more frequent in renal (31%) and mixed (19%) patients. D-dimer elevation was one of the most striking findings, present in 25% overall but highest in renal (36%) and mixed (28%) groups. Soluble fibrin-monomer complexes (SFMC) were increased in 9%, most common in renal (19%) and mixed (11%) cases.
|
4. Discussion
- The analysis of our findings demonstrates that Henoch–Schönlein purpura (HSP) in children manifests with significant clinical and laboratory heterogeneity, which largely depends on the prevailing clinical form of the disease. The predominance of cutaneous-articular and mixed variants in our cohort is consistent with the results of numerous international studies, which emphasize that these forms constitute the majority of pediatric cases. At the same time, the renal form, although less common, is of particular clinical importance due to its potential for long-term complications and progression to chronic kidney disease.The obtained laboratory data revealed a high frequency of dysproteinemia (58%) and an increase in α₂- and γ-globulins (61%), which confirms the involvement of immune-inflammatory mechanisms in the pathogenesis of HSP. Elevated liver transaminases (48%) observed in nearly half of patients indicate systemic involvement beyond the vascular component, which has also been described in earlier studies. Importantly, creatinine elevation (6%) was almost exclusively associated with the renal form, highlighting the need for close nephrological monitoring in this subgroup of patients [7].Changes in the hemostatic system, recorded in 63% of children, represent one of the key pathogenetic aspects of HSP. Shortened activated partial thromboplastin time (APTT), elevated prothrombin index (PTI), increased fibrinogen levels, and higher concentrations of D-dimer and soluble fibrin-monomer complexes indicate a hypercoagulable state, which correlates with the severity of the disease and the presence of nephritic or mixed forms. Similar findings have been emphasized in several European and Asian studies, which underline the contribution of hypercoagulation and endothelial dysfunction to the development of severe complications in HSP [4,6,8,10].Taken together, the results suggest that the spectrum of clinical manifestations in HSP is closely related to immunological and hemostatic disturbances. The identification of laboratory predictors—such as dysproteinemia, hypercoagulability markers, and early biochemical signs of renal impairment—may be crucial for stratifying patients by risk and developing individualized therapeutic strategies.
5. Conclusions
- The study of clinical and laboratory characteristics of Henoch–Schönlein purpura in children has demonstrated that the disease is characterized by pronounced variability depending on the predominant clinical form. The cutaneous-articular and mixed variants were the most frequently observed, whereas the renal form, though less common, carried the greatest risk for long-term complications.Laboratory analyses revealed a high prevalence of dysproteinemia, elevation of α₂- and γ-globulins, and signs of systemic inflammation, all of which confirm the central role of immune-inflammatory processes in the pathogenesis of the disease. Hypercoagulability, observed in more than half of the cases, was strongly associated with the severity of the clinical course and was particularly evident in children with renal and mixed forms of HSP.The obtained data underscore the importance of comprehensive clinical and laboratory assessment in pediatric patients with HSP. Early identification of laboratory abnormalities, particularly immunological and hemostatic disturbances, allows clinicians to stratify patients by risk and optimize management strategies. These findings highlight the need for integrated diagnostic approaches and timely therapeutic interventions aimed at preventing progression to severe complications, especially renal involvement.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML