Ulzhabaev Javlon Abdugani ugli
Andijan State Medical Institute, Uzbekistan
Correspondence to: Ulzhabaev Javlon Abdugani ugli, Andijan State Medical Institute, Uzbekistan.
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Polymorphism G-455A of the FGB gene is associated with changes in fibrinogen concentration in pregnant women, especially in obstetric hemorrhages. Genotype -455AA is associated with a decrease in fibrinogen levels, which can lead to hypocoagulant disorders of the hemostasis system.
Keywords:
Dysfibrinogenemia, FGB, Polymorphic variants -455G/ A, Obstetric bleeding, Coagulation disorders
Cite this paper: Ulzhabaev Javlon Abdugani ugli, FGB Gene Polymorphism (G-455A) and Its Association with Obstetric Bleeding During Cesarean Section, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2795-2798. doi: 10.5923/j.ajmms.20251508.79.
1. Introduction
The problem of maintaining and improving women's reproductive health remains the focus of the medical community and the World Health Organization, which are developing programs with new conceptual approaches to address it [2,4,5,6,7,10,13]. Nevertheless, reproductive disorders and obstetric hemorrhages continue to be a pressing problem for both developed countries and countries with less favorable conditions.Thrombophilia plays a significant role in pregnancy pathologies, the contribution of which, according to some data, is from 40 to 80%. Untimely detection of thrombophilia can lead to various clinical complications [1,3,11,12], including obstetric bleeding in women in labor [8,9] and hidden hemostasis disorders are especially dangerous, which, in the absence of timely diagnosis, can cause an unfavorable pregnancy outcome.Determination of fibrinogen level is a standard test in the study of the blood coagulation system. Unlike genetic polymorphism analysis, this test is more accessible and can serve as an important marker for the detection of clinically significant genetically determined hemostasis disorders. Genetic factors, including FGB polymorphisms (-455G/A), along with physiological and exogenous causes, play a key role in pathological changes in fibrinogen levels. These changes are associated with reproductive disorders in women, accompanied by disorders of the blood coagulation system and fluctuations in plasma fibrinogen concentrations [5,9]. The aim of the study was to establish a link between FGB gene polymorphisms (-455G/A) and obstetric bleeding in women with hemostasis disorders.
2. Material and Methods
The main group consisted of 80 patients with cesarean section, which were divided into two subgroups: Subgroup I (n = 55) with intrapartum emergency abdominal delivery, Subgroup II (n = 25) with planned abdominal delivery. In all women, infectious (including sexually transmitted) diseases and extragenital pathology were not detected at the time of examination. All patients were analyzed for anamnestic, clinical and laboratory data and hemostasis parameters at the time of treatment. The control group consisted of 85 practically healthy pregnant women with natural childbirth. Molecular genetic study of polymorphic variants -455g/A in the FGB gene was carried out according to modified protocols using oligonucleotide primers (Metabion, Germany) by the polymerase chain reaction method. The amplification products of DNA fragments of the FGB-455 G/A gene were subject to hydrolytic cleavage.Statistical calculations were performed using standard Microsoft software packages. Excel 2016. To assess the reliability of quantitative indicators, the arithmetic mean and the error of the arithmetic mean (m ± m) were determined.In our study, we analyzed the distribution frequency of alleles and genotypes of the FGB gene polymorphism (G-455A) in the main group of 80 pregnant women and the control group of 85 pregnant women with normal pregnancy. In the main group, the frequency of the wild homozygous genotype G/G was 93.75%, the heterozygous genotype G/A was 3.75%, and the mutant homozygous genotype A/A was 2.5%. In the control group, these figures were 97.65% for G/G and 2.35% for G/A, respectively (Fig. 1).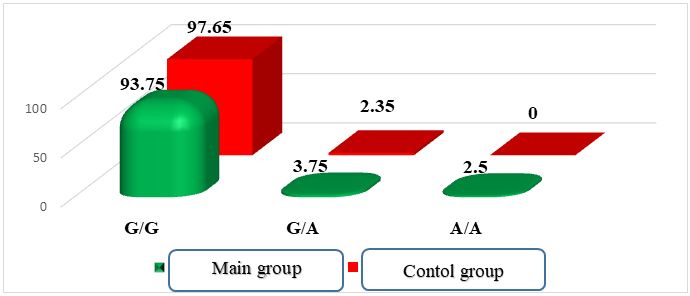 | Figure 1. Distribution of FGB gene genotypes in the main and control groups of patients |
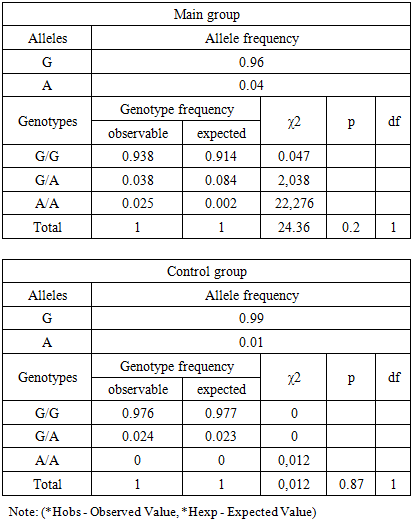
When analyzing the distribution of FGB gene alleles, it was found that the wild-type G allele was predominant in the control group of pregnant women with a frequency of 98.82%, while in the main group its frequency was 95.63%.Mutant allele frequency In the main group it was 4.38%, while in the control group it was 0.0%.The distribution of genotypes of the G-455A polymorphism of the FGB gene in pregnant women of the main and control groups in our study corresponded to the Hardy- Weinberg equilibrium (HWE). The frequencies of the G and A alleles were 0.96 and 0.04 in the main group and 0.99 and 0.01 in the control group, respectively.Table 1. Frequency distribution of genotypes of the G-455A polymorphism of the FGB gene in accordance with the Hardy- Weinberg equilibrium (observed and expected results)
 |
| |
|
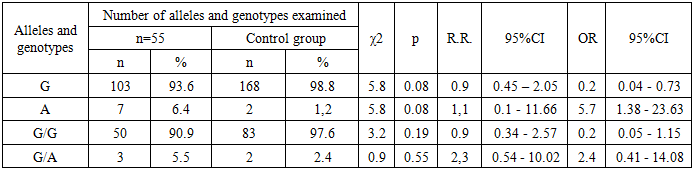
The analysis of the data presented in the table shows that in the main group for the FGB gene polymorphism (-455 G/A) the observed frequency of the homozygous genotype G/G (Hobs) was 0.938%, while the expected one (Hexp) was 0.914% (χ² = 0.047; p = 0.2). For the genotypes G/A and A/A the observed frequencies (Hobs) were 0.038% and 0.025%, while the expected ones (Hexp) were 0.084% and 0.002%, respectively (χ² = 2.038; p = 0.2 for G/A and χ² = 22.276; p = 0.2 for A/A). No statistically significant differences were found (D = -0.55 and D = 0.01, respectively). These results are consistent with the Hardy -Weinberg equilibrium, and the probability of systematic errors in the study remains low (χ² < 3.85; p > 0.05).In the control group, the frequencies of the G/G, G/A, and A/A genotypes for this polymorphism were 0.976/0.977, 0.024/0.023, and 0.0/0.0, respectively (χ² = 0.012; p = 0.87), which also indicates a low probability of systematic errors during the study.In the main group, the observed frequency of heterozygous genotypes G/A of the FGB gene (-455 G/A) exceeded the expected one (0.04 versus 0.08; D = -0.55). A similar trend was noted in the control group, where the observed frequency slightly exceeded the expected one (0.02 versus 0.02; D = 0.01).When comparing the frequencies of the G/G, G/A, and A/A genotypes of the G-455A polymorphism of the FGB gene between the main and control groups, it was found that the wild genotype G/G was more common in pregnant women in the control group, while the heterozygous genotype G/A prevailed in the main group. The mutant homozygous genotype A/A was not detected in any of the groups. The frequencies of the G/G, G/A, and A/A genotypes in the main group were 93.8%, 3.8%, and 0.0%, respectively, while in the control group they were 97.6%, 2.4%, and 0.0%, respectively.In the control group, the frequency of detection of the wild genotype G/G was slightly higher (χ² = 1.5; p = 0.47; RR = 1.0; 95% CI: 0.36–2.54; OR = 0.4; 95% CI: 0.07–1.8). In the main group, the heterozygous genotype G/A prevailed (χ² = 0.3; p = 0.82; RR = 1.6; 95% CI: 0.38–6.71; OR = 1.6; 95% CI: 0.27–9.78).Statistical analysis showed that, according to the odds ratio (OR), the mutant allele A (OR = 3.8; 95% CI: 0.87–16.88) and the heterozygous genotype G/A (OR = 1.6; 95% CI: 0.27–9.78) can be considered as risk factors for complications during cesarean section, but these data are not statistically significant (χ² = 0.3; p = 0.82).We analyzed the differences in the frequency of occurrence of genotypes and alleles of the G-455A polymorphism of the FGB gene between Group I (n = 55) and the control group of patients participating in the study (Table 2).Table 2. Frequency of occurrence of genotypes and alleles of the FGB gene (G455A) in patients of subgroup I (n= 55) (probability-control model)
 |
| |
|
Analysis of the data presented in the table revealed that in the control group the frequency of the G allele was higher than in group I and was 98.8% versus 93.6%, respectively (χ² = 5.8; p = 0.08; RR = 0.9; 95% CI: 0.45–2.05; OR = 0.2; 95% CI: 0.04–0.73). In contrast, the frequency of the allele A was higher in subgroup I compared to the control group (6.4% vs. 1.2%, χ² = 5.8; p = 0.08; RR = 1.1; 95% CI: 0.1–11.66; OR = 5.7; 95% CI: 1.38–23.63). The frequency of the homozygous G/G genotype in the control group exceeded that of subgroup I (97.6% vs. 90.9%, χ² = 3.2; p = 0.19; RR = 0.90; 95% CI: 0.34–2.57; OR = 0.2; 95% CI: 0.05–1.15).In subgroup I, the frequency of the heterozygous genotype G/A was higher than in the control group (5.5% versus 2.4%, χ² = 0.9; p = 0.55; RR = 2.3; 95% CI: 0.54–10.02; OR = 2.4; 95% CI: 0.41–14.08). According to the study data, the indicators of the A allele and the heterozygous genotype G/A, expressed through the odds ratio (OR) and relative risk (RR), may indicate a potential risk of developing intraoperative haemostatic complications, however these results are statistically insignificant (χ² = 0.9; p = 0.55).Efficiency of predicting the presence of a mutant allele And the FGB gene in the main group was AUC = 0.52 (SE = 0.99; SP = 0.04; OR = 3.84; 95% CI = 0.87–16.85; p = 0.22). In pregnant women of group I, this indicator was AUC = 0.53; SE = 0.99; SP = 0.06; OR = 5.71; 95% CI = 1.38–23.64; p = 0.22.When constructing a prognostic model for the FGB gene alleles, it was found that the sensitivity of mutant alleles in the main group reached a high level of 99%, while the specificity was only 10%. The area under the ROC curve (AUC) was 0.52. These AUC values indicate that the FGB gene polymorphism (G-455A) as an independent model for predicting intraoperative The incidence of haemostatic complications does not have statistical significance for all groups.The efficiency of predicting the heterozygous G/A genotype of the FGB gene in the main group was AUC = 0.51 (SE = 0.04; SP = 0.98; OR = 1.62; 95% CI = 0.27–9.87; p = 0.48). For pregnant women in group I, the indicators were as follows: AUC = 0.52; SE = 0.05; SP = 0.98; OR = 2.39; 95% CI = 0.41–14.01; p = 0.39.
3. Conclusions
High fibrinogen levels are associated with increased blood clotting, but some studies indicate that mutations in the FGB gene, including the G-455A polymorphism, may be associated with bleeding conditions such as congenital afibrinogenemia or hypofibrinogenemia, causing fibrinogen deficiency or dysfunction [Karpenko O., Netiazhenko V., 2020]. There is also evidence that the A allele of the G-455A polymorphism of β-fibrinogen is associated with an increased risk of bleeding.Our statistical data did not confirm the significant role of the G-455A polymorphism of the FGB gene in the development of intraoperative hemostatic complications in the Uzbek population. Although the odds ratio (OR) and relative risk (RR) values indicated a possible risk of bleeding in Uzbek women with mutant alleles and heterozygous genotypes, these results did not reach statistical significance when analyzed by the chi-square method (χ² < 3.84, p > 0.05).Thus, bleeding during and after surgical interventions has a multifactorial nature, and its causes remain unclear in many patients. This complicates diagnosis and treatment, and also leads to psychological trauma both for patients suffering from this pathology and for medical specialists.
References
| [1] | Karpenko, O.O., Netyazhenko, V.Z. (2020). Influence of mutations in the integrin beta-3 gene in curical diabetes type 2 on the activity of platelet hemostasis in patients in gostria and chronic coronary syndromes / vol. 73 no. 3 (2020): Problems endocrine pathology / Clinical endocrinology. |
| [2] | Mukhamedova B, et al. Vestnik Extreme Medicine 2016; 4. |
| [3] | Jastrzebska M, et al. J Physiol Pharmacol 2019; 70(2): 175-185. https://doi.org/10.26402/jpp.2019.2.01. |
| [4] | Kafian S, et al. Scandinavian J Clin Lab Invest 2019; 79(7): 507-512. https://doi.org/10.1080/00365513.2019.1663554. |
| [5] | Grinstein Y, et al. Genetic testing and molecular biomarkers 2018; 2 2(4): 2 59-265. https://doi.org/10.1089/gtmb.2017.0177. |
| [6] | Tsantes A, et al. Thrombosis Res 2019; 180: 47-54. https://doi.org/10.1016/j.thromres.2019.06.001. |
| [7] | Zotova TYu, et al. Bull Experiment Biol Med 2016; 161(3): 334-338. https://doi.org/10.1007/s10517-016-3408-0. |
| [8] | Mjandina GI, et al. Pul's 2014; 16(4). |
| [9] | Christopher FN, Benjamin EH, Ferro A. PLoS One 2014; 9(7). https://doi.org/10.1371/journal.pone.0100239. |
| [10] | Christopher FN, Ferro A. Brit J Clin Pharmacol 2014; 77(3): 446-457. https://doi.org/10.1111/bcp.12204. |
| [11] | Salem Abdel Halim A, et al. Blood Transfusion 2014; 12(1): s281. https://doi.org/10.2450/2013.0220-12. |
| [12] | Muslim JeF, et al. Terapevt Arh 2017; 89(5): 74-78. |
| [13] | Proscia C, et al. Int Cardiol 2015; 7(3): 283. |



 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML
