-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2769-2771
doi:10.5923/j.ajmms.20251508.74
Received: Aug. 5, 2025; Accepted: Aug. 26, 2025; Published: Aug. 30, 2025

Comparative Characterization of Liver Fibrosis and Its Collagen Markers in Patients with Tuberculosis and Chronic Viral Hepatitis
Yitmasova Tuhfa Davlatovna1, Obloqulov Abdurashid Raximovich2, Usmonov Isomiddin Haydarovich3
1PhD Candidate, Department of Phthisiology and Pulmonology, Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan
2Doctor of Medical Sciences, Professor, Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan
3Associate Professor, Republican Specialized Scientific and Practical Medical Center of Phthisiology and Pulmonology, Tashkent, Uzbekistan
Correspondence to: Yitmasova Tuhfa Davlatovna, PhD Candidate, Department of Phthisiology and Pulmonology, Bukhara State Medical Institute named after Abu Ali ibn Sino, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Coinfection with tuberculosis (TB) and chronic viral hepatitis (CVH) is presumed to accelerate the progression of liver fibrosis. The aim was to compare the baseline state of liver fibrosis in three groups: TB and CVH coinfection (Group 1, n=59), TB monoinfection (Group 2, n=33), and CVH monoinfection (Group 3, n=30). Fibrosis was assessed using FibroTest and by measuring serum collagen types I and IV. Results showed a clear gradient of fibrosis severity. Advanced fibrosis/cirrhosis (F3-F4) was present in 42.4% of the coinfection group, significantly higher than in the CVH monoinfection group (13.3%) and the TB monoinfection group (3%) (p<0.01). Collagen I and IV levels were also highest in Group 1. Conclusion: TB infection is an independent factor that exacerbates liver fibrogenesis in the context of CVH. A thorough pre-treatment assessment of fibrosis in coinfected patients is of critical clinical importance.
Keywords: Tuberculosis, Chronic viral hepatitis, Coinfection, Liver fibrosis, FibroTest, Collagen type I, Collagen type IV, Comparative analysis
Cite this paper: Yitmasova Tuhfa Davlatovna, Obloqulov Abdurashid Raximovich, Usmonov Isomiddin Haydarovich, Comparative Characterization of Liver Fibrosis and Its Collagen Markers in Patients with Tuberculosis and Chronic Viral Hepatitis, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2769-2771. doi: 10.5923/j.ajmms.20251508.74.
1. Introduction
- Tuberculosis (TB) and chronic viral hepatitis (CVH) are two global epidemics that continue to place an immense burden on healthcare systems worldwide [1,2]. The convergence of these two infections in a single patient, i.e., coinfection, creates distinct clinical challenges due to their interaction [3,4]. Liver fibrosis is the universal outcome of chronic liver diseases, including CVH, and its progression ultimately leads to cirrhosis and its life-threatening complications [6]. In CVH, the main driver of fibrogenesis is the persistent inflammation and immune response caused by the virus [7]. However, the influence of tuberculosis infection on this process is of particular interest. TB, although primarily a pulmonary disease, is renowned for its systemic inflammatory response, characterized by high levels of pro-inflammatory cytokines such as TNF-α [8]. These cytokines can activate hepatic stellate cells, thereby enhancing fibrogenesis. This posits a "double-hit" hypothesis, whereby the simultaneous impact of both infections drastically accelerates liver damage.Assessing the degree of fibrosis is crucial for determining treatment strategies. While liver biopsy remains the "gold standard," it is increasingly challenged by its invasiveness and limitations [9], leading to the growing popularity of non-invasive methods [5]. These include algorithms like FibroTest [10] and direct markers of fibrogenesis, such as collagen types I and IV [7]. Many studies have focused on the hepatotoxicity of anti-tuberculosis drugs [11,12]. However, there is a lack of comparative studies on the baseline state of the liver in patients with different infectious statuses before treatment initiation. Such an analysis would allow for the determination of the extent to which TB itself affects fibrosis and how significantly it is potentiated by concomitant CVH.The aim of the study: To perform a comparative assessment of the state of liver fibrosis using non-invasive markers in three groups of patients before the start of anti-tuberculosis treatment: those with TB and CVH coinfection, those with TB only, and those with CVH only.
2. Materials and Methods
- Study Design and Patient Population:This single-center, prospective, comparative study enrolled a total of 122 patients, who were divided into three groups:1. Group 1 (Coinfection): 59 patients diagnosed with drug-susceptible pulmonary tuberculosis and chronic viral hepatitis (B and/or C).2. Group 2 (TB monoinfection): 33 patients diagnosed with drug-susceptible pulmonary tuberculosis only, without CVH.3. Group 3 (CVH monoinfection): 30 patients diagnosed with chronic viral hepatitis only, without tuberculosis (control).Inclusion and Exclusion Criteria:• Inclusion criteria: Age from 18 to 65 years; confirmed diagnosis for the respective group (TB via GeneXpert MTB/RIF, CVH via HBsAg/Anti-HCV positive for >6 months); written informed consent.• Exclusion criteria: HIV infection, alcohol abuse, other chronic liver diseases, decompensated comorbidities, pregnancy, prior anti-tuberculosis or antiviral treatment.Methods of Examination:All examinations for patients in Groups 1 and 2 were conducted before the initiation of anti-tuberculosis treatment.• "FibroTest": The fibrosis stage (F0-F4) was determined using the BioPredictive (France) algorithm according to the METAVIR scale.• Collagen Measurement: Serum levels of type I and type IV collagen were measured using the enzyme-linked immunosorbent assay (ELISA) method with standard test kits (results in ng/mL).• General Examinations: Clinical and biochemical blood analyses.Statistical Analysis:Data were analyzed using SPSS software. For inter-group comparisons, ANOVA or the Kruskal-Wallis test (for quantitative data) and the chi-square (χ²) test (for qualitative data) were used. A p-value of <0.05 was considered statistically significant.
3. Results
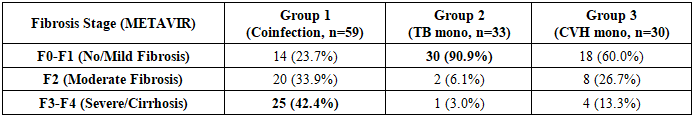
- All three groups were comparable in terms of age and gender (p>0.05).Assessment of liver fibrosis via FibroTest revealed stark differences between the groups (Table 1).
|
|
4. Discussion
- The results of this study clearly demonstrate the interaction of infectious agents in the development of liver fibrosis. The data show a distinct gradient of fibrosis severity: (TB+CVH) > (CVH mono) > (TB mono). This indicates that coinfection has a synergistic, mutually aggravating negative effect.The minimal level of fibrosis in patients with only tuberculosis suggests that TB infection itself, despite causing systemic inflammation, is not sufficient to induce significant liver fibrosis. However, it acts as a powerful trigger in the context of CVH. When the potent cytokine storm (TNF-α, etc.) characteristic of TB is superimposed on the chronic inflammation caused by CVH, the fibrogenesis processes in the liver are dramatically activated. This is confirmed by the highest indicators for both FibroTest and the direct fibrosis markers, collagens, in the coinfection group.A crucial aspect of our study is that we assessed the pre-treatment condition. This means that coinfected patients already have advanced liver fibrosis before they even begin taking highly hepatotoxic anti-tuberculosis drugs. This places them in an extremely high-risk group for developing severe forms of drug-induced liver injury.Therefore, our results provide clear recommendations for clinical practice: it is essential to screen every patient diagnosed with TB for CVH markers. If coinfection is detected, a thorough assessment of the liver fibrosis stage is mandatory before starting treatment. In such cases, standard treatment regimens should be used with caution, under constant hepatoprotective cover, and with meticulous monitoring of liver function.
5. Conclusions
- 1. Coinfection with tuberculosis and chronic viral hepatitis is characterized by a significantly more severe degree of liver fibrosis compared to either CVH or TB monoinfections.2. TB infection itself does not lead to significant liver fibrosis but acts as an independent risk factor that dramatically accelerates the process of fibrogenesis in the context of CVH.3. A mandatory assessment of the liver fibrosis stage using non-invasive markers (FibroTest, collagen I and IV) is necessary in coinfected patients before starting anti-tuberculosis treatment, to manage them as a high-risk group and to individualize their treatment strategy.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML