-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2749-2752
doi:10.5923/j.ajmms.20251508.69
Received: Aug. 2, 2025; Accepted: Aug. 23, 2025; Published: Aug. 30, 2025

The Dynamics of Aphasia (Paraphasia and Perseveration) During the Stroke Recovery Period
Munisa Rasulova
Lecturer , Department of Neurology and Medical Psychology, Tashkent State Medical University, Tashkent, Uzbeksitan
Correspondence to: Munisa Rasulova, Lecturer , Department of Neurology and Medical Psychology, Tashkent State Medical University, Tashkent, Uzbeksitan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

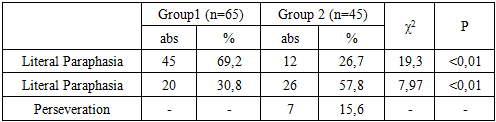
This study investigates the clinical presentation and temporal progression of aphasia—specifically, literal paraphasia, verbal paraphasia, and perseveration—during the acute and recovery phases of ischemic stroke. Aphasia remains one of the most disabling consequences of stroke, affecting nearly one-third of patients and significantly hindering their ability to communicate, thereby disrupting personal, social, and emotional well-being. The research involved 110 aphasic patients treated between 2021 and 2024 at the multidisciplinary clinic of the Tashkent Medical Academy, divided into two groups: 65 in the acute stage and 45 in the recovery stage. Analysis revealed that literal (phonemic) paraphasia was significantly more common in the acute phase (69.2%) than in the recovery phase (26.7%), indicating that early post-stroke speech impairments are largely phonological in nature. In contrast, verbal (semantic) paraphasia increased during the recovery stage (57.8% versus 30.8%), suggesting a shift toward higher-level language processing deficits as neural reorganization occurs. Perseveration, characterized by the involuntary repetition of words or syllables, was not observed during the acute stage but emerged in 15.6% of patients in the recovery group, indicating deeper cortical involvement and the possible effects of insufficient rehabilitation. These findings demonstrate the dynamic nature of post-stroke aphasia and underscore the need for phase-specific and individualized rehabilitation strategies. Emphasis should be placed on correcting phonemic errors during the acute phase and addressing semantic deficits and stereotyped speech patterns during recovery. Without timely and appropriate intervention, these impairments may worsen and lead to persistent communication dysfunction, adversely affecting reintegration and quality of life. The statistically significant differences observed (χ² = 19.3 and χ² = 7.97; P < 0.01) further validate the clinical relevance of these symptom patterns and support their use in evaluating rehabilitation outcomes.
Keywords: Cerebrovascular disease, Stroke, Paraphasia, Perseveration, Recovery period
Cite this paper: Munisa Rasulova, The Dynamics of Aphasia (Paraphasia and Perseveration) During the Stroke Recovery Period, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2749-2752. doi: 10.5923/j.ajmms.20251508.69.
Article Outline
1. Introduction
- Each year, approximately 12.2 million individuals worldwide are affected by stroke [1] As many as 40% of individuals who survive a stroke initially develop aphasia [2], a condition that impairs both language comprehension and production. Since language is fundamental to communication in familial, social, and professional settings, aphasia often leads to substantial psychosocial challenges [3] and a diminished quality of life related to health [4]. Speech and language therapy has been shown to be an effective intervention for individuals with aphasia [5,6,7], and meta-analyses of extensive patient-level data indicate that those who receive therapy that is intensive, frequent, and personalized tend to experience more favorable outcomes [8]. In real-world clinical settings, individuals with aphasia often receive only minimal therapy [9,10,11], and the limited availability of clinical resources may be used suboptimally due to insufficient knowledge about which specific treatments are most effective for different patient profiles [12,13]. Providing effective and personalized aphasia treatment that maximizes the use of available resources requires a comprehensive understanding of the individual characteristics of people with aphasia—such as their sociodemographic background, type and severity of stroke, and specific features of their language impairment—as well as insight into the quality and type of care they receive and the outcomes they value and attain. At present, there is a lack of routinely collected data to inform this understanding [14,15], and only limited benchmarks exist to measure therapy success from the patient’s perspective, making it difficult to determine when meaningful progress has been achieved [16]. Speech is a fundamental means of human communication, a marker of etiquette and moral values, and an indicator of intellectual development. It is one of the most essential functions that distinguishes humans. Normal speech production depends on the coordinated function of various parts of the brain. Speech impairments are observed in approximately one-third of stroke patients. Deficiencies in speech significantly impact the patient’s personal, family, and social life, leading to social and psychological maladaptation.
2. The Aim of the Study
- To assess the clinical manifestation and dynamics of aphasia (literal and verbal paraphasia, and perseveration) during the stroke recovery period.
3. Materials and Methods
- Between 2021 and 2024, we studied 110 patients with post-stroke aphasia who were treated under inpatient conditions at the intensive neurology and neurology departments of the multidisciplinary clinic of the Tashkent Medical Academy. For the purpose of investigating speech disorders, the patients were divided into two groups: Group 1: 65 patients with aphasia in the acute stage of stroke; Group 2: 45 patients with aphasia in the recovery stage of stroke. During the stroke recovery period, the most common speech symptoms observed were paraphasia and perseveration. Paraphasias are characterized by the incorrect or inappropriate use of sounds or words, often involving their substitution with irrelevant or mismatched speech elements.When individual, unstable sounds are incorrectly replaced (e.g., saying “TALEB” or “TALBE” instead of “TABLE”), this is referred to as literal (phonemic) paraphasia. Patients with literal paraphasia may misuse or omit certain phonemes or add new ones. These substitutions result from disruptions in the sensory or motor control of speech.When an entire word is replaced with another word that is phonologically or semantically similar (e.g., using “MAP” instead of “CAP” or “HAT” instead of “KAT”), this is known as verbal (semantic) paraphasia. In such cases, patients tend to substitute target words with alternatives that are meaningfully close, relying only on partial cues. For example, instead of saying “water,” a patient might say “to drink” or “that thing.” These verbal paraphasias are considered to result from semantic and mnestic disturbances associated with widespread damage to the frontal, temporal, and parietal lobes. The presence of paraphasias in stroke patients indicates impaired comparative processing of individual phonemes and words. The nature of paraphasias can vary across aphasia types, often providing important diagnostic clues.Perseveration refers to the involuntary, persistent repetition of a word, syllable, or even motor activity. It is typically due to the inertia of neural processes, where the signal to terminate a speech act is delayed. Clinically, perseveration may resemble clonic stuttering, manifesting as frequent, involuntary repetitions of a sound or syllable (e.g., “mamamamachine”).
4. Results and Discussion
- During the stroke recovery phase, 65 patients demonstrated paraphasic speech patterns. Paraphasia, in this context, was defined as the inaccurate or inappropriate use of speech sounds or words in response to a given speech task, often involving substitution with irrelevant elements.The misuse, omission, or addition of individual phonemes was classified as literal paraphasia, stemming from disruptions in sensory or motor speech control mechanisms.As patients progressed in recovery, their speech showed a tendency to substitute entire words with semantically related alternatives — a phenomenon categorized as verbal paraphasia.
|
5. Conclusions
- In summary, the dynamics of aphasia during stroke recovery reveal key differences in the manifestation of speech disorders between the acute and recovery phases:1. Literal paraphasia (phoneme-level errors) was significantly more common during the acute phase (69.2%) and markedly decreased during the recovery phase (26.7%), indicating phonemic improvement over time.2. Verbal paraphasia (semantic substitutions) was less frequent in the acute phase (30.8%) but increased during recovery (57.8%), reflecting an expansion of the speech lexicon despite ongoing semantic inaccuracies.3. Perseveration (involuntary repetition of a word or sound) was observed only during the recovery phase (15.6%), indicating delayed onset of deeper cortical damage when rehabilitation was inadequate.These findings emphasize the need for individualized and staged rehabilitation approaches in post-stroke aphasia treatment. Attention should be paid to phonemic errors during the acute phase and to semantic and stereotyped errors during the recovery period. If appropriate rehabilitation measures are not implemented in time, speech disorders may worsen, progressing to severe forms such as perseveration, which significantly impair social integration and quality of life.The results of statistical analysis (χ² = 19.3 and χ² = 7.97; P < 0.01) confirm the reliability of these findings and demonstrate their importance as key indicators in evaluating the effectiveness of various rehabilitation strategies.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML