-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2745-2748
doi:10.5923/j.ajmms.20251508.68
Received: Aug. 2, 2025; Accepted: Aug. 21, 2025; Published: Aug. 30, 2025

Clinical and Immunological Changes in Young Children with Community-Acquired Pneumonia Complicated by Infectious-Toxic Shock
Aliev A. L.1, Quziyev D. V.2, Yuldoshev S. V.3
1Tashkent Medical University, Uzbekistan
2Assistant of the Department of Propaedeutics of Children's Diseases and Outpatient Pediatrics, Andijan State Medical Institute, Uzbekistan
3Student of Andijan State Medical Institute the Faculty of Pediatrics, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

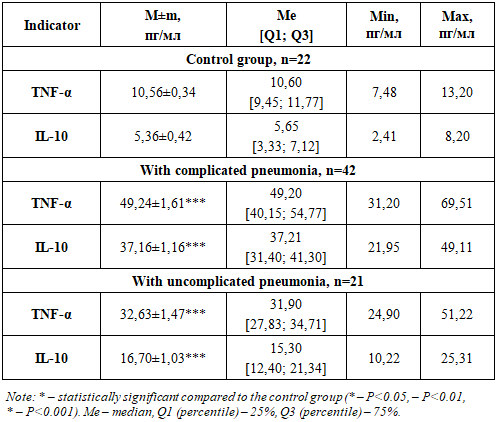
The study included 63 children with a confirmed diagnosis of pneumonia (main group – 42 children under one year of age with complicated pneumonia; comparison group – 21 children under one year of age with uncomplicated pneumonia). Twenty-two practically healthy children constituted the control group. The concentrations of IL-10 and TNF-α in blood serum were determined by solid-phase enzyme-linked immunosorbent assay (ELISA) using test systems manufactured by AO “Vector-Best” (Novosibirsk, Russia). Hypersecretion of the studied cytokines was established in patients of both groups. TNF-α contributes to the body’s fight against infection, which was observed in the group of children with uncomplicated pneumonia. Excessive production of TNF-α in children with complicated pneumonia may lead to negative effects, such as increased vascular permeability, which can cause pulmonary edema and hemorrhages. In addition, an elevated level of IL-10 in both complicated and uncomplicated pneumonia in the examined children is likely part of a complex immune response aimed at regulating inflammation and limiting tissue damage.
Keywords: Immunity, Pneumonia, Children under one year of age, Cytokines, Serum, Imbalance
Cite this paper: Aliev A. L., Quziyev D. V., Yuldoshev S. V., Clinical and Immunological Changes in Young Children with Community-Acquired Pneumonia Complicated by Infectious-Toxic Shock, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2745-2748. doi: 10.5923/j.ajmms.20251508.68.
1. Introduction
- Pneumonia (PN) is a severe infectious and inflammatory disease, accounting for 80% of pulmonary pathology, with high mortality among children, especially preterm and newborn infants. In recent years, cases of pneumonia with extremely severe and “fulminant” progression have increasingly been registered in newborns, and despite the use of modern antibacterial therapy, a high mortality rate persists (6.1–11). Therefore, the study of the etiological structure, clinical features, course, and treatment of pneumonia in children under current conditions is of great theoretical and practical significance.A key role in regulating the immune response of the body to the invasion of pathogenic flora belongs to cytokines, which stimulate a local protective inflammatory reaction with the production of inflammatory mediators. In the case of their hyperproduction, a cascade systemic inflammatory response develops, up to infectious-toxic shock, sepsis, enhanced procoagulant activity, and multiple organ dysfunction [4,9,19].The course of pneumonia depends on the concentration and ratio of pro- and anti-inflammatory cytokines. Their elevated levels may correspond to a severe course of pneumonia [1,2,7].Despite numerous studies and research, the immune response, including the cytokine profile in children under one year of age with complicated pneumonia, remains insufficiently explored, and this process has yet to be fully clarified. At the same time, the nature of immune disturbances at different stages of the inflammatory process has not been studied comprehensively and is interpreted ambiguously. Therefore, the study of cytokines is of particular interest, since they regulate the intensity and duration of the immune response, as well as the nature of the inflammatory process, ensuring both positive and negative immunoregulation.Based on the above, the aim of this study was to clarify and investigate certain key pro- and anti-inflammatory mediators of the immune response in children under one year of age with different courses of pneumonia.
2. Materials and Methods
- This study included 63 children with a confirmed diagnosis of pneumonia, among whom 42 infants under one year of age with complicated pneumonia, admitted to the intensive care unit, comprised the main group, while 21 infants under one year of age with uncomplicated pneumonia comprised the comparison group. Twenty-two practically healthy children formed the control group. The study involved children aged 3 to 12 months.Immunological examinations of the children were carried out at a multidisciplinary pediatric medical institution in the Andijan region.The concentration of the pro-inflammatory tumor necrosis factor alpha (TNF-α) and the anti-inflammatory interleukin-10 (IL-10) in peripheral blood serum was determined by solid-phase enzyme-linked immunosorbent assay (ELISA) using test systems produced by AO “VECTOR-BEST” (Novosibirsk, Russia). Quantitative evaluation of the results was performed by constructing a calibration curve that reflects the dependence of optical density on concentration for the standard antigen, allowing comparison with the studied samples.Statistical processing of the obtained data was carried out using the computer program Statistica 6.0. The data were analyzed using standard approaches, and the results are presented as the sample mean (M) and standard error of the mean (m); the median (Me), characterizing the central tendency; and the upper and lower quartiles, characterizing the spread of values in 50% of the respondents (Q1–Q3), where Q1 is the 25th percentile, Me is the 50th percentile, and Q3 is the 75th percentile. The significance of differences in mean values (P) between the compared indicators was assessed using Student’s t-test.
3. Results and Discussion
- One of the leading causes of complicated and prolonged pneumonia at present is the alteration of the body’s immunological reactivity [5]. The immune response in complicated pneumonia in children under one year of age represents a complex research problem.As is known, the organization of the immune system undergoes age-related changes. Critical periods of development of immunobiological reactivity (milestones) represent stages during which the impact of antigens may trigger a disproportionate or even paradoxical immune response. This response may either be insufficient for effective protection (hypo- or anergic) or excessive (hyperergic). The selected group of children (from 3 to 12 months) in the present study corresponds to the second critical period of immune system development.Literature data indicate a multitude of factors that make the study of immune response in complicated pneumonia in children under one year particularly difficult. Firstly, in this age group, immunological mechanisms are still in the process of formation, and their function may differ significantly from the immune response in adults or older children. Secondly, pneumonia may be caused by various pathogens, such as bacteria, viruses, or fungi, which further complicates the study of immune response.In pulmonary diseases, cytokines are involved in the infectious-inflammatory process at the level of both immune mechanisms themselves and the effector component, largely determining the direction, severity, and outcome of the pathological process [8].In the context of pneumonia, cytokines perform several key functions—they act as mediators of inflammation, regulators of the immune response, agents of recruitment and activation of immune cells, growth and regeneration factors, and immune regulators.It should be noted that initially all children participating in the immunological studies were divided into groups of 3 to 6 months and 6 to 12 months. However, in the course of the research, the results obtained did not show significant differences; therefore, all children in the main and comparison groups were subsequently combined accordingly into the above-mentioned groupings of 3 to 12 months.The obtained results are presented in Table 1 below.
|
4. Conclusions
- Understanding the immune response in complicated pneumonia in children under one year of age is of great importance for developing effective methods of treatment and prevention. This requires further research, including clinical studies, analysis of immunological mechanisms and molecular pathways, as well as the use of modern biotechnological approaches.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML