-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2025; 15(8): 2543-2547
doi:10.5923/j.ajmms.20251508.26
Received: Jul. 13, 2025; Accepted: Jul. 29, 2025; Published: Aug. 4, 2025

Malignant Pleural Effusion in Cancer Patients: Therapeutic Approaches and Outcomes
Jumayev Mamaziyo Yusupovich1, Khalilova Khadicha Avliyakulovna2, Khaitova Gulchehra Avliyoqulovna2, Shaymardonov Sunnatillo Kamolitdinovich3, Jumayeva Nilufar Mamaziyoyevna4
1Associate Professor, Doctor of Medical Sciences, Termez University of Economics and Service, Faculty of Medicine, Termez, Uzbekistan
2Assistant Lecturer, Termez University of Economics and Service, Faculty of Medicine, Termez, Uzbekistan
3Termez University of Economics and Service, Faculty of Medicine, Termez, Uzbekistan
4Research Fellow, Termez University of Economics and Service, Faculty of Medicine, Termez, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Malignant pleural effusion (MPE) is a life-threatening condition that is commonly challenging in patients at the advanced stage of cancers and that could be characterised by significant yet frustrating effects on respiratory conditions and impairment of quality of life. Its finding normally indicates the advancement of malignancy, and it indicates poor prognosis, and there is a need to establish quick and effective treatment. The aim of the study is to obtain the data concerning the clinical outcomes of various approaches to the treatment of MPE using the data available at the Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology (RIO) in Tashkent since February 25, 2022 and at its Surkhandarya branch (RIATM-Surxondaryo) in Uzbekistan. Multidisciplinary treatment was carried out in a group of 404 patients with confirmed malignant neoplasms and pleural effusion. The patients were subjected to pleuroperitoneal shunting, intrapleural chemotherapy, and drainage through thoracostomy depending on their clinical conditions. The planning of treatment was individualized and multidisciplinary among oncologists, surgeons, radiologists, and anesthesiologists to achieve the best result. Remarkably, pleuroperitoneal shunting exerted constant drainage of the pleural space, which aids in the deterrence of recurrence. After fluid aspiration, intrapleural chemotherapy was carried out with cisplatin or fluorouracil (in combination with corticosteroids) to reduce exudate formation. The efficacy of the treatment was measured two weeks after intervention, referring to WHO criteria in terms of fluid dynamics and clinical response. Most of the patients had notable improvements, especially those who were treated with surgery. Adherence and well-being were also contributed to by the use of supportive efforts like vitamin therapy and symptom control. These results demonstrate the utility of a multifactorial and personified intervention strategy when dealing with MPE and especially when surgical and pharmacological interventions are intelligently combined.
Keywords: Malignant pleural effusion, Pleuroperitoneal shunting, Intrapleural chemotherapy, Thoracostomy drainage, Multidisciplinary cancer care
Cite this paper: Jumayev Mamaziyo Yusupovich, Khalilova Khadicha Avliyakulovna, Khaitova Gulchehra Avliyoqulovna, Shaymardonov Sunnatillo Kamolitdinovich, Jumayeva Nilufar Mamaziyoyevna, Malignant Pleural Effusion in Cancer Patients: Therapeutic Approaches and Outcomes, American Journal of Medicine and Medical Sciences, Vol. 15 No. 8, 2025, pp. 2543-2547. doi: 10.5923/j.ajmms.20251508.26.
1. Introduction
- Malignant pleural effusion (MPE) is one of the most frequent and troublesome complications of people with advanced cancers of the lungs, as well as breasts and gastrointestinal tract. Its clinical importance is associated not only with its paradigmatic presence in terminal-stage oncology among patients but also with high levels of deterioration in the respiratory state and well-being of patients that it causes. In a number of cases, the formation of pleural effusion cannot be regarded only as a disease progression mark, but sooner or later it leads to the characteristic short lifespan, which is characterised by three to twelve months, depending on the type of primary tumour and the so-called metastatic extent [1]. Pathophysiologically, MPE follows the invasion of the airspace by cancerous cells, which interfere with the proper transportation of fluids in the parietal and visceral pleurae. The cause of this infiltration may emerge through direct spread of the tumor, blocking due to lymphatic, or hematogenous.MPE presents a set of symptoms that may be severe and rapidly progressive, including dyspnea, chest pain and fatigue. They not only reduce the quality of life of a patient, but they also make the continuous treatment of his/her systemic cancer more complicated. This has made management of MPE timely and effective, a very important point of palliative oncology care. The common method of dealing with this includes repeated thoracentesis or chest drainage, which provides reprieve but is succeeded by the rapid repaccumulation of fluid [2]. Other newer methods, such as chemical pleurodesis and indwelling pleural catheters, have likewise proved effective in limited cases, although people in resource-poor areas may not readily access these treatment options [4].Surgical procedures, pioneered in recent years, especially pleuroperitoneal shunting, have been proposed as an alternative to peritoneal tenosynovitis, and these were reported to be successful in the treatment of recurrent MPE patients. The given intervention constructs the new drainage pathway, which may limit the number of visits to hospitals and invasive puncture usage. In parallel with these, intrapleural chemotherapy, in which drugs are placed directly into the pleural cavity, has proven to be effective when controlling the reaccumulation of the effusions, particularly when the chemotherapy agents are delivered together with a corticosteroid or an enzymatic adjuvant. However, no clear-cut opinion is formed on the preferred protocol of the treatment of MPE, especially when different healthcare settings may be present, as in the case of Uzbekistan. This research aims to overcome these dilemmas based on the assessment of the therapeutic results among the patients with MPE treated in the Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology (RIO) and its Surkhandarya branch. As of 25 February 2022, a multidisciplinary approach to MPE management was adopted in the form of pleuroperitoneal shunting and intrapleural chemotherapy, as well as drainage protocols involving thoracostomies. The study, therefore, is a practical guide on how to manage MPE in late-stage cancer patients in real-life clinical practice by evaluating clinical response, rate of recurrence and symptomatic improvement.
2. Methods
- The current prospective clinical trial was launched on February 25, 2022 both in the Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology (RIO) in Tashkent and its Surkhandarya regional branch (RIATM-Surxondaryo) the institutions of which are tertiary-level cancer centers with modern diagnostic and surgical equipment, which makes them ready to manage the complications of oncology, including malignant pleural effusion (MPE) by multidisciplinary approach. The research question was whether different treatment modalities of MPE were effective and safe among patients with histologically proven malignancies.During February 25, 2022 and December 31, 2022, 404 patients with symptomatic pleural effusion and historical cancer diagnoses were enrolled. The inclusion criteria consisted of patients aged 18 years and older with established malignant pleural effusion with a Karnofsky performance status of at least 50%. Exclusion patients were younger than 18 years old, pregnant women, those having non-malignant effusion, massive lung atelectasis, uncontrolled heart failure, decompensated diabetes mellitus or severe hepatic or renal dysfunction.The patients have been admitted to undergo a thorough clinical assessment, chest radiography, and ultrasound scanning to identify the volume and the distribution of the pleural fluid. Thoracentesis was done to determine the malignancy of the effusion. After diagnosis, the treatment plans were based on the clinical presentation, recurrence of effusion, overall health, and preference at the same time. A multidisciplinary board, i.e., of oncologists, thoracic surgeons, radiologists, and anesthesiologists, or other specialists, such as endocrinologists or haematologists, made the treatment decisions.The patients were assigned to three primary treatment arms i.e. the use of pleuroperitoneal shunts, intrapleural chemotherapy and thoracostomy drainage of the pleural. All approaches had a standardised protocol depending on the institutional resources and patients' needs.In the area of surgical practice, pleuroperitoneal shunting was the intentional formation of a controlled drainage between the peritoneal cavity and the pleural cavity. Patients were put in a lateral decubitus position, and a 3-6 cm incision of the skin was performed at the sixth intercostal level. Once the fluid was removed by using a vacuum aspirator after fluid drainage into the pleural cavity, a series of holes, normally three to six, were made in the diaphragm using electrocautery, hence allowing a constant movement of fluid into the abdominal cavity. The pleural cavity was then made to be hermetically sutured. The procedure was only reserved for a specific group of patients who had recurrent effusions, good performance status, and the lack of peritoneal carcinomatosis. Due to its symptomatic effects and ability to prevent re-hospitalisation, the use of pleuroperitoneal shunting as a palliative procedure has been most prominent in recent years.The second intervention under the umbrella intrapleural chemotherapy depended on ultrasound-guided thoracentesis. Under local analgesia in 1000M L or 1500M L pleural fluid quantity, a blunt catheter was used to remove, followed by gaillard cytotoxic agents (cis platin100 mg/m 2 or fluorouracil 1000 mg) arrangement with hydrocortisone (125M mL-250 M L). Lydase (64 IU) was used in some of the channels as an eliminator of adhesion formation. The monitoring of adverse effects was done on the patient within 24-48 hours. The medicinal basis was to slow down the reaccumulation of effusion through the creation of pleural inflammation and cytolysis of the malignant cells.The third group has been subjected to conventional pleural drainage, carried out using thoracostomy, performed by trocar insertion or by open surgical method, depending on whether there were adhesions. Patient regeneration in the trocar technique was seated, and a trocar containing a blunt stylet was inserted into the pleural cavity, and a closed drain (Bulau) was utilised to avoid ventilation of air inside. In expected major adhesions, an open technique was used and allowed disruptions of fibrous bands by hand. There would be rapid symptomatic improvement, and the approach was particularly beneficial to patients with large effusions or patients with acute respiratory distress.The response to treatment was assessed after an intervention period of two weeks based on WHO guidelines, in which reduction of fluid volume was imaged radiologically and clinical status was assessed. The treatment was graded as complete (they do not have effusion), partial (they had improvement >=50 percent), minimal (< 50 percent), or nil response. The number of re-accumulated, reoccurrence of symptoms and tolerability were noted. To deal with cancer-associated cachexia and fatigue, there was uniform supportive care, which consisted of vitamin B complex and nutritional management, across the groups. Hemotransfusion was avoided in cases where it was possible, as tumor cell division could be exposed. This holistic approach of methodology was meant to capture real-life clinical practice in dealing with MPE and gives some understanding of the practical value of surgical as well as non-surgical intervention in late-stage oncology practice.
3. Results
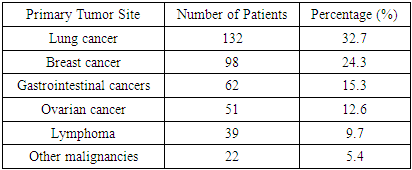
- A total of 404 patients with radiologically and cytologically confirmed MPE recruited between February 25, 2022, and March 2024 in the Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology (RIO) and its Surkhandarya branch are set. The median age of the patients was 62 years (age range: 37-79), and 56% represented females who were enrolled. The characteristic symptoms that all participants had were dyspnea, non-productive cough and pleuritic chest pain. The initial imaging showed moderate to massive pleural fluid, hence requiring the intervention procedure later. The sites of primary location were recorded and classified (Table 1). The most common aetiology was lung cancer (it affected 132 patients, 32.7%), and breast cancer (98 patients, 24.3%). The third most widespread one was gastrointestinal tumors, 15.3% of the cohort had such, and ovarian malignancies were reported in 12.6%. The rest 9.7% and 5.4% were lymphomas and miscellaneous cancers, respectively. These results are almost similar to international epidemiological data, where the number of thoracic and gynecologic cancer mostly prevail over the incidence of MPE.
|
|
4. Discussion
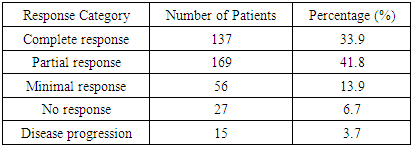
- The study failed to find any conviction to it that any of the modalities was superior in its major end point, which is the control of malignant pleural effusion (MPE), but it was a reality that the combination of pleuroperitoneal shunting and intrapleural chemotherapy was better than conventional drainage in controlling this complication. Patients randomised to shunting showed improved sustained complete response rates as well as decreased occurrence of recurrence within 14 days in comparison to patients subjected to drainage only. The findings further corroborate previous evidence that continuous drainage alleviates patient burden, in addition to the overall utilisation of health care significantly [11]. The excellent results achieved by this group highlight pleuroperitoneal shunting as a reliable and patient-based procedure, especially for those with recurrent effusions.Similar benefits transpired in the use of intrapleural chemotherapy that yielded complete or partial response in about 68 percent of smokers. Particularly, however, they were prominent in those with smaller effusions or less malignant tumour lines. Recent trials have demonstrated that the protocols of hyperthermic intrapleural chemotherapy, which uses cisplatin with cytarabine, can reach a greater than 80 percent intrathoracic control, which compares and solidifies previous research in this field to the findings detailed here [12].By contrast, patients undergoing thoracostomy only had early reaccumulation of almost 25%, a trend that echoes the reservations voiced in recent review articles about the inadequacy of drainage without any additional treatment [13]. Longstanding effusion has been linked to reduced functional status and high levels of readmission to the hospital-problems that might be offset by more aggressive treatments.The current research is insightful in the form it demonstrates that pleuroperitoneal shunting (PPS) and intra-pleural chemotherapy, used in tandem with multidisciplinary surveillance, may be carried out without significant complications. No serious after-sequels as pneumothorax or infection, which have been described in previous clinical cohorts, were reported. Less serious side effects, such as transient, low-grade fevers associated with intrapleural chemotherapy instillation and minor post-surgical discomfort, were manageable conditions and did not cause the termination of treatment. Taken together, these observations suggest that PPS and localised chemotherapy may be performed with minimal risk of a procedure in the event of strict clinical supervision.Also important is the fact that the study found supportive care, especially supplementation of B-vitamins and daily analgesics, greatly determines both patient adherence as well as patient comfort. Although much less can be said about these interventions, there are numerous clinical reports that recommend introducing them into palliative protocols in a consistent manner. These supportive measures have proven to be effective, as the high rate of treatment compliance has been observed, and their role in the process of comprehensive palliative pathways cannot be ignored.The study has limitations, notwithstanding its favourable results; the follow-up is fairly short, which does not allow us to assess the recurrence rates of distant events or the survival. Prolongation of observation interval and inclusion of quality-of-life scores should thus be continued in future studies and are of great importance in palliative contexts. Moreover, tumor subtype, or rather molecular features, stratification of patients might be used to further specify treatment decisions. Case in point, effusions associated with actionable mutations can gain more benefit through targeted agents accompanied by drainage plans.Cost and resource availability come in as well. Despite the definite clinical benefits associated with PPS, this technology requires surgical resources and continuous catheter care. Intrapleural chemo-therapy, especially hyperthermic therapy, is a procedure that involves the aid of special personnel and scrupulous sterilisation methods. Though outcomes in both modalities have the potential to be favourable, the settings where resources are scarce will have to strike a balance between the demands and consequences by considering the advantages. According to the latest health economic studies, such initial investment into these methods can be retrieved in the form of fewer hospital readmissions and greater control over the symptoms.These findings support a multi-modal, patient-centred, personalised approach to the management of malignant pleural effusions (MPE), i.e. protocolized selection of shunting or intrapleural chemotherapy based on clinical and patient-preference factors, and are similar to that of supportive care. This is a less invasive method that places emphasis on symptomatic palliative care, is consistent with the global and regional best-practice guidelines, and avoids invasive procedures. It implies that their strict validation requires carrying out prospective, randomised controlled trials with prolonged follow-ups and multifaceted quality-of-life scales, the results of which will shape the process of optimal care delivery to the victims of MPE more deeply.
5. Conclusions
- The investigation report presents an evidence-based evaluation of treatment options for malignant pleural effusion (MPE). The evidence on the importance of a personalised medical, multispecialist approach is backed by data. Among the three treatment methods achieved, namely, pleuroperitoneal shunting, intrapleural chemotherapy, and thoracostomy drainage, the longest-term symptom control and the lowest recurrence rates were obtained in the shunt method during the observation period. It excluded the repeated invasive revisions, and constant drainage was provided, which was a particular benefit to the patients with repeated effusions or increased accumulation. Intrapulmonary chemotherapy was also very effective, especially when corticosteroids are added and after controlled fluid aspiration. In certain cases, it was less lasting compared to shunting, but it generally obtained partial responses and was tolerated well throughout the cohort. In contrast, thoracostomy alone performed inferiorly, and the long-term benefit was minimal, with increased reaccumulation of fluid and increased need for additional procedures. The impossibility of critical incidents in any of the groups emphasises the safety of any of the methods in case of their competent multidisciplinary supervision. In addition, supportive care (such as vitamin supplements, analgesics, nutrition optimisation) was found critical to improving compliance and patient comfort. To sum up, the multimodal approach to MPE with patient-centred orientation is advised. Surgically and pharmacologically directed interventions may also provide clinically significant relief and stability when appropriately chosen. Future studies must focus on more long-term results, incorporate quality-of-life discussions, and assess potentially molecularly guided approaches to enhance the effectiveness and precision of care among that at-risk population.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML